Abstract
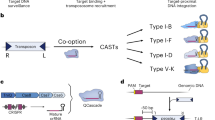
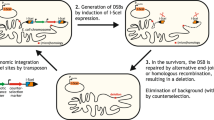
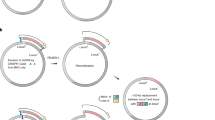
An increasing number of microbial genomes have been completely sequenced, and functional analyses of these genomic sequences are under way. To facilitate these analyses, we have developed a genome-engineering tool for determining essential genes and minimizing bacterial genomes. We made two large pools of independent transposon mutants in Escherichia coli using modified Tn5 transposons with two different selection markers and precisely mapped the chromosomal location of 800 of these transposons. By combining a mapped transposon mutation from each of the mutant pools into the same chromosome using phage P1 transduction and then excising the flanked genomic segment by Cre-mediated loxP recombination, we obtained E. coli strains in which large genomic fragments (59–117 kilobases) were deleted. Some of these individual deletions were then combined into a single “cumulative deletion strain” that lacked 287 open reading frames (313.1 kilobases) but that nevertheless exhibited normal growth under standard laboratory conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Schmid, M.B., Kapur, N., Isaacson, D.R., Lindroos, P. & Sharpe, C. Genetic analysis of temperature-sensitive lethal mutants of Salmonella typhimurium. Genetics 123, 625–663 (1989).
Arigoni, F. et al. A genome-based approach for the identification of essential bacterial genes. Nat. Biotechnol. 16, 851–856 (1998).
Mushegian, A.R. & Koonin, E.V. A minimal gene set for cellular life derived by comparison of complete bacterial genomes. Proc. Natl. Acad. Sci. USA 93, 10268–10273 (1996).
Winzeler, E.A. et al. Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285, 901–906 (1999).
Itaya, M. An estimation of minimal genome size required for life. FEBS Lett. 362, 257–260 (1995).
Akerley, B.J. et al. A genome-scale analysis for identification of genes required for growth or survival of Haemophilus influenzae. Proc. Natl. Acad. Sci. USA 99, 966–971 (2002).
Blattner, F.R. et al. The complete genome sequence of Escherichia coli K-12. Science 277, 1453–1474 (1997).
Tao, H., Bausch, C., Richmond, C., Blattner, F.R. & Conway, T. Functional genomics: expression analysis of Escherichia coli growing on minimal and rich media. J. Bacteriol. 181, 6425–6440 (1999).
Koob, M.D., Shaw, A.J. & Cameron, D.C. Minimizing the genome of Escherichia coli. Ann. N. Y. Acad. Sci. 745, 1–3 (1994).
Yoon, Y.G., Cho, J.H. & Kim, S.C. Cre/loxP-mediated excision and amplification of the Escherichia coli genome. Genet. Anal. 14, 89–95 (1998).
Datsenko, K.A. & Wanner, B.L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 97, 6640–6645 (2000).
Posfai, G. et al. In vivo excision and amplification of large segments of the Escherichia coli genome. Nucleic Acids Res. 22, 2392–2398 (1994).
Hasan, N., Koob, M. & Szybalski, W. Escherichia coli genome targeting, I. Cre-lox-mediated in vitro generation of ori- plasmids and their in vivo chromosomal integration and retrieval. Gene 150, 51–56 (1994).
Kolisnychenko, V. et al. Engineering a reduced Escherichia coli genome. Genome Res. 12, 640–647 (2002).
Szybalski, W. From the double-helix to novel approaches to the sequencing of large genomes. Gene 135, 279–290 (1993).
Pratt, L.A. & Kolter, R. Genetic analysis of Esherichia coli biofilm formation: roles of flagella, motility, chemotaxis and type I pili. Mol. Microbiol. 30, 285–293 (1998).
Lawrence, J.G. & Ochman, H. Molecular archaeology of the Escherichia coli genome. Proc. Natl. Acad. Sci. USA 95, 9413–9417 (1998).
Armstrong, K.A. A 37 × 103 molecular weight plasmid-encoded protein is required for replication and copy number control in the plasmid pSC101 and its temperature-sensitive derivatives pHS1. J. Mol. Biol. 175, 331–348 (1984).
Thompson, J.R., Register, E., Curotto, J., Kurtz, M. & Kelly, R. An improved protocol for the preparation of yeast cells for transformation by electroporation. Yeast 14, 565–571 (1998).
Grant, S.G., Jessee, J., Bloom, F.R. & Hanahan, D. Differential plasmid rescue from transgenic mouse DNAs into Escherichia coli methylation-restriction mutants. Proc. Natl. Acad. Sci. USA 87, 4645–4649 (1990).
Goryshin, I.Y., Jendrisak, J., Hoffman, L.M., Meis, R. & Reznikoff, W.S. Insertional transposon mutagenesis by electroporation of released Tn5 transposition complexes. Nat. Biotechnol. 18, 97–100 (2000).
Sambrook, J. & Russell, D.W. Molecular Cloning: A Laboratory Manual, Edn. 3 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2001).
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. & Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Miller, J.H. A Short Course in Bacterial Genetics: A Laboratory Manual and Handbook for Escherichia coli and Related Bacteria (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1992).
Acknowledgements
This work was supported in part by grants from the National Research Laboratory Program (2000-N-NL-01-C-160) and the Molecular Medicine Research Program (00-J-MM-01-B-01) from the Korean Ministry of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Yu, B., Sung, B., Koob, M. et al. Minimization of the Escherichia coli genome using a Tn5-targeted Cre/loxP excision system. Nat Biotechnol 20, 1018–1023 (2002). https://doi.org/10.1038/nbt740
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt740
This article is cited by
-
Trimming the genomic fat: minimising and re-functionalising genomes using synthetic biology
Nature Communications (2023)
-
Advances and prospects in metabolic engineering of Escherichia coli for L-tryptophan production
World Journal of Microbiology and Biotechnology (2022)
-
Insights into the structure of Escherichia coli outer membrane as the target for engineering microbial cell factories
Microbial Cell Factories (2021)
-
CRISPR RNA-guided integrases for high-efficiency, multiplexed bacterial genome engineering
Nature Biotechnology (2021)
-
Exploring the fitness benefits of genome reduction in Escherichia coli by a selection-driven approach
Scientific Reports (2020)