Abstract
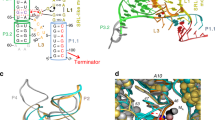
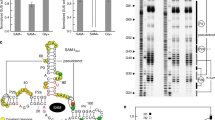
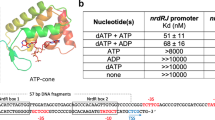
The biosynthesis of several protein cofactors is subject to feedback regulation by riboswitches1,2,3. Flavin mononucleotide (FMN)-specific riboswitches4,5, also known as RFN elements6, direct expression of bacterial genes involved in the biosynthesis and transport of riboflavin (vitamin B2) and related compounds. Here we present the crystal structures of the Fusobacterium nucleatum riboswitch bound to FMN, riboflavin and antibiotic roseoflavin7. The FMN riboswitch structure, centred on an FMN-bound six-stem junction, does not fold by collinear stacking of adjacent helices, typical for folding of large RNAs. Rather, it adopts a butterfly-like scaffold, stapled together by opposingly directed but nearly identically folded peripheral domains. FMN is positioned asymmetrically within the junctional site and is specifically bound to RNA through interactions with the isoalloxazine ring chromophore and direct and Mg2+-mediated contacts with the phosphate moiety. Our structural data, complemented by binding and footprinting experiments, imply a largely pre-folded tertiary RNA architecture and FMN recognition mediated by conformational transitions within the junctional binding pocket. The inherent plasticity of the FMN-binding pocket and the availability of large openings make the riboswitch an attractive target for structure-based design of FMN-like antimicrobial compounds. Our studies also explain the effects of spontaneous and antibiotic-induced deregulatory mutations and provided molecular insights into FMN-based control of gene expression in normal and riboflavin-overproducing bacterial strains.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Protein Data Bank
Data deposits
The coordinates of the X-ray structures of the FMN riboswitch are deposited in the RCSB Protein Data Bank under the following accession numbers: bound to FMN, transcribed RNA, 3F2Q; bound to FMN, two-strand composition, 3F4E; bound to riboflavin, 3F4G; bound to roseoflavin, 3F4H; [Ir(NH3)6]3+-soaked, 3F2T; Cs+-soaked 3F2X; Ba2+-soaked, 3F2W; Mn2+-soaked, 3F2Y; [Co(NH3)6]3+-soaked, 3F30.
References
Nudler, E. & Mironov, A. S. The riboswitch control of bacterial metabolism. Trends Biochem. Sci. 29, 11–17 (2004)
Serganov, A. & Patel, D. J. Ribozymes, riboswitches and beyond: regulation of gene expression without proteins. Nature Rev. Genet. 8, 776–790 (2007)
Winkler, W. C. & Breaker, R. R. Regulation of bacterial gene expression by riboswitches. Annu. Rev. Microbiol. 59, 487–517 (2005)
Mironov, A. S. et al. Sensing small molecules by nascent RNA: a mechanism to control transcription in bacteria. Cell 111, 747–756 (2002)
Winkler, W. C., Cohen-Chalamish, S. & Breaker, R. R. An mRNA structure that controls gene expression by binding FMN. Proc. Natl Acad. Sci. USA 99, 15908–15913 (2002)
Gelfand, M. S., Mironov, A. A., Jomantas, J., Kozlov, Y. I. & Perumov, D. A. A conserved RNA structure element involved in the regulation of bacterial riboflavin synthesis genes. Trends Genet. 15, 439–442 (1999)
Blount, K. F. & Breaker, R. R. Riboswitches as antibacterial drug targets. Nature Biotechnol. 24, 1558–1564 (2006)
Barrick, J. E. & Breaker, R. R. The distributions, mechanisms, and structures of metabolite-binding riboswitches. Genome Biol. 8, R239 (2007)
Serganov, A., Polonskaia, A., Phan, A. T., Breaker, R. R. & Patel, D. J. Structural basis for gene regulation by a thiamine pyrophosphate-sensing riboswitch. Nature 441, 1167–1171 (2006)
Thore, S., Leibundgut, M. & Ban, N. Structure of the eukaryotic thiamine pyrophosphate riboswitch with its regulatory ligand. Science 312, 1208–1211 (2006)
Edwards, T. E. & Ferre-D’Amare, A. R. Crystal structures of the thi-box riboswitch bound to thiamine pyrophosphate analogs reveal adaptive RNA-small molecule recognition. Structure 14, 1459–1468 (2006)
Gilbert, S. D., Rambo, R. P., Van Tyne, D. & Batey, R. T. Structure of the SAM-II riboswitch bound to S-adenosylmethionine. Nature Struct. Mol. Biol. 15, 177–182 (2008)
Montange, R. K. & Batey, R. T. Structure of the S-adenosylmethionine riboswitch regulatory mRNA element. Nature 441, 1172–1175 (2006)
Nahvi, A. et al. Genetic control by a metabolite binding mRNA. Chem. Biol. 9, 1043–1049 (2002)
Burgess, C. M., Smid, E. J., Rutten, G. & van Sinderen, D. A general method for selection of riboflavin-overproducing food grade micro-organisms. Microb. Cell Fact. 5, 24 (2006)
Kapatral, V. et al. Genome sequence and analysis of the oral bacterium Fusobacterium nucleatum strain ATCC 25586. J. Bacteriol. 184, 2005–2018 (2002)
Vitreschak, A. G., Rodionov, D. A., Mironov, A. A. & Gelfand, M. S. Regulation of riboflavin biosynthesis and transport genes in bacteria by transcriptional and translational attenuation. Nucleic Acids Res. 30, 3141–3151 (2002)
Agmon, I., Bashan, A., Zarivach, R. & Yonath, A. Symmetry at the active site of the ribosome: structural and functional implications. Biol. Chem. 386, 833–844 (2005)
Jaeger, L., Verzemnieks, E. J. & Geary, C. The UA_handle: a versatile submotif in stable RNA architectures. Nucleic Acids Res 10.1093/nar/gkn911 (28 November 2008)
Lescoute, A. & Westhof, E. The interaction networks of structured RNAs. Nucleic Acids Res. 34, 6587–6604 (2006)
Krasilnikov, A. S. & Mondragon, A. On the occurrence of the T-loop RNA folding motif in large RNA molecules. RNA 9, 640–643 (2008)
Nagaswamy, U. & Fox, G. E. Frequent occurrence of the T-loop RNA folding motif in ribosomal RNAs. RNA 8, 1112–1119 (2002)
Selmer, M. et al. Structure of the 70S ribosome complexed with mRNA and tRNA. Science 313, 1935–1942 (2006)
Batey, R. T., Gilbert, S. D. & Montange, R. K. Structure of a natural guanine-responsive riboswitch complexed with the metabolite hypoxanthine. Nature 432, 411–415 (2004)
Serganov, A. et al. Structural basis for discriminative regulation of gene expression by adenine- and guanine-sensing mRNAs. Chem. Biol. 11, 1729–1741 (2004)
Garst, A. D., Heroux, A., Rambo, R. P. & Batey, R. T. Crystal structure of the lysine riboswitch regulatory mRNA element. J. Biol. Chem. 283, 22347–22351 (2008)
Serganov, A., Huang, L. & Patel, D. J. Structural insights into amino acid binding and gene control by a lysine riboswitch. Nature 455, 1263–1267 (2008)
Fan, P., Suri, A. K., Fiala, R., Live, D. & Patel, D. J. Molecular recognition in the FMN-RNA aptamer complex. J. Mol. Biol. 258, 480–500 (1996)
Burgess, C., O’Connell-Motherway, M., Sybesma, W., Hugenholtz, J. & van Sinderen, D. Riboflavin production in Lactococcus lactis: potential for in situ production of vitamin-enriched foods. Appl. Environ. Microbiol. 70, 5769–5777 (2004)
Wickiser, J. K., Winkler, W. C., Breaker, R. & Crothers, D. M. The speed of RNA transcription and metabolite binding kinetics operate an FMN riboswitch. Mol. Cell 18, 49–60 (2005)
Serganov, A. et al. Ribosomal protein S15 from Thermus thermophilus: cloning, sequencing, overexpression of the gene and RNA-binding properties of the protein. Eur. J. Biochem. 246, 291–300 (1997)
de La Fortelle, E. & Bricogne, G. in Methods in Enzymology 472–494 (Academic Press, 1997)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997)
Feig, A. L. & Uhlenbeck, O. C. in The RNA World 2nd edn (eds Gesteland, R. F., Cech, T. R. & Atkins, J. F.) 287–319 (Cold Spring Harbor Laboratory Press, 1999)
Serganov, A., Polonskaia, A., Ehresmann, B., Ehresmann, C. & Patel, D. J. Ribosomal protein S15 represses its own translation via adaptation of an rRNA-like fold within its mRNA. EMBO J. 8, 1898–1908 (2003)
Acknowledgements
We thank the personnel of beamline X29 at the Brookhaven National Laboratory and beamline 24-ID-C at the Advanced Photon Source, Argonne National Laboratory, funded by the US Department of Energy. We thank L. Jaeger (University of California, Santa Barbara) for discussions and O. Ouerfelli (Memorial Sloan-Kettering Cancer Center, New York) for the synthesis of iridium hexamine. D.J.P. was supported by funds from the National Institutes of Health.
Author Contributions L.H. crystallized the F. nucleatum FMN riboswitch and performed binding experiments, A.S. determined the structures and performed footprinting experiments, and A.S. and D.J.P. wrote the manuscript with the assistance of L.H. All authors discussed the results and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-19 with Legends, Supplementary Tables 1-2 and Supplementary References. This file was replaced on February 4th, 2009 to correct 2 chemical formulae. (PDF 9097 kb)
Rights and permissions
About this article
Cite this article
Serganov, A., Huang, L. & Patel, D. Coenzyme recognition and gene regulation by a flavin mononucleotide riboswitch. Nature 458, 233–237 (2009). https://doi.org/10.1038/nature07642
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07642
This article is cited by
-
An RNA aptamer that shifts the reduction potential of metabolic cofactors
Nature Chemical Biology (2022)
-
Targeting RNA structures with small molecules
Nature Reviews Drug Discovery (2022)
-
Riboswitch-mediated regulation of riboflavin biosynthesis genes in prokaryotes
3 Biotech (2022)
-
RPocket: an intuitive database of RNA pocket topology information with RNA-ligand data resources
BMC Bioinformatics (2021)
-
Accelerated cryo-EM-guided determination of three-dimensional RNA-only structures
Nature Methods (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.