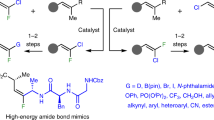
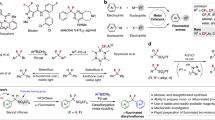
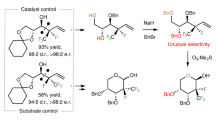
Abstract
Despite the growing importance of organofluorines as pharmaceuticals and agrochemicals, the stereoselective introduction of fluorine into many prominent classes of natural products and chemotherapeutic agents is difficult. One long-standing unsolved challenge is the enantioselective aldol reaction of fluoroacetate to enable access to fluorinated analogues of medicinally relevant acetate-derived compounds, such as polyketides and statins. Herein we present fluoromalonic acid halfthioesters as biomimetic surrogates of fluoroacetate and demonstrate their use in highly stereoselective aldol reactions that proceed under mild organocatalytic conditions. We also show that the methodology can be extended to formal aldol reactions with fluoroacetaldehyde and consecutive aldol reactions. The synthetic utility of the fluorinated aldol products is illustrated by the synthesis of a fluorinated derivative of the top-selling drug atorvastatin. The results show the prospects of the method for the enantioselective introduction of fluoroacetate to access a wide variety of highly functionalized fluorinated compounds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ojima, I. Fluorine in Medicinal Chemistry and Chemical Biology (Blackwell Publishing Ltd, 2009).
Müller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: looking beyond intuition. Science 317, 1881–1886 (2007).
Purser, S., Moore, P. R., Swallow, S. & Gouverneur, V. Fluorine in medicinal chemistry. Chem. Soc. Rev. 37, 320–330 (2008).
O'Hagan, D. Understanding organofluorine chemistry. An introduction to the C–F bond. Chem. Soc. Rev. 37, 308–319 (2008).
Wang, J. et al. Fluorine in pharmaceutical industry: fluorine-containing drugs introduced to the market in the last decade (2001–2011). Chem. Rev. 114, 2432–2506 (2013).
Fried, J. & Sabo, E. F. 9α-Fluoro derivatives of cortisone and hydrocortisone. J. Am. Chem. Soc. 76, 1455–1456 (1954).
Walker, M. C. & Chang, M. C. Y. Natural and engineered biosynthesis of fluorinated natural products. Chem. Soc. Rev. 43, 6527–6536 (2014).
Furuya, T., Kamlet, A. S. & Ritter, T. Catalysis for fluorination and trifluoromethylation. Nature 473, 470–477 (2011).
Yang, X., Wu, T., Phipps, R. J. & Toste, F. D. Advances in catalytic enantioselective fluorination, mono-, di-, and trifluoromethylation, and trifluoromethylthiolation reactions. Chem. Rev. 115, 826–870 (2015).
Khosla, C., Herschlag, D., Cane, D. E. & Walsh, C. T. Assembly line polyketide synthases: mechanistic insights and unsolved problems. Biochemistry 53, 2875–2883 (2014).
Mahrwald, R. Modern Methods in Stereoselective Aldol Reactions (Wiley-VCH Verlag GmbH & Co., 2013).
Welch, J. T., Seper, K., Eswarakrishnan, S. & Samartino, J. Preparation of α-fluoro enolates and their use in the directed aldol reaction. J. Org. Chem. 49, 4720–4721 (1984).
Welch, J. T. & Plummer, J. S. The stereoselective aldol condensation of α-fluoroacetates. Synth. Comm. 19, 1081–1090 (1989).
Linderman, R. J. & Graves, D. M. Oxidation of fluoroalkyl-substituted carbinols by the Dess–Martin reagent. J. Org. Chem. 54, 661–668 (1989).
Huang, X.-T. & Chen, Q.-Y. Ethyl α-fluoro silyl enol ether: stereoselective synthesis and its aldol reaction with aldehydes and ketones. J. Org. Chem. 67, 3231–3234 (2002).
Saadi, J., Akakura, M. & Yamamoto, H. Rapid, one-pot synthesis of β-siloxy-α-haloaldehydes. J. Am. Chem. Soc. 133, 14248–14251 (2011).
Walker, M. C. et al. Expanding the fluorine chemistry of living systems using engineered polyketide synthase pathways. Science 341, 1089–1094 (2013).
Orlandi, S., Benaglia, M. & Cozzi, F. Cu(II)-catalyzed enantioselective aldol condensation between malonic acid hemithioesters and aldehydes. Tetrahedron Lett. 45, 1747–1749 (2004).
Magdziak, D. et al. Catalytic enantioselective thioester aldol reactions that are compatible with protic functional groups. J. Am. Chem. Soc. 127, 7284–7285 (2005).
Ricci, A. et al. Organocatalytic enantioselective decarboxylative addition of malonic half thioesters to imines. Adv. Synth. Catal. 349, 1037–1040 (2007).
Lubkoll, J. & Wennemers, H. Mimicry of polyketide synthases—enantioselective 1,4-addition reactions of malonic acid half-thioesters to nitroolefins. Angew. Chem. Int. Ed. 46, 6841–6844 (2007).
