Abstract
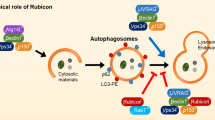
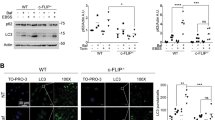
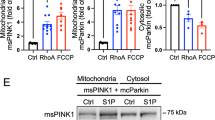
Here we show that Mst1, a proapoptotic kinase, impairs protein quality control mechanisms in the heart through inhibition of autophagy. Stress-induced activation of Mst1 in cardiomyocytes promoted accumulation of p62 and aggresome formation, accompanied by the disappearance of autophagosomes. Mst1 phosphorylated the Thr108 residue in the BH3 domain of Beclin1, which enhanced the interaction between Beclin1 and Bcl-2 and/or Bcl-xL, stabilized the Beclin1 homodimer, inhibited the phosphatidylinositide 3-kinase activity of the Atg14L-Beclin1-Vps34 complex and suppressed autophagy. Furthermore, Mst1-induced sequestration of Bcl-2 and Bcl-xL by Beclin1 allows Bax to become active, thereby stimulating apoptosis. Mst1 promoted cardiac dysfunction in mice subjected to myocardial infarction by inhibiting autophagy, associated with increased levels of Thr108-phosphorylated Beclin1. Moreover, dilated cardiomyopathy in humans was associated with increased levels of Thr108-phosphorylated Beclin1 and signs of autophagic suppression. These results suggest that Mst1 coordinately regulates autophagy and apoptosis by phosphorylating Beclin1 and consequently modulating a three-way interaction among Bcl-2 proteins, Beclin1 and Bax.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Levine, B. & Klionsky, D.J. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev. Cell 6, 463–477 (2004).
Nakai, A. et al. The role of autophagy in cardiomyocytes in the basal state and in response to hemodynamic stress. Nat. Med. 13, 619–624 (2007).
Yan, L. et al. Autophagy in chronically ischemic myocardium. Proc. Natl. Acad. Sci. USA 102, 13807–13812 (2005).
Matsui, Y. et al. Distinct roles of autophagy in the heart during ischemia and reperfusion: roles of AMP-activated protein kinase and Beclin 1 in mediating autophagy. Circ. Res. 100, 914–922 (2007).
Zhu, H. et al. Cardiac autophagy is a maladaptive response to hemodynamic stress. J. Clin. Invest. 117, 1782–1793 (2007).
Shih, H., Lee, B., Lee, R.J. & Boyle, A.J. The aging heart and post-infarction left ventricular remodeling. J. Am. Coll. Cardiol. 57, 9–17 (2011).
Pattingre, S. et al. Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122, 927–939 (2005).
Mizushima, N. & Komatsu, M. Autophagy: renovation of cells and tissues. Cell 147, 728–741 (2011).
Liang, X.H. et al. Protection against fatal Sindbis virus encephalitis by beclin, a novel Bcl-2-interacting protein. J. Virol. 72, 8586–8596 (1998).
Kihara, A., Noda, T., Ishihara, N. & Ohsumi, Y. Two distinct Vps34 phosphatidylinositol 3-kinase complexes function in autophagy and carboxypeptidase Y sorting in Saccharomyces cerevisiae. J. Cell Biol. 152, 519–530 (2001).
Itakura, E., Kishi, C., Inoue, K. & Mizushima, N. Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol. Biol. Cell 19, 5360–5372 (2008).
Funderburk, S.F., Wang, Q.J. & Yue, Z. The Beclin 1–VPS34 complex – at the crossroads of autophagy and beyond. Trends Cell Biol. 20, 355–362 (2010).
Pan, D. The Hippo signaling pathway in development and cancer. Dev. Cell 19, 491–505 (2010).
Yamamoto, S. et al. Activation of Mst1 causes dilated cardiomyopathy by stimulating apoptosis without compensatory ventricular myocyte hypertrophy. J. Clin. Invest. 111, 1463–1474 (2003).
Del Re, D.P. et al. Proapoptotic Rassf1A/Mst1 signaling in cardiac fibroblasts is protective against pressure overload in mice. J. Clin. Invest. 120, 3555–3567 (2010).
Odashima, M. et al. Inhibition of endogenous Mst1 prevents apoptosis and cardiac dysfunction without affecting cardiac hypertrophy after myocardial infarction. Circ. Res. 100, 1344–1352 (2007).
