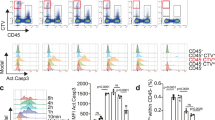
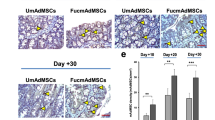
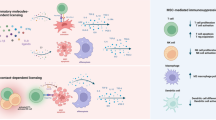
Abstract
Human multipotent mesenchymal stromal cells (MSCs) exhibit multilineage differentiation potential, support hematopoiesis, and inhibit proliferation and effector function of various immune cells. On the basis of these properties, MSC are currently under clinical investigation in a range of therapeutic applications including tissue repair and immune-mediated disorders such as graft-versus-host-disease refractory to pharmacological immunosuppression. Although initial clinical results appear promising, there are significant concerns that application of MSC might inadvertently suppress antimicrobial immunity with an increased risk of infection. We demonstrate here that on stimulation with inflammatory cytokines human MSC exhibit broad-spectrum antimicrobial effector function directed against a range of clinically relevant bacteria, protozoal parasites and viruses. Moreover, we identify the tryptophan catabolizing enzyme indoleamine 2,3-dioxygenase (IDO) as the underlying molecular mechanism. We furthermore delineate significant differences between human and murine MSC in that murine MSC fail to express IDO and inhibit bacterial growth. Conversely, only murine but not human MSC express inducible nitric oxide synthase on cytokine stimulation thus challenging the validity of murine in vivo models for the preclinical evaluation of human MSC. Collectively, our data identify human MSC as a cellular immunosuppressant that concurrently exhibits potent antimicrobial effector function thus encouraging their further evaluation in clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284: 143–147.
Meisel R, Zibert A, Laryea M, Gobel U, Daubener W, Dilloo D . Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood 2004; 103: 4619–4621.
Spaggiari GM, Capobianco A, Abdelrazik H, Becchetti F, Mingari MC, Moretta L . Mesenchymal stem cells inhibit natural killer-cell proliferation, cytotoxicity, and cytokine production: role of indoleamine 2,3-dioxygenase and prostaglandin E2. Blood 2008; 111: 1327–1333.
Ball LM, Bernardo ME, Roelofs H, Lankester A, Cometa A, Egeler RM et al. Cotransplantation of ex vivo expanded mesenchymal stem cells accelerates lymphocyte recovery and may reduce the risk of graft failure in haploidentical hematopoietic stem-cell transplantation. Blood 2007; 110: 2764–2767.
Wolf D, Wolf AM . Mesenchymal stem cells as cellular immunosuppressants. Lancet 2008; 371: 1553–1554.
Nauta AJ, Fibbe WE . Immunomodulatory properties of mesenchymal stromal cells. Blood 2007; 110: 3499–3506.
Kang HS, Habib M, Chan J, Abavana C, Potian JA, Ponzio NM et al. A paradoxical role for IFN-gamma in the immune properties of mesenchymal stem cells during viral challenge. Exp Hematol 2005; 33: 796–803.
Sundin M, Orvell C, Rasmusson I, Sundberg B, Ringden O, Le Blanc K . Mesenchymal stem cells are susceptible to human herpesviruses, but viral DNA cannot be detected in the healthy seropositive individual. Bone Marrow Transplant 2006; 37: 1051–1059.
Deak E, Ruster B, Keller L, Eckert K, Fichtner I, Seifried E et al. Suspension medium influences interaction of mesenchymal stromal cells with endothelium and pulmonary toxicity after transplantation in mice. Cytotherapy 2010; 12: 260–264.
Tropel P, Noel D, Platet N, Legrand P, Benabid AL, Berger F . Isolation and characterisation of mesenchymal stem cells from adult mouse bone marrow. Exp Cell Res 2004; 295: 395–406.
Daubener W, Wanagat N, Pilz K, Seghrouchni S, Fischer HG, Hadding U . A new, simple, bioassay for human IFN-gamma. J Immunol Methods 1994; 168: 39–47.
Ding AH, Nathan CF, Stuehr DJ . Release of reactive nitrogen intermediates and reactive oxygen intermediates from mouse peritoneal macrophages. Comparison of activating cytokines and evidence for independent production. J Immunol 1988; 141: 2407–2412.
Schroten H, Spors B, Hucke C, Stins M, Kim KS, Adam R et al. Potential role of human brain microvascular endothelial cells in the pathogenesis of brain abscess: inhibition of Staphylococcus aureus by activation of indoleamine 2,3-dioxygenase. Neuropediatrics 2001; 32: 206–210.
Daubener W, Spors B, Hucke C, Adam R, Stins M, Kim KS et al. Restriction of Toxoplasma gondii growth in human brain microvascular endothelial cells by activation of indoleamine 2,3-dioxygenase. Infect Immun 2001; 69: 6527–6531.
Adams O, Besken K, Oberdorfer C, MacKenzie CR, Takikawa O, Daubener W . Role of indoleamine-2,3-dioxygenase in alpha/beta and gamma interferon-mediated antiviral effects against herpes simplex virus infections. J Virol 2004; 78: 2632–2636.
Parsons CH, Szomju B, Kedes DH . Susceptibility of human fetal mesenchymal stem cells to Kaposi sarcoma-associated herpesvirus. Blood 2004; 104: 2736–2738.
