Abstract
Hexagonal boron nitride (h-BN) has attracted significant attention because of its superior properties as well as its potential as an ideal dielectric layer for graphene-based devices. The h-BN films obtained via chemical vapour deposition in earlier reports are always polycrystalline with small grains because of high nucleation density on substrates. Here we report the successful synthesis of large single-crystal h-BN grains on rational designed Cu–Ni alloy foils. It is found that the nucleation density can be greatly reduced to 60 per mm2 by optimizing Ni ratio in substrates. The strategy enables the growth of single-crystal h-BN grains up to 7,500 μm2, approximately two orders larger than that in previous reports. This work not only provides valuable information for understanding h-BN nucleation and growth mechanisms, but also gives an effective alternative to exfoliated h-BN as a high-quality dielectric layer for large-scale nanoelectronic applications.
Similar content being viewed by others
Introduction
Hexagonal boron nitride (h-BN) is a representative two-dimensional (2D) crystal with a band gap of ~6 eV (ref. 1). Owing to the strong covalent sp2 bonds in the plane, it has very high mechanical strength, thermal conductivity and chemical stability2,3,4,5,6. The h-BN layer with atomically smooth surface free of dangling bonds/charge traps makes it an ideal dielectric substrate for field effect transistor7 and spacer for novel tunnelling devices8,9. The characters of h-BN make it possible to realize ballistic electronics at room temperature10,11. Normally, uniform crystalline h-BN flakes are mostly obtained by mechanical exfoliation from h-BN single crystals7,11,12,13,14,15. However, the flakes are obtained through a highly skilled manual process—mechanical exfoliation and transferring, which is not a scalable method for practical applications. So far, many efforts have been taken on various substrates such as Ni16,17,18,19,20,21, Cu3,21,22,23,24,25,26,27, Pt28,29, Ru30,31 and Co32 to obtain large h-BN crystals via the chemical vapour deposition (CVD) process. However, those grains in h-BN films are very small (usually <50 μm2) because of high nucleation density at the early growth stages10,11,15,16,17,19,22. The small grains lead to high density of grain boundaries and dangling bonds, which are known as structural defects in h-BN33,34. The defects definitely give rise to higher surface roughness/charge impurities, and then degrade the performance of electronic devices35. Therefore, the growth of high-quality, large h-BN crystals is essential in the commercial-level manufacture for its further application in electronic, photonic and mechanical devices.
Here we report the successful synthesis of large-size single-crystal h-BN grains on rationally designed binary Cu–Ni alloy. It is observed that under our growth conditions the nucleation density can be dramatically decreased by introducing Ni to the Cu substrate. Ten to twenty atom % of Ni can reduce the nucleation density down to 60 mm2, and h-BN grains of ~7,500 μm2 in area can be obtained. The grains are about two orders larger than those in previous reports10,11,15,16,17,19,22. The introduction of Ni is also proved to enhance the decomposition of poly-aminoborane and help synthesize high-quality h-BN grains free of nanoparticles. The characterizations such as Auger electron spectroscopy (AES), atomic force microscopy (AFM) and transmission electron microscopy (TEM) confirm that h-BN grains are uniform. Our results demonstrate that the CVD-grown h-BN layer with large grain can help effectively preserve the intrinsic properties of graphene, and will certainly facilitate the further application of wafer-scale electronics.
Results
h-BN growth
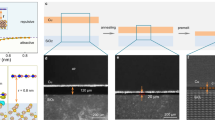
In our work, Cu–Ni alloy was used as the catalytic substrate for h-BN growth. The alloy was obtained by annealing Ni-coated Cu foils at 1,050 °C for 2 h in flowing hydrogen. Ammonia borane (H3BNH3, also called borazane) was supplied as the precursor for h-BN growth. By heating the precursor to 70–90 °C, borazane began to dissociate and the products were carried into the reaction chamber in a H2 flow at a pressure of ~50 Pa. The growth temperature was ~1,050 °C. The schematic of process flow is illustrated in Fig. 1a, and the experimental details are given in Supplementary Fig. 1.