Gautreau, A. et al. Isolation and characterization of an aggresome determinant in the NF2 tumor suppressor. J. Biol. Chem. 278, 6235–6242 (2003).
Rote, K.V. & Rechsteiner, M. Degradation of microinjected proteins: effects of lysosomotropic agents and inhibitors of autophagy. J. Cell Physiol. 116, 103–110 (1983).
Hariharan, N. et al. Deacetylation of FoxO by Sirt1 plays an essential role in mediating starvation-induced autophagy in cardiac myocytes. Circ. Res. 107, 1470–1482 (2010).
Roggo, L. et al. Membrane transport in Caenorhabditis elegans: an essential role for VPS34 at the nuclear membrane. EMBO J. 21, 1673–1683 (2002).
Maiuri, M.C. et al. Functional and physical interaction between Bcl-XL and a BH3-like domain in Beclin1. EMBO J. 26, 2527–2539 (2007).
Li, X. et al. Imperfect interface of Beclin1 coiled-coil domain regulates homodimer and heterodimer formation with Atg14L and UVRAG. Nat. Commun. 3, 662 (2012).
Zhong, Y. et al. Distinct regulation of autophagic activity by Atg14L and Rubicon associated with Beclin 1–phosphatidylinositol-3-kinase complex. Nat. Cell Biol. 11, 468–476 (2009).
Matsunaga, K. et al. Two Beclin 1-binding proteins, Atg14L and Rubicon, reciprocally regulate autophagy at different stages. Nat. Cell Biol. 11, 385–396 (2009).
Sun, Q. et al. Identification of Barkor as a mammalian autophagy-specific factor for Beclin 1 and class III phosphatidylinositol 3-kinase. Proc. Natl. Acad. Sci. USA 105, 19211–19216 (2008).
Zalckvar, E. et al. DAP-kinase-mediated phosphorylation on the BH3 domain of beclin 1 promotes dissociation of beclin 1 from Bcl-XL and induction of autophagy. EMBO Rep. 10, 285–292 (2009).
Oberstein, A., Jeffrey, P.D. & Shi, Y. Crystal structure of the Bcl-XL-Beclin 1 peptide complex: Beclin 1 is a novel BH3-only protein. J. Biol. Chem. 282, 13123–13132 (2007).
Ross, C.A. & Poirier, M.A. Opinion: what is the role of protein aggregation in neurodegeneration? Nat. Rev. Mol. Cell Biol. 6, 891–898 (2005).
Tannous, P. et al. Intracellular protein aggregation is a proximal trigger of cardiomyocyte autophagy. Circulation 117, 3070–3078 (2008).
Harvey, K. & Tapon, N. The Salvador-Warts-Hippo pathway—an emerging tumour-suppressor network. Nat. Rev. Cancer 7, 182–191 (2007).
Wei, Y., Pattingre, S., Sinha, S., Bassik, M. & Levine, B. JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol. Cell 30, 678–688 (2008).
He, C. et al. Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis. Nature 481, 511–515 (2012).
Noble, C.G., Dong, J.M., Manser, E. & Song, H. Bcl-xL and UVRAG cause a monomer-dimer switch in Beclin1. J. Biol. Chem. 283, 26274–26282 (2008).
Mizushima, N. et al. In vivo analysis of autophagy in response to nutrient starvation using transgenic mice expressing a fluorescent autophagosome marker. Mol. Biol. Cell 15, 1101–1111 (2004).
Oh, S. et al. Crucial role for Mst1 and Mst2 kinases in early embryonic development of the mouse. Mol. Biol. Cell 29, 6309–6320 (2009).
Qu, X. et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J. Clin. Invest. 112, 1809–1820 (2003).
Ago, T. et al. A redox-dependent pathway for regulating class II HDACs and cardiac hypertrophy. Cell 133, 978–993 (2008).
Whelan, R.S. et al. Bax regulates primary necrosis through mitochondrial dynamics. Proc. Natl. Acad. Sci. USA 109, 6566–6571 (2012).
Komatsu, M. et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441, 880–884 (2006).
Yabuta, N. et al. Structure, expression, and chromosome mapping of LATS2, a mammalian homologue of the Drosophila tumor suppressor gene lats/warts. Genomics 63, 263–270 (2000).