Sato K, Ozaki K, Oh I, Meguro A, Hatanaka K, Nagai T et al. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood 2007; 109: 228–234.
Ren G, Zhang L, Zhao X, Xu G, Zhang Y, Roberts AI et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell 2008; 2: 141–150.
Muller A, Heseler K, Schmidt SK, Spekker K, Mackenzie CR, Daubener W . The missing link between indoleamine 2,3-dioxygenase mediated antibacterial and immunoregulatory effects. J Cell Mol Med 2009; 13: 1125–1135.
Aggarwal S, Pittenger MF . Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005; 105: 1815–1822.
Plumas J, Chaperot L, Richard MJ, Molens JP, Bensa JC, Favrot MC . Mesenchymal stem cells induce apoptosis of activated T cells. Leukemia 2005; 19: 1597–1604.
Krampera M, Cosmi L, Angeli R, Pasini A, Liotta F, Andreini A et al. Role for interferon-gamma in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells 2006; 24: 386–398.
Haniffa MA, Wang XN, Holtick U, Rae M, Isaacs JD, Dickinson AM et al. Adult human fibroblasts are potent immunoregulatory cells and functionally equivalent to mesenchymal stem cells. J Immunol 2007; 179: 1595–1604.
Sudres M, Norol F, Trenado A, Gregoire S, Charlotte F, Levacher B et al. Bone marrow mesenchymal stem cells suppress lymphocyte proliferation in vitro but fail to prevent graft-versus-host disease in mice. J Immunol 2006; 176: 7761–7767.
Badillo AT, Peranteau WH, Heaton TE, Quinn C, Flake AW . Murine bone marrow derived stromal progenitor cells fail to prevent or treat acute graft-versus-host disease. Br J Haematol 2008; 141: 224–234.
Prigozhina TB, Khitrin S, Elkin G, Eizik O, Morecki S, Slavin S . Mesenchymal stromal cells lose their immunosuppressive potential after allotransplantation. Exp Hematol 2008; 36: 1370–1376.
Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 2008; 371: 1579–1586.
Kaandorp CJ, Dinant HJ, van de Laar MA, Moens HJ, Prins AP, Dijkmans BA . Incidence and sources of native and prosthetic joint infection: a community based prospective survey. Ann Rheum Dis 1997; 56: 470–475.
Sia IG, Berbari EF . Infection and musculoskeletal conditions: osteomyelitis. Best Pract Res Clin Rheumatol 2006; 20: 1065–1081.
Muller I, Vaegler M, Holzwarth C, Tzaribatchev N, Pfister SM, Schutt B et al. Secretion of angiogenic proteins by human multipotent mesenchymal stromal cells and their clinical potential in the treatment of avascular osteonecrosis. Leukemia 2008; 22: 2054–2061.
Hebart H, Einsele H . Clinical aspects of CMV infection after stem cell transplantation. Hum Immunol 2004; 65: 432–436.
Acknowledgements
We thank D Klostermann for technical assistance. Moreover, we want to acknowledge M Herten (Department of Orthopedics, Heinrich-Heine-University Düsseldorf) for performing chondrogenic differentiation assays and P Verde (Coordination Center for Clinical Trials, Heinrich-Heine-University Düsseldorf) for expert statistical advice. Human CMV was kindly provided by C Sinzger (Institute for Medical Virology, University of Tübingen, Germany) and Toxoplasma gondii tachyzoites were obtained from M Saathoff and HM Seitz (Institute for Medical Parasitology, University of Bonn, Germany). This work was supported by German Federal Ministry of Education and Research (BMBF) Grants nos. 01GN0990 (RM, DD), 01GM0873 (RM, DD), 01GN0951 (RS, WD), 01GN0952 (RH) and 01GN0949 (JS), German Research Council (DFG) Grant no. RU729 (WD) and the ‘Elterninitiative Kinderkrebsklinik Düsseldorf e.V.’ (RM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Author contributions
RM and WD designed research; RM, SB, KH, ÖD, HB, CW, SS, WS, MJ, RS, RH, JS, DD and WD performed research; RM, RS and WD analyzed data; and RM, DD and WD wrote the paper.
Supplementary Information accompanies the paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Meisel, R., Brockers, S., Heseler, K. et al. Human but not murine multipotent mesenchymal stromal cells exhibit broad-spectrum antimicrobial effector function mediated by indoleamine 2,3-dioxygenase. Leukemia 25, 648–654 (2011). https://doi.org/10.1038/leu.2010.310
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2010.310
Keywords
This article is cited by
-
Cancer and COVID-19: unravelling the immunological interplay with a review of promising therapies against severe SARS-CoV-2 for cancer patients
Journal of Hematology & Oncology (2023)
-
Mesenchymal stem cells in the treatment of osteogenesis imperfecta
Cell Regeneration (2023)
-
Therapeutic potential of mesenchymal stem/stromal cells (MSCs)-based cell therapy for inflammatory bowel diseases (IBD) therapy
European Journal of Medical Research (2023)
-
Canine umbilical cord tissue derived mesenchymal stem cells naturally express mRNAs of some antimicrobial peptides
Veterinary Research Communications (2023)
-
Can mesenchymal stem/stromal cells and their secretomes combat bacterial persisters?
World Journal of Microbiology and Biotechnology (2023)