(a) Schematic illustration showing the procedure of h-BN growth. (b–e) Typical SEM images of h-BN grains grown on Cu–Ni alloy foils with 15 atom % Ni at 1,085 °C for 10, 30, 60 and 90 min, respectively. The red arrows in b show the sites of the h-BN grains, while the inset shows the enlarged image of one as-grown h-BN grain. The scale bars in b–e are 20 μm, and in inset in b is 2 μm.
Figure 1b–e shows the scanning electron microscopy (SEM) images of h-BN grown at 1,085 °C on Cu–Ni alloy substrates with 15 atom % Ni. It can be clearly seen that the h-BN grains nucleate, grow up and finally coalesce into a continuous film. The typical lengths of side for triangle h-BN grains after 10 and 40 min of growth were 5 and 60 μm, respectively, while grains with the length of side up to 130 μm were obtained after 60 min. All the h-BN grains obtained are in triangular shape (Fig. 1b–d), which implies that these grains are all single crystals with nitrogen-terminated zigzag edges16,17,23. h-BN grains growing across alloy grain boundaries are also frequently observed, suggesting that under the conditions h-BN growth is surface-mediated23.
Experiments were carried out to understand the influence of Ni concentration on h-BN growth. Figure 2a shows a typical SEM image of h-BN grains grown on pure Cu foil for 10 min at 1,050 °C. It could be observed that the nucleation density of the h-BN grains is so high that the triangular-shaped grains with only ~10–15 μm in the length of side are about to coalesce. Figure 2b–d shows SEM images of h-BN grains grown for 60 min on Cu–Ni alloy foils with 10, 20 and 30 atom % Ni, respectively. And the relationship between the largest grain size and growth time is given in Fig. 2e, while the relationship between the number of grains and growth time is shown in Fig. 2f. It can be found that the nucleation density decreased notably after introducing Ni element. On Cu–Ni alloy substrate with 10–20 atom % Ni, the largest length of side for single-crystal h-BN grains reaches ~70–90 μm with a lateral growth rate of ~1.2 μm min−1. The nucleation density dropped further on 30 atom % Ni substrates, but the lateral growth rate dropped to ~0.3 μm min−1, with the largest length of side for grains only achieved ~30 μm. The same experiments were also taken on Ni foils and Cu–Ni alloy foils with 50 atom % Ni; however, in the present process window, h-BN grains were not observed even after 90 min of growth for both cases (see Supplementary Fig. 2).
(a) SEM images of h-BN grains grown on Cu foil for 10 min at 1,050 °C. (b–d) SEM images of h-BN grains grown for 60 min on Cu–Ni alloy foils with (b) 10 atom %, (c) 20 atom % and (d) 30 atom % Ni at 1,050 °C. The scale bars are 20 μm in a–d. (e) Dependence of the maximum h-BN grain size on Ni concentration and growth time. (f) Dependence of the number of h-BN grains on Ni concentration and growth time.
The growth mechanism of h-BN on Cu is mainly surface-mediated23,25, while on Ni, it is neither surface-limited nor dominated by segregation and precipitation of B and N, but rather depends on surface chemistry of Ni–B and Ni–N18. Our experimental parameters of h-BN growth on Cu–Ni substrate with 10–20 atom % Ni are based on the surface-mediated mechanism on Cu foils. With further increase in Ni content, new growth mechanism based on solid-gas reactions involving Ni–B and Ni–N sets in, so that the growth of h-BN would move entirely to a different process window, accounting for decreased growth rate or even inability for phase formation.
As shown in Figs 1b–d and 2e,f, it is also discovered that nucleation continued with its rate increased nonlinearly with time, while the early nuclei developed in size during growth. Similar phenomenon has also been reported for h-BN growth on Cu foils23. It is therefore clear that the key to success for large grain size is to reduce the initial nucleation density and to suppress the continuous nucleation during growth.
Mechanisms
Figure 3a–d compares the typical surface morphologies of h-BN grown on Cu (10 min at 1,050 °C) and Cu–Ni alloys (25 min at 1,050 °C, 5–20 atom % Ni). Many nanoparticles (appearing white in SEM) are observed on Cu surface, while the density decreases dramatically with the increasing of Ni concentration in substrates. The nanoparticles disperse randomly among h-BN grains or accumulate along its perimeters, and are statically correlated to the h-BN grain nucleation (see Supplementary Fig. 3).
(a) SEM images showing the morphology of a Cu foil after the growth of h-BN for 10 min at 1,050 °C. (b–d) SEM images of Cu–Ni alloy foils with (b) 5 atom %, (c) 10 atom % and (d) 20 atom % Ni after growth of h-BN for 25 min at 1,050 °C. (e) Schematic diagram showing the structural development from dissociation of borazane to h-BN. (f) The molecular dynamic simulation of H2BNH2 dissociation on the surface of different substrates. The scale bars in a–d are 5 μm.
As illustrated in Fig. 3e, the borazane precursor (H3BNH3) decomposes into gaseous borazine (B3N3H6) and solid poly-aminoborane (BH2NH2)n upon heating to 70~90 °C (refs 23, 24, 36, 37, 38, 39, 40). Poly-aminoborane can hardly develop into h-BN unless a very high temperature of 1,170–1,500 °C is applied if without catalytic substrates40. Previous studies indicate that on catalytic surface the resulting polymer undergoes a two-stage weight loss process: 2D cross-linking reaction of B-H and N-H groups is initiated from 125 to 200 °C, and then dehydrogenation from unaligned chain branches continues from 600 to 1,100 °C (refs 20, 36). The reactions involved suggest that the nanoparticles are most likely a complex mixture of poly-aminoborane and its partially dehydrogenated derivatives, consistent with earlier reports23,24,26,41 and our Auger investigations (Supplementary Fig. 3a–e).
To have a better understanding on the suppressed formation of nanoparticles at higher Ni content, we carried out simplified density functional theory (DFT) calculations of decomposition energy of (BH2NH2)n=1 on substrates with different compositions (Fig. 3f, details of the calculations are given in Supplementary Fig. 4). The calculation shows that dehydrogenation of BH2NH2 becomes easier by increasing the Ni content in our alloy substrates in a certain range. On the other hand, the decomposition energy of BN, dehydrogenated derivative of BH2NH2, drops more evidently by introducing Ni to the Cu substrate. It is apparent that the introduction of Ni can enhance the decomposition of poly-aminoborane, which helps the reactions of desorption or the formation of Ni–B and Ni–N phases18, consistent with the weak h-BN growth on Cu–Ni alloy with 30 atom % Ni. To enable growth of h-BN on Cu–Ni alloy with high Ni content, one would have to increase the precursor supply; thus, the growth of h-BN enters into a completely different process window18,19,21,23,24.
Characterization
Figure 4a–c shows the SEM image and the corresponding B(KLL) and N(KLL) Auger electron maps of a typical h-BN grain grown on Cu–Ni alloy foil with 15 atom % Ni. It is found that distributions of B and N elements are homogeneous in the grains. Figure 4d presents the AES spectra taken in the blue and magenta dotted areas shown in Fig. 4a, while the insets show the corresponding B(KLL) and N(KLL) spectra, respectively. The B(KLL) and N(KLL) peaks measured on h-BN grain are at 176 and 385 eV, respectively, in agreement with those given in previous reports42,43. It can also be seen that the BN-coated area (blue dot) shows a much weaker peak of O(KLL) than the uncoated area (magenta dot), proving the high oxidation resistance of the h-BN film44. Besides homogeneous h-BN grains, some triangular-shaped h-BN ad-layers with size of more than 10 μm were occasionally observed (Supplementary Fig. 5). The edges of the ad-layers are parallel to those of the first layer, implying strict stacking orders of the h-BN multilayers.
(a) SEM image of a typical h-BN grain grown on a Cu–Ni alloy foil. (b,c) The corresponding B(KLL) and N(KLL) Auger electron maps, respectively. (d) AES spectra taken in the dotted areas shown in a. The insets show the corresponding B(KLL) and N(KLL) spectra, respectively. (e) AFM image of a corner of a h-BN grain transferred on the SiO2/Si substrate. The inset shows the height distribution along the black line in e. (f) High-resolution transmission electron microscopy image of h-BN film on TEM grid with the black arrows showing that it is of monolayer thickness. The inset shows the corresponding selected area electron diffraction pattern. Scale bars are (a) 10 μm, (e) 1 μm and (f) 5 nm.
Figure 4e shows the AFM image of a corner of a triangular-shaped h-BN grain transferred on the SiO2/Si substrate. The height of the transferred film is ~0.455 nm, consistent with that reported for single-layer h-BN23,24,28,29. Figure 4f shows a typical TEM image obtained at the edge of the h-BN film on a TEM grid, confirming that the h-BN prepared is monolayered. The selected area electron diffraction pattern presented in the inset of Fig. 4f shows a set of characteristic sixfold symmetric spots, indicating that the h-BN grain is well crystallized. TEM images containing two to four atomic layers were also occasionally detected, as shown in Supplementary Fig. 6. More characterizations of the h-BN grains, including X-ray photoemission spectroscopy (XPS), optical image and optical absorption spectrum are presented in Supplementary Fig. 7.
Application
We fabricated graphene/h-BN heterostructure to evaluate the quality of CVD-grown h-BN (the transferring method is given in Supplementary Fig. 8). Figure 5a shows the optical image of graphene/h-BN heterostructure on a 90-nm SiO2/Si substrate, with the Raman spectra taken in different areas presented in Fig. 5b. The spectrum taken in the surface of h-BN (magenta dot) exhibits a characteristic peak at 1,370 cm−1 because of the E2g phonon mode of h-BN45, consistent with previous report on the Raman signature of mechanical exfoliated monolayer h-BN films13. The peak near 1,450 cm−1 is from the third-order transverse optic mode of the silicon substrate46,47. Both the spectra taken in the surfaces of graphene/SiO2 (olive dot) and graphene/h-BN (violet dot) indicate that the graphene grain is a well-crystallized monolayer with few defects48,49.
(a) Optical image of graphene/h-BN grains on a 90-nm SiO2/Si substrate. (b) Raman spectra taken from the dotted areas shown in a. (c) Resistivity of graphene/SiO2/Si and graphene/h-BN Hall bar devices versus the back-gate voltage at room temperature, respectively. The values of the extracted carrier mobility are indicated in insets. (d) Graphene/h-BN Hall device exhibits half-integer quantum Hall effect measured at 4 K under a magnetic field B=9 T. The scale bar in a is 10 μm.
Graphene/h-BN and contrastive graphene/SiO2/Si Hall devices were then made to further characterize the electronic properties. Figure 5c displays the resistivity curves of typical graphene/h-BN and graphene/SiO2/Si back-gate field-effect transistors, with the calculated mobility read 6,213 and 3,625 cm2V−1s−1, respectively. The effectiveness of the CVD-grown h-BN as dielectric substrate for graphene devices is thus well proved. Moreover, as given in Fig. 5d, through the graphene/h-BN Hall device the half-integer quantum Hall effect is also observed, further confirming the high quality of both h-BN and graphene grains. As continuous h-BN films containing more than one atomic layers were synthesized and used as substrates, the mobility of our CVD-grown graphene is further improved to ~10,000 cm2V−1s−1 (see Supplementary Fig. 9).
Discussion
In summary, we successfully grow uniform large single-crystal h-BN grains via the CVD method. By rationally designing the Cu–Ni alloy substrate, the nucleation density of h-BN can be greatly decreased to 60 per mm2. Single-crystal h-BN grains with 7,500 μm2 are obtained, which are two orders larger in area than previous reports. Characterizations such as AES, AFM and TEM confirm the uniformity of h-BN grains. Electrical transport measurements show that the CVD-grown h-BN layers exhibit excellent dielectric performance. The success brings in hope in the successive CVD growth of high-quality h-BN, as well as fabrication of graphene/h-BN heterostructure or super lattices, which are important for both fundamental research as well as device applications.
Methods
Deposition of Ni film on a Cu foil
An 8 cm × 8 cm Cu foil (25 μm, 99.8%, Alfa-Aesar) was first electrochemically polished to reduce the surface roughness. The polishing solution consisted of 500 ml of water, 250 ml of ethanol, 250 ml of orthophosphoric acid, 50 ml of isopropyl alcohol and 5 g of urea. After 90 s of electrochemical polishing with the current set at 10A, the thickness of the Cu foil reduced to ~20 μm. Then, the polished Cu foil was loaded into an annealing tube and annealed at 1,050 °C for 2 h in a mixed Ar/H2 (400/100 sccm) flow under atmospheric pressure to further increase its surface flatness and grain size.
After annealing, the Cu foil was electroplated with a layer of Ni. The electrolytic solution was a mixture of 1 l of water, 280 g of NiSO4·6 H2O, 8 g of NiCl2·6 H2O, 4 g of NaF and 30 g of H3BO3. With the current density set at 0.01 A cm−2, a Ni film with a certain thickness was deposited on the Cu foil at a rate of 200 nm min−1.
Growth of h-BN grains
The Ni-coated Cu foils were loaded into a 5-cm diameter CVD-fused quartz tube as shown in Supplementary Fig. 1a. The distance between precursor and substrates is ~60 cm. The annealing and growth process is shown in Supplementary Fig. 1b. The substrates were first heated to 1,050 °C and maintained for 2 h in a H2 flow at a total pressure of ~5 kPa. After that, the two metals were completely interdiffused to Cu–Ni alloy foils, with the atomic proportions determined by the thickness of the deposited Ni film before annealing. For h-BN growth, the temperature was changed to the specified value in the range of 1,050–1,090 °C, and the total pressure was maintained at 50 Pa. At the same time, borazane precursor placed in an Al2O3 boat was heated to 70–90 °C by a heating lamp. After growth, both the heating furnace and the heating lamp were quickly cooled down to room temperature.
Characterizations
h-BN grains were characterized by SEM (Zeiss Supra55, operated at 3 kV), AES (PHI670, operated at 10 kV), AFM (Bruker Dimension Icon, contact mode), TEM (FEI Tecnai G2, 200 kV), Raman spectroscopy (Renishaw, 514 nm laser wavelength), XPS (AXIS UltraDLD) and UV–vis spectrophotometer (EV300).
DFT calculation
All the calculations are performed within the framework of DFT as implemented in the Vienna Ab initio Simulation Package. Electronic exchange and correlation are included through the generalized gradient approximation in the Perdew–Burke–Ernzerhof form. The interaction between valence electrons and ion cores is described by the projected augmented wave method and the energy cutoff for the plane wave functions is 400 eV. All structures were optimized until the maximum force component on each atom is less than 0.02 eV Å−1. The vacuum layer inside the super cell is kept as large as 14 Å to avoid the interaction of the adjacent unit cell. The slab model is composed of 4 × 4 repeating unit cells and the 2 × 2 × 1 k-point mesh is used for the calculation.
The dehydrogenation energies, Edh, of each dehydrogenated borazane on the surfaces are calculated by Edh=E(Sub-BN)+2 × E(H2)–E(Sub)–E(H2BNH2), in which E(Sub-BN) is the total energy of fully dehydrogenated borazane adsorbed on the substrate; E(Sub) and E(H2BNH2) are the energies of substrate and separated H2BNH2, respectively; E(H2) is the energy of hydrogen molecule in the vacuum. The decomposition energies are calculated by Edc=E(Sub-B–N)–E(Sub-BN), in which E(Sub-B–N) and E(Sub-BN) are the energies of separated B, N atoms adsorbed on the substrate and BN cluster adsorbed on the substrate, respectively.
