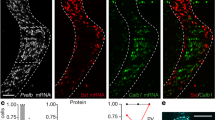
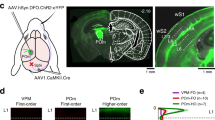
Abstract
Descending projections from sensory areas of the cerebral cortex are among the largest pathways in the brain, suggesting that they are important for subcortical processing. Although corticofugal inputs have been shown to modulate neuronal responses in the thalamus and midbrain, the behavioral importance of these changes remains unknown. In the auditory system, one of the major descending pathways is from cortical layer V pyramidal cells to the inferior colliculus in the midbrain. We examined the role of these neurons in experience-dependent recalibration of sound localization in adult ferrets by selectively killing the neurons using chromophore-targeted laser photolysis. When provided with appropriate training, animals normally relearn to localize sound accurately after altering the spatial cues available by reversibly occluding one ear. However, this ability was lost after eliminating corticocollicular neurons, whereas normal sound-localization accuracy was unaffected. The integrity of this descending pathway is therefore critical for learning-induced localization plasticity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Gilbert, C.D., Li, W. & Piech, V. Perceptual learning and adult cortical plasticity. J. Physiol. (Lond.) 587, 2743–2751 (2009).
Dahmen, J.C. & King, A.J. Learning to hear: plasticity of auditory cortical processing. Curr. Opin. Neurobiol. 17, 456–464 (2007).
Edeline, J.M. & Weinberger, N.M. Thalamic short-term plasticity in the auditory system: associative returning of receptive fields in the ventral medial geniculate body. Behav. Neurosci. 105, 618–639 (1991).
Tzounopoulos, T. & Kraus, N. Learning to encode timing: mechanisms of plasticity in the auditory brainstem. Neuron 62, 463–469 (2009).
Suga, N. & Ma, X. Multiparametric corticofugal modulation and plasticity in the auditory system. Nat. Rev. Neurosci. 4, 783–794 (2003).
Sillito, A.M., Jones, H.E., Gerstein, G.L. & West, D.C. Feature-linked synchronization of thalamic relay cell firing induced by feedback from the visual cortex. Nature 369, 479–482 (1994).
Krupa, D.J., Ghazanfar, A.A. & Nicolelis, M.A. Immediate thalamic sensory plasticity depends on corticothalamic feedback. Proc. Natl. Acad. Sci. USA 96, 8200–8205 (1999).
Xiao, Z. & Suga, N. Modulation of cochlear hair cells by the auditory cortex in the mustached bat. Nat. Neurosci. 5, 57–63 (2002).
Perrot, X. et al. Evidence for corticofugal modulation of peripheral auditory activity in humans. Cereb. Cortex 16, 941–948 (2006).
Alvarado, J.C., Stanford, T.R., Vaughan, J.W. & Stein, B.E. Cortex mediates multisensory, but not unisensory, integration in superior colliculus. J. Neurosci. 27, 12775–12786 (2007).
Luo, F., Wang, Q., Kashani, A. & Yan, J. Corticofugal modulation of initial sound processing in the brain. J. Neurosci. 28, 11615–11621 (2008).
Suga, N. Role of corticofugal feedback in hearing. J. Comp. Physiol. A Neuroethol. Sens. Neural. Behav. Physiol. 194, 169–183 (2008).
Ma, X. & Suga, N. Plasticity of bat's central auditory system evoked by focal electric stimulation of auditory and/or somatosensory cortices. J. Neurophysiol. 85, 1078–1087 (2001).
Yan, J., Zhang, Y. & Ehret, G. Corticofugal shaping of frequency tuning curves in the central nucleus of the inferior colliculus of mice. J. Neurophysiol. 93, 71–83 (2005).
Yan, J. & Ehret, G. Corticofugal modulation of midbrain sound processing in the house mouse. Eur. J. Neurosci. 16, 119–128 (2002).
Ma, X. & Suga, N. Corticofugal modulation of duration-tuned neurons in the midbrain auditory nucleus in bats. Proc. Natl. Acad. Sci. USA 98, 14060–14065 (2001).
Zhou, X. & Jen, P.H. Corticofugal modulation of directional sensitivity in the midbrain of the big brown bat, Eptesicus fuscus. Hear. Res. 203, 201–215 (2005).
Nakamoto, K.T., Jones, S.J. & Palmer, A.R. Descending projections from auditory cortex modulate sensitivity in the midbrain to cues for spatial position. J. Neurophysiol. 99, 2347–2356 (2008).
Macklis, J.D. Transplanted neocortical neurons migrate selectively into regions of neuronal degeneration produced by chromophore-targeted laser photolysis. J. Neurosci. 13, 3848–3863 (1993).
Magavi, S.S., Leavitt, B.R. & Macklis, J.D. Induction of neurogenesis in the neocortex of adult mice. Nature 405, 951–955 (2000).
King, A.J., Doubell, T.P. & Schnupp, J.W.H. The shape of ears to come: dynamic coding of auditory space. Trends Cog. Sci. 5, 261–270 (2001).
Chase, S.M. & Young, E.D. Cues for sound localization are encoded in multiple aspects of spike trains in the inferior colliculus. J. Neurophysiol. 99, 1672–1682 (2008).
Jenkins, W.M. & Merzenich, M.M. Role of cat primary auditory cortex for sound-localization behavior. J. Neurophysiol. 52, 819–847 (1984).
Kavanagh, G.L. & Kelly, J.B. Contributions of auditory cortex to sound localization in the ferret (Mustela putorius). J. Neurophysiol. 57, 1746–1766 (1987).
Heffner, H.E. & Heffner, R.S. Effect of bilateral auditory cortex lesions on sound localization in Japanese macaques. J. Neurophysiol. 64, 915–931 (1990).
Spierer, L., Tardif, E., Sperdin, H., Murray, M.M. & Clarke, S. Learning-induced plasticity in auditory spatial representations revealed by electrical neuroimaging. J. Neurosci. 27, 5474–5483 (2007).
Nodal, F.R., Bajo, V.M., Parsons, C.H., Schnupp, J.W.H. & King, A.J. Sound localization behavior in ferrets: comparison of acoustic orientation and approach-to-target responses. Neuroscience 154, 397–408 (2008).
Thompson, G.C. & Masterton, R.B. Brain stem auditory pathways involved in reflexive head orientation to sound. J. Neurophysiol. 41, 1183–1202 (1978).
Lomber, S.G., Payne, B.R. & Cornwell, P. Role of the superior colliculus in analyses of space: superficial and intermediate layer contributions to visual orienting, auditory orienting, and visuospatial discriminations during unilateral and bilateral deactivations. J. Comp. Neurol. 441, 44–57 (2001).
Kacelnik, O., Nodal, F.R., Parsons, C.H. & King, A.J. Training-induced plasticity of auditory localization in adult mammals. PLoS Biol. 4, e71 (2006).
Bajo, V.M., Nodal, F.R., Bizley, J.K., Moore, D.R. & King, A.J. The ferret auditory cortex: descending projections to the inferior colliculus. Cereb. Cortex 17, 475–491 (2007).
Winer, J.A., Larue, D.T., Diehl, J.J. & Hefti, B.J. Auditory cortical projections to the cat inferior colliculus. J. Comp. Neurol. 400, 147–174 (1998).
King, A.J., Jiang, Z.D. & Moore, D.R. Auditory brainstem projections to the ferret superior colliculus: anatomical contribution to the neural coding of sound azimuth. J. Comp. Neurol. 390, 342–365 (1998).
Knudsen, E.I., Esterly, S.D. & Knudsen, P.F. Monaural occlusion alters sound localization during a sensitive period in the barn owl. J. Neurosci. 4, 1001–1011 (1984).
King, A.J., Parsons, C.H. & Moore, D.R. Plasticity in the neural coding of auditory space in the mammalian brain. Proc. Natl. Acad. Sci. USA 97, 11821–11828 (2000).
Bauer, R.W., Matuzsa, J.L., Blackmer, F. & Glucksberg, S. Noise localization after unilateral attenuation. J. Acoust. Soc. Am. 40, 441–444 (1966).
Florentine, M. Relation between lateralization and loudness in asymmetrical hearing losses. J. Am. Audiol. Soc. 1, 243–251 (1976).
Van Wanrooij, M.M. & Van Opstal, A.J. Sound localization under perturbed binaural hearing. J. Neurophysiol. 97, 715–726 (2007).
Mrsic-Flogel, T.D., King, A.J. & Schnupp, J.W.H. Encoding of virtual acoustic space stimuli by neurons in ferret primary auditory cortex. J. Neurophysiol. 93, 3489–3503 (2005).
Woods, T.M., Lopez, S.E., Long, J.H., Rahman, J.E. & Recanzone, G.H. Effects of stimulus azimuth and intensity on the single-neuron activity in the auditory cortex of the alert macaque monkey. J. Neurophysiol. 96, 3323–3337 (2006).
Jiang, W., Jiang, H. & Stein, B.E. Two corticotectal areas facilitate multisensory orientation behavior. J. Cogn. Neurosci. 14, 1240–1255 (2002).
Hofman, M. & van Opstal, A.J. Binaural weighting of pinna cues in human sound localization. Exp. Brain Res. 148, 458–470 (2003).
Jin, C., Corderoy, A., Carlile, S. & van Schaik, A. Contrasting monaural and interaural spectral cues for human sound localization. J. Acoust. Soc. Am. 115, 3124–3141 (2004).
Eyding, D., Macklis, J.D., Neubacher, U., Funke, K. & Wörgötter, F. Selective elimination of corticogeniculate feedback abolishes the electroencephalogram dependence of primary visual cortical receptive fields and reduces their spatial specificity. J. Neurosci. 23, 7021–7033 (2003).
Davis, K.A., Ramachandran, R. & May, B.J. Single-unit responses in the inferior colliculus of decerebrate cats. II. Sensitivity to interaural level differences. J. Neurophysiol. 82, 164–175 (1999).
Pollak, G.D., Burