Abstract
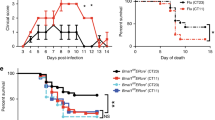
The circadian system is an important regulator of immune function. Human inflammatory lung diseases frequently show time-of-day variation in symptom severity and lung function, but the mechanisms and cell types underlying these effects remain unclear. We show that pulmonary antibacterial responses are modulated by a circadian clock within epithelial club (Clara) cells. These drive circadian neutrophil recruitment to the lung via the chemokine CXCL5. Genetic ablation of the clock gene Bmal1 (also called Arntl or MOP3) in bronchiolar cells disrupts rhythmic Cxcl5 expression, resulting in exaggerated inflammatory responses to lipopolysaccharide and an impaired host response to Streptococcus pneumoniae infection. Adrenalectomy blocks rhythmic inflammatory responses and the circadian regulation of CXCL5, suggesting a key role for the adrenal axis in driving CXCL5 expression and pulmonary neutrophil recruitment. Glucocorticoid receptor occupancy at the Cxcl5 locus shows circadian oscillations, but this is disrupted in mice with bronchiole-specific ablation of Bmal1, leading to enhanced CXCL5 expression despite normal corticosteroid secretion. The therapeutic effects of the synthetic glucocorticoid dexamethasone depend on intact clock function in the airway. We now define a regulatory mechanism that links the circadian clock and glucocorticoid hormones to control both time-of-day variation and the magnitude of pulmonary inflammation and responses to bacterial infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Martinez, F.J., Donohue, J.F. & Rennard, S.I. The future of chronic obstructive pulmonary disease treatment–difficulties of and barriers to drug development. Lancet 378, 1027–1037 (2011).
Scheiermann, C. et al. Adrenergic nerves govern circadian leukocyte recruitment to tissues. Immunity 37, 290–301 (2012).
Scheiermann, C., Kunisaki, Y. & Frenette, P.S. Circadian control of the immune system. Nat. Rev. Immunol. 13, 190–198 (2013).
Keller, M. et al. A circadian clock in macrophages controls inflammatory immune responses. Proc. Natl. Acad. Sci. USA 106, 21407–21412 (2009).
Gibbs, J.E. et al. The nuclear receptor REV-ERBα mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines. Proc. Natl. Acad. Sci. USA 109, 582–587 (2012).
Wang, X., Reece, S.P., Van Scott, M.R. & Brown, J.M. A circadian clock in murine bone marrow-derived mast cells modulates IgE-dependent activation in vitro. Brain Behav. Immun. 25, 127–134 (2011).
Shackelford, P.G. & Feigin, R.D. Periodicity of susceptibility to pneumococcal infection: influence of light and adrenocortical secretions. Science 182, 285–287 (1973).
Silver, A.C., Arjona, A., Walker, W.E. & Fikrig, E. The circadian clock controls Toll-like receptor 9-mediated innate and adaptive immunity. Immunity 36, 251–261 (2012).
Halberg, F., Johnson, N.E.A., Brown, B.W. & Bittner, J.J. Susceptibility rhythm to E. coli endotoxin and bioassay. Proc. Soc. Exp. Biol. Med. 103, 142–144 (1960).
Castanon-Cervantes, O. et al. Dysregulation of inflammatory responses by chronic circadian disruption. J. Immunol. 185, 5796–5805 (2010).
Bellet, M.M. et al. Circadian clock regulates the host response to Salmonella. Proc. Natl. Acad. Sci. USA 110, 9897–9902 (2013).
Witko-Sarsat, V., Rieu, P., Descamps-Latscha, B., Lesavre, P. & Halbwachs-Mecarelli, L. Neutrophils: molecules, functions and pathophysiological aspects. Lab. Invest. 80, 617–653 (2000).
Perkins, N.D. Integrating cell-signalling pathways with NF-κB and IKK function. Nat. Rev. Mol. Cell Biol. 8, 49–62 (2007).
Ota, T., Fustin, J.M., Yamada, H., Doi, M. & Okamura, H. Circadian clock signals in the adrenal cortex. Mol. Cell. Endocrinol. 349, 30–37 (2012).
Dickmeis, T. Glucocorticoids and the circadian clock. J. Endocrinol. 200, 3–22 (2009).
Nader, N., Chrousos, G.P. & Kino, T. Circadian rhythm transcription factor CLOCK regulates the transcriptional activity of the glucocorticoid receptor by acetylating its hinge region lysine cluster: potential physiological implications. FASEB J. 23, 1572–1583 (2009).
Lamia, K.A. et al. Cryptochromes mediate rhythmic repression of the glucocorticoid receptor. Nature 480, 552–556 (2011).
Han, D.H., Lee, Y.J., Kim, K., Kim, C.J. & Cho, S. Modulation of glucocorticoid receptor induction properties by core circadian clock proteins. Mol. Cell. Endocrinol. 383, 170–180 (2014).
Aschoff, J. in Circadian Clocks (ed. Aschoff, J.) 95–111 (Amsterdam, North-Holland, 1965).
Paladino, N., Leone, M.J., Plano, S.A. & Golombek, D.A. Paying the circadian toll: the circadian response to LPS injection is dependent on the Toll-like receptor 4. J. Neuroimmunol. 225, 62–67 (2010).
Kadioglu, A. et al. Host cellular immune response to pneumococcal lung infection in mice. Infect. Immun. 68, 492–501 (2000).
Yoo, S.H. et al. PERIOD2:LUCIFERASE real-time reporting of circadian dynamics reveals persistent circadian oscillations in mouse peripheral tissues. Proc. Natl. Acad. Sci. USA 101, 5339–5346 (2004).
Elizur, A. et al. Clara cells impact the pulmonary innate immune response to LPS. Am. J. Physiol. Lung Cell. Mol. Physiol. 293, L383–L392 (2007).
Hirota, T. et al. Identification of small molecule activators of cryptochrome. Science 337, 1094–1097 (2012).
Gibbs, J.E. et al. Circadian timing in the lung; a specific role for bronchiolar epithelial cells. Endocrinology 150, 268–276 (2009).
Li, S. et al. Foxp1/4 control epithelial cell fate during lung development and regeneration through regulation of anterior gradient 2. Development 139, 2500–2509 (2012).
Guilding, C. et al. Suppressed cellular oscillations in after-hours mutant mice are associated with enhanced circadian phase-resetting. J. Physiol. (Lond.) 591, 1063–1080 (2013).
Jeyaseelan, S. et al. Induction of CXCL5 during inflammation in the rodent lung involves activation of alveolar epithelium. Am. J. Respir. Cell Mol. Biol. 32, 531–539 (2005).
Mei, J. et al. CXCL5 regulates chemokine scavenging and pulmonary host defense to bacterial infection. Immunity 33, 106–117 (2010).
John, S. et al. Interaction of the glucocorticoid receptor with the chromatin landscape. Mol. Cell 29, 611–624 (2008).
Dalm, S., Brinks, V., van der Mark, M.H., de Kloet, E.R. & Oitzl, M.S. Non-invasive stress-free application of glucocorticoid ligands in mice. J. Neurosci. Methods 170, 77–84 (2008).
The ENCODE Project Consortium. et al. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012).
Vyas, S. et al. Insights into negative regulation by the glucocorticoid receptor from genome-wide profiling of inflammatory cistromes. Mol. Cell 49, 1–3 (2013).
Grommes, J. & Soehnlein, O. Contribution of neutrophils to acute lung injury. Mol. Med. 17, 293–307 (2011).
Nakagome, K., Matsushita, S. & Nagata, M. Neutrophilic inflammation in severe asthma. Int. Arch. Allergy Immunol. 158 (suppl. 1) 96–102 (2012).
Quint, J.K. & Wedzicha, J.A. The neutrophil in chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 119, 1065–1071 (2007).
Eash, K.J., Greenbaum, A.M., Gopalan, P.K. & Link, D.C. CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow. J. Clin. Invest. 120, 2423–2431 (2010).
Suratt, B.T. et al. Role of the CXCR4/SDF-1 chemokine axis in circulating neutrophil homeostasis. Blood 104, 565–571 (2004).
Kobayashi, Y. Neutrophil infiltration and chemokines. Crit. Rev. Immunol. 26, 307–316 (2006).
Mei, J. et al. Cxcr2 and Cxcl5 regulate the IL-17/G-CSF axis and neutrophil homeostasis in mice. J. Clin. Invest. 122, 974–986 (2012).
Tuller, T., Atar, S., Ruppin, E., Gurevich, M. & Achiron, A. Common and specific signatures of gene expression and protein-protein interactions in autoimmune diseases. Genes Immun. 14, 67–82 (2013).
Heit, B., Tavener, S., Raharjo, E. & Kubes, P. An intracellular signaling hierarchy determines direction of migration in opposing chemotactic gradients. J. Cell Biol. 159, 91–102 (2002).
Hwang, J.W., Sundar, I.K., Yao, H., Sellix, M.T. & Rahman, I. Circadian clock function is disrupted by environmental tobacco/cigarette smoke, leading to lung inflammation and injury via a SIRT1-BMAL1 pathway. FASEB J. 28, 176–194 (2014).
Vasu, V.T., Cross, C.E. & Gohil, K. Nr1d1, an important circadian pathway regulatory gene, is suppressed by cigarette smoke in murine lungs. Integr. Cancer Ther. 8, 321–328 (2009).
Smith, J.B. & Herschman, H.R. Glucocorticoid-attenuated response genes encode intercellular mediators, including a new C–X-C chemokine. J. Biol. Chem. 270, 16756–16765 (1995).
Li, H. et al. Cre-mediated recombination in mouse Clara cells. Genesis 46, 300–307 (2008).
McGrath, E.E. et al. TNF-related apoptosis-inducing ligand (TRAIL) regulates inflammatory neutrophil apoptosis and enhances resolution of inflammation. J. Leukoc. Biol. 90, 855–865 (2011).
Meng, Q.J. et al. Ligand modulation of REV-ERBα function resets the peripheral circadian clock in a phasic manner. J. Cell Sci. 121, 3629–3635 (2008).
Lee, T.I., Johnstone, S.E. & Young, R.A. Chromatin immunoprecipitation and microarray-based analysis of protein location. Nat. Protoc. 1, 729–748 (2006).
Okamoto, K., Onai, K. & Ishiura, M. RAP, an integrated program for monitoring bioluminescence and analyzing circadian rhythms in real time. Anal. Biochem. 340, 193–200 (2005).
Acknowledgements
We thank J.S. Takahashi, Southwestern University, for the gift of the Per2-luc mice, F. Scott and A. Hughes for help with imaging lung tissue slices and J. Woodburn for technical help. We thank M.H. Hastings for comments on the manuscript. The work was supported by research grants to A.L. from GlaxoSmithKline and the Biotechnology and Biological Sciences Research Council, UK (BB/D004357/1, BB/K003119/1 and BB/K003097/1), to D.R. from the Wellcome Trust and the UK National Institute for Health Research Musculoskeletal Biomedical Research Unit Manchester, as well as the US National Institute of Diabetes and Digestive and Kidney Diseases grant P01 DK59820 to F.D. and US National Institutes of Health grants 1RO1HL105834 and 1RO1AI099479 to G.S.W.
Author information
Authors and Affiliations
Contributions
J.G., D.R., S.F. and A.L. conceived the project. J.G., D.R., G.S.W. and A.L. designed the experiments, and J.G., D.R. and A.L. wrote the paper. J.G. performed all in vivo experiments, oversaw all breeding programs, as well as measures of cellular infiltrates, qPCR gene and cytokine protein expression studies and all statistical analyses; L.I. carried out all adrenalectomies and the associated in vivo studies and downstream analyses; L.M. performed transfection studies; M.P. undertook studies of human bronchiolar epithelial cells; J.M. and G.S.W. undertook all studies of CXCL5-null mice; N.Y. performed ChIP studies; T.P. analyzed DEX-responsive genes in the lung; T.B. and T.H. led pulmonary S. pneumoniae infection studies; F.D. developed the Ccsp-icre mouse model; B.S. and N.B. managed the transgenic animal colony and genotyping; and B.S. undertook the radioactive in situ hybridization studies.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14 and Supplementary Tables 1–4 (PDF 2169 kb)
CCSP-bmal–/– lung video.
Video showing bioluminescence over time in a lung slice from a CCSP-bmal–/– mouse on a PER2::luc background. The slice was recorded for 3 d. (AVI 18181 kb)
Wild-type lung video.
Video showing bioluminescence over time in a lung slice from a wild-type mouse on a PER2::luc background. The slice was recorded for 3 d. (AVI 18181 kb)
Rights and permissions
About this article
Cite this article
Gibbs, J., Ince, L., Matthews, L. et al. An epithelial circadian clock controls pulmonary inflammation and glucocorticoid action. Nat Med 20, 919–926 (2014). https://doi.org/10.1038/nm.3599
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3599
This article is cited by
-
Investigation of the rhythmic recruitment of tear neutrophils to the ocular surface and their phenotypes
Scientific Reports (2024)
-
Club cell protein (CC16) in serum as an effect marker for small airway epithelial damage caused by diesel exhaust and blasting fumes in potash mining
International Archives of Occupational and Environmental Health (2024)
-
Glucocorticoids coordinate the bladder peripheral clock and diurnal micturition pattern in mice
Communications Biology (2023)
-
Circadian clock molecule REV-ERBα regulates lung fibrotic progression through collagen stabilization
Nature Communications (2023)
-
Circadian regulation of liver function: from molecular mechanisms to disease pathophysiology
Nature Reviews Gastroenterology & Hepatology (2023)