Fabrication and measurement of graphene Hall devices
CVD-grown monolayer graphene grains with a typical size of 50 μm were transferred on as-grown h-BN grains on Cu–Ni alloy, followed by the etching of Cu–Ni and then the graphene/h-BN grains were transferred on a highly doped p-type silicon wafer with a 300-nm SiO2-capping layer. For comparison, some graphene grains were transferred directly on the SiO2/Si substrate. After that, a standard electron beam lithography technique was employed to pattern the Hall bar structure. The contact electrodes (10 nm Au/50 nm Ti) were deposited through electron beam evaporation. Both electrical transport and magnetotransport measurements were carried out in a PPMS-9 T system from Quantum Design.
Additional information
How to cite this article: Lu. G, et al. Synthesis of large single-crystal hexagonal boron nitride grains on Cu–Ni alloy. Nat. Commun. 6:6160 doi: 10.1038/ncomms7160 (2015).
References
Kubota, Y., Watanabe, K., Tsuda, O. & Taniguchi, T. Deep ultraviolet light-emitting hexagonal boron nitride synthesized at atmospheric pressure. Science 317, 932–934 (2007).
Chen, Y., Zou, J., Campbell, S. J. & Le Caer, G. Boron nitride nanotubes: pronounced resistance to oxidation. Appl. Phys. Lett. 84, 2430–2432 (2004).
Song, L. et al. Large scale growth and characterization of atomic hexagonal boron nitride layers. Nano Lett. 10, 3209–3215 (2010).
Kho, J. G., Moon, K. T., Kim, J. H. & Kim, D. P. Properties of boron nitride (BxNy) films produced by the spin-coating process of polyborazine. J. Am. Ceram. Soc. 83, 2681–2683 (2000).
Lipp, A., Schwetz, K. A. & Honold, K. Hexagonal boron nitride: fabrication, properties and application. J. Eur. Ceram. Soc. 5, 3–9 (1989).
Tao, O. et al. Thermal transport in hexagonal boron nitride nanoribbons. Nanotechnology 21, 245701 (2010).
Dean, C. R. et al. Boron nitride substrates for high-quality graphene electronics. Nat. Nanotechnol. 5, 722–726 (2010).
Britnell, L. et al. Field-effect tunneling transistor based on vertical graphene heterostructures. Science 335, 947–950 (2012).
Britnell, L. et al. Resonant tunnelling and negative differential conductance in graphene transistors. Nat. Commun. 4, 1794 (2013).
Feldman, B. E., Martin, J. & Yacoby, A. Broken-symmetry states and divergent resistance in suspended bilayer graphene. Nat. Phys. 5, 889–893 (2009).
Garcia, A. G. F. et al. Effective cleaning of hexagonal boron nitride for graphene devices. Nano Lett. 12, 4449–4454 (2012).
Pacile, D., Meyer, J. C., Girit, C. O. & Zettl, A. The two-dimensional phase of boron nitride: few-atomic-layer sheets and suspended membranes. Appl. Phys. Lett. 92, 133107 (2008).
Gorbachev, R. V. et al. Hunting for monolayer boron nitride: optical and Raman signatures. Small 7, 465–468 (2011).
Tang, S. et al. Nucleation and growth of single crystal graphene on hexagonal boron nitride. Carbon. NY. 50, 329–331 (2012).
Lee, C. et al. Frictional characteristics of atomically thin sheets. Science 328, 76–80 (2010).
Auwärter, W., Muntwiler, M., Osterwalder, J. & Greber, T. Defect lines and two-domain structure of hexagonal boron nitride films on Ni(111). Surf. Sci. 545, L735–L740 (2003).
Auwärter, W., Suter, H. U., Sachdev, H. & Greber, T. Synthesis of one monolayer of hexagonal boron nitride on Ni(111) from B-trichloroborazine (ClBNH)3 . Chem. Mater. 16, 343–345 (2003).
Ismach, A. et al. Toward the controlled synthesis of hexagonal boron nitride films. ACS Nano 6, 6378–6385 (2012).
Lee, Y.-H. et al. Growth selectivity of hexagonal-boron nitride layers on Ni with various crystal orientations. RSC Adv. 2, 111–115 (2012).
Shi, Y. et al. Synthesis of few-layer hexagonal boron nitride thin film by chemical vapor deposition. Nano Lett. 10, 4134–4139 (2010).
Gibb, A., Alem, N. & Zettl, A. Low pressure chemical vapor deposition synthesis of hexagonal boron nitride on polycrystalline metal foils. Phys. Status Solidi B 250, 2727–2731 (2010).
Guo, N. et al. Controllable growth of triangular hexagonal boron nitride domains on copper foils by an improved low-pressure chemical vapor deposition method. Nanotechnology 23, 415605 (2012).
Kim, K. K. et al. Synthesis of monolayer hexagonal boron nitride on Cu foil using chemical vapor deposition. Nano Lett. 12, 161–166 (2012).
Wang, L. et al. Monolayer hexagonal boron nitride films with large domain size and clean interface for enhancing the mobility of graphene-based field-effect transistors. Adv. Mater. 26, 1559–1564 (2014).
Tay, R. Y. et al. Growth of large single-crystalline two-dimensional boron nitride hexagons on electropolished copper. Nano Lett. 14, 839–846 (2014).
Han, J., Lee, J. Y., Kwon, H. & Yeo, J. S. Synthesis of wafer-scale hexagonal boron nitride monolayers free of aminoborane nanoparticles by chemical vapor deposition. Nanotechnology 25, 145604 (2014).
Tay, R. Y. et al. A systematic study of the atmospheric pressure growth of large-area hexagonal crystalline boron nitride film. J. Mater. Chem. C 2, 1650–1657 (2014).
Kim, G. et al. Growth of high-crystalline, single-layer hexagonal boron nitride on recyclable platinum foil. ACS Nano 13, 1834–1839 (2013).
Gao, Y. et al. Repeated and controlled growth of monolayer, bilayer and few-layer hexagonal boron nitride on Pt foils. ACS Nano 7, 5199–5206 (2013).
Corso, M. et al. Boron nitride nanomesh. Science 303, 217–220 (2004).
Goriachko, A. et al. Self-assembly of a hexagonal boron nitride nanomesh on Ru(0001). Langmuir 23, 2928–2931 (2007).
Orofeo, C. M., Suzuki, S., Kageshima, H. & Hibino, H. Growth and low-energy electron microscopy characterization of monolayer hexagonal boron nitride on epitaxial cobalt. Nano Res. 6, 335–347 (2013).
Gibb, A. L. et al. Atomic resolution imaging of grain boundary defects in monolayer chemical vapor deposition-grown hexagonal boron nitride. J. Am. Chem. Soc. 135, 6758–6761 (2013).
Cretu, O., Lin, Y.-C. & Suenaga, K. Evidence for active atomic defects in monolayer hexagonal boron nitride: a new mechanism of plasticity in two-dimensional materials. Nano Lett. 14, 1064–1068 (2014).
Bresnehan, M. S. et al. Integration of hexagonal boron nitride with quasi-freestanding epitaxial graphene: toward wafer-scale, high-performance devices. ACS Nano 6, 5234–5241 (2012).
Kim, D. P. et al. Synthesis and characterization of poly-(aminoborane) as a new boron nitride precursor. Polym. Adv. Technol. 10, 702–712 (1999).
Wolf, G., Baumann, J., Baitalow, F. & Hoffmann, F. P. Calorimetric process monitoring of thermal decomposition of B-N-H compounds. Thermochim. Acta 343, 19–25 (2000).
Baitalow, F., Baumann, J., Wolf, G., Jaenicke-Rossler, K. & Leitner, G. Thermal decomposition of B-N-H compounds investigated by using combined thermoanalytical methods. Thermochim. Acta 391, 159–168 (2002).
Baumann, J., Baitalow, E. & Wolf, G. Thermal decomposition of polymeric aminoborane (H2BNH2)x under hydrogen release. Thermochim. Acta 430, 9–14 (2005).
Frueh, S. et al. Pyrolytic decomposition of ammonia borane to boron nitride. Inorg. Chem. 50, 783–792 (2011).
Wang, Y. T., Yamamoto, Y., Kiyono, H. & Shimada, S. Effect of ambient gas and temperature on crystallization of boron nitride spheres prepared by vapor phase pyrolysis of ammonia borane. J. Am. Ceram. Soc. 92, 787–792 (2009).
Hanke, G. & Muller, K. Low-energy Auger transitions of boron in several boron-compounds. J. Vac. Sci. Technol. A 2, 964–968 (1984).
Xu, M., Fujita, D., Chen, H. & Hanagata, N. Formation of monolayer and few-layer hexagonal boron nitride nanosheets via surface segregation. Nanoscale 3, 2854–2858 (2011).
Liu, Z. et al. Ultrathin high-temperature oxidation-resistant coatings of hexagonal boron nitride. Nat. Commun. 4, 2541 (2013).
Arenal, R. et al. Raman spectroscopy of single-wall boron nitride nanotubes. Nano Lett. 6, 1812–1816 (2006).
Spizzirri, P. G. et al. Nano-Raman spectroscopy of silicon surfaces http://arxiv.org/abs/1002.2692 (2010).
Han, G. H. et al. Continuous growth of hexagonal graphene and boron nitride in-plane heterostructures by atmospheric pressure chemical vapor deposition. ACS Nano 7, 10129–10138 (2013).
Ferrari, A. C. et al. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 97, 187401 (2006).
Graf, D. et al. Spatially resolved Raman spectroscopy of single- and few-layer graphene. Nano Lett. 7, 238–242 (2007).
Acknowledgements
We thank X.F. Zhang for providing CVD-grown graphene on Cu, J. Chen, X.Y. Liu and Q.J. Sun for the help in device fabrications. The work in Shanghai Institute of Microsystem and Information Technology, Chinese Academy of Sciences is partially supported by the National Science and Technology Major Projects of China (Grant No. 2011ZX02707), Chinese Academy of Sciences (Grant No. KGZD-EW-303 and XDB04010500), CAS International Collaboration and Innovation Program on High Mobility Materials Engineering, the National Natural Science Foundation of China (Grant No. 11304337, 11104303, 11274333, 11204339 and 61136005) and the projects from Science and Technology Commission of Shanghai Municipality (Grant No. 12JC1410100 and 12JC1403900).
Author information
Authors and Affiliations
Contributions
M.J. and X.X. supervised the research work. T.W. conceived the project. T.W. and G.L. designed the experiments. G.L. performed h-BN growth, transfer and characterization. F.D. and Q.Y. performed the DFT calculations. H.W. and H.W. fabricated the graphene electronic devices and carried out the transport measurements. T.W., X.X. and G.L. analysed the experimental data and designed the figures. H.W., G.L., T.W., X.X. and M.J. co-wrote the manuscript and all authors contributed to the critical discussions of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-9 (PDF 349 kb)
Rights and permissions
About this article
Cite this article
Lu, G., Wu, T., Yuan, Q. et al. Synthesis of large single-crystal hexagonal boron nitride grains on Cu–Ni alloy. Nat Commun 6, 6160 (2015). https://doi.org/10.1038/ncomms7160
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms7160
This article is cited by
-
Progress on boron nitride nanostructure materials: properties, synthesis and applications in hydrogen storage and analytical chemistry
Journal of Nanostructure in Chemistry (2023)
-
Towards n-type conductivity in hexagonal boron nitride
Nature Communications (2022)
-
Growth, coalescence, and etching of two-dimensional overlayers on metals modulated by near-surface Ar nanobubbles
Nano Research (2022)
-
Abnormal anti-oxidation behavior of hexagonal boron nitride grown on copper
Nano Research (2022)
-
Intrinsic toughening and stable crack propagation in hexagonal boron nitride
Nature (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.