Maejima, Y. et al. Nitric oxide inhibits ischemia/reperfusion-induced myocardial apoptosis by modulating cyclin A-associated kinase activity. Cardiovasc. Res. 59, 308–320 (2003).
Matsushima, S. et al. Increased oxidative stress in the nucleus caused by Nox4 mediates oxidation of HDAC4 and cardiac hypertrophy. Circ. Res. 112, 651–663 (2013).
Hayashi, K. & Kojima, C. pCold-GST vector: a novel cold-shock vector containing GST tag for soluble protein production. Protein Expr. Purif. 62, 120–127 (2008).
Matsui, Y. et al. Lats2 is a negative regulator of myocyte size in the heart. Circ. Res. 103, 1309–1318 (2008).
Shen, D. et al. Novel cell- and tissue-based assays for detecting misfolded and aggregated protein accumulation within aggresomes and inclusion bodies. Cell Biochem. Biophys. 60, 173–185 (2011).
Gillooly, D.J. et al. Localization of phosphatidylinositol 3-phosphate in yeast and mammalian cells. EMBO J. 19, 4577–4588 (2000).
Shi, C.S. & Kehrl, J.H. TRAF6 and A20 regulate lysine 63-linked ubiquitination of Beclin1 to control TLR4-induced autophagy. Sci. Signal. 3, ra42 (2010).
Sciarretta, S. et al. Rheb is a critical regulator of autophagy during myocardial ischemia: pathophysiological implications in obesity and metabolic syndrome. Circulation 125, 1134–1146 (2012).
Acknowledgements
We thank C. Brady for critical reading of the manuscript and Y. Ikeda, Y. Matsuda and N. Tamura for their excellent technical assistance. We also thank N. Mizushima (University of Tokyo) for Tg-GFP-LC3 mice, T. Yoshimori (Osaka University) for mRFP-GFP-LC3 plasmid, C. Kojima (Nara Institute of Science and Technology) for pCold-GST plasmid, M. Komatsu (Tokyo Metropolitan Institute of Medical Science) for Atg7flox/flox mice, R. Kitsis (Albert Einstein College of Medicine) for Bax−/−; Bak1−/− MEFs, and B. Levine (University of Texas Southwestern) for Becn1+/− mice. This work was supported in part by US Public Health Service grants HL59139, HL67724, HL69020, HL91469, HL102738 and AG27211, the Foundation of Leducq Transatlantic Network of Excellence (J.S.), American Heart Association Scientist Development Grant 12SDG12070262 and the Banyu Fellowship Program sponsored by Banyu Life Science Foundation International (Y.M.).
Author information
Authors and Affiliations
Contributions
Y.M. designed and performed experiments, analyzed data and wrote the manuscript. S.K. designed and performed experiments and analyzed data. P.Z. performed animal surgery. T.L. and H.L. performed experiments and analyzed data regarding proteomic analyses. A.I., S.S., D.P.D.R., and C.-P.H. performed experiments and analyzed data. D.K.Z. wrote the manuscript. D.-S.L. provided the animals and cells and supervised the project. M.I. supervised the project. J.S. designed the studies, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 74070 kb)
Rights and permissions
About this article
Cite this article
Maejima, Y., Kyoi, S., Zhai, P. et al. Mst1 inhibits autophagy by promoting the interaction between Beclin1 and Bcl-2. Nat Med 19, 1478–1488 (2013). https://doi.org/10.1038/nm.3322
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3322
This article is cited by
-
Protective effects of melatonin on myocardial microvascular endothelial cell injury under hypertensive state by regulating Mst1
BMC Cardiovascular Disorders (2023)
-
Ser14 phosphorylation of Bcl-xL mediates compensatory cardiac hypertrophy in male mice
Nature Communications (2023)
-
Porphyromonas gingivalis, a periodontal pathogen, impairs post-infarcted myocardium by inhibiting autophagosome–lysosome fusion
International Journal of Oral Science (2023)
-
Fibroblast growth factor 18 alleviates stress-induced pathological cardiac hypertrophy in male mice
Nature Communications (2023)
-
Mitophagy as a mitochondrial quality control mechanism in myocardial ischemic stress: from bench to bedside
Cell Stress and Chaperones (2023)