Bae, H. Y., Sim, J. H., Lee, J.-W., List, B. & Song, C. E. Organocatalytic enantioselective decarboxylative aldol reaction of malonic acid half thioesters with aldehydes. Angew. Chem. Int. Ed. 52, 12143–12147 (2013).
Nakamura, S., Sano, M., Toda, A., Nakane, D. & Masuda, H. Organocatalytic enantioselective decarboxylative reaction of malonic acid half thioesters with cyclic N-sulfonyl ketimines by using N-heteroarenesulfonyl cinchona alkaloid amides. Chem. Eur. J. 21, 3929–3932 (2015).
Furutachi, M., Mouri, S., Matsunaga, S. & Shibasaki, M. A heterobimetallic Ni/La-salan complex for catalytic asymmetric decarboxylative 1,4-addition of malonic acid half-thioester. Chem. Asian J. 5, 2351–2354 (2010).
Pan, Y. et al. Expanding the utility of Brønsted base catalysis: biomimetic enantioselective decarboxylative reactions. Chem. Eur. J. 17, 8363–8370 (2011).
Rigo, B., Fasseur, D., Cauliez, P. & Couturier, D. Reaction of trimethylsilyl derivatives with Meldrum's acid: a new and easy monofunctionalization of malonic acid. Tetrahedron Lett. 30, 3073–3076 (1989).
Pohl, N. L., Gokhale, R. S., Cane, D. E. & Khosla, C. Synthesis and incorporation of an N-acetylcysteamine analogue of methylmalonyl–CoA by a modular polyketide synthase. J. Am. Chem. Soc. 120, 11206–11207 (1998).
Arakawa, Y., Fritz, S. P. & Wennemers, H. Organocatalytic stereoselective synthesis of acyclic γ-nitrothioesters with all-carbon quaternary stereogenic centers. J. Org. Chem. 79, 3937–3945 (2014).
Bahlinger, A., Fritz, S. P. & Wennemers, H. Stereoselective metal-free synthesis of β-amino thioesters with tertiary and quaternary stereogenic centers. Angew. Chem. Int. Ed. 53, 8779–8783 (2014).
Brak, K. & Jacobsen, E. N. Asymmetric ion-pairing catalysis. Angew. Chem. Int. Ed. 52, 534–561 (2013).
Bordwell, F. G. Equilibrium acidities in dimethyl sulfoxide solution. Acc. Chem. Res. 21, 456–463 (1988).
Krautwald, S., Sarlah, D., Schafroth, M. A. & Carreira, E. M. Enantio- and diastereodivergent dual catalysis: α-allylation of branched aldehydes. Science 340, 1065–1068 (2013).
Kitagaki, J., Shi, G., Miyauchi, S., Murakami, S. & Yang, Y. Cyclic depsipeptides as potential cancer therapeutics. Anti-Cancer Drugs 26, 259–271 (2015).
Oláh, G. & Kuhn, S. Darstellung und Untersuchung Organischer Fluorverbindungen XXI. Darstellung von Fluoracetaldehyd und Aliphatischen Fluormethylketonen. Chem. Ber. 89, 864–865 (1956).
Tabernero, L., Bochar, D. A., Rodwell, V. W. & Stauffacher, C. V. Substrate-induced closure of the flap domain in the ternary complex structures provides insights into the mechanism of catalysis by 3-hydroxy-3-methylglutaryl–CoA reductase. Proc. Natl Acad. Sci. USA 96, 7167–7171 (1999).
Wang, Z. et al. Molecular modeling studies of atorvastatin analogues as HMGR inhibitors using 3D-QSAR, molecular docking and molecular dynamics simulations. Bioorg. Med. Chem. Lett. 24, 3869–3876 (2014).
Acknowledgements
We are grateful to the Swiss National Science Foundation for financial support of this work. We thank N. Trapp and the SMoCC facility of D-CHAB at ETH Zurich for recording the X-ray crystal structure. We dedicate this article to Professor Albert Eschenmoser on the occasion of his 90th birthday.
Author information
Authors and Affiliations
Contributions
J.S. conducted the experiments. J.S. and H.W. conceived and designed the project, analysed the data and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 6459 kb)
Rights and permissions
About this article
Cite this article
Saadi, J., Wennemers, H. Enantioselective aldol reactions with masked fluoroacetates. Nature Chem 8, 276–280 (2016). https://doi.org/10.1038/nchem.2437
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2437
This article is cited by
-
Catalytic length-controlled oligomerization with synthetic programmable templates
Nature Synthesis (2023)
-
Chemoenzymatic synthesis of fluorinated polyketides
Nature Chemistry (2022)
-
Enzymatic synthesis of fluorinated compounds
Applied Microbiology and Biotechnology (2021)
-
α-Fluorination of carbonyls with nucleophilic fluorine
Nature Chemistry (2019)
-
Enantioselective decarboxylative chlorination of β-ketocarboxylic acids
Nature Communications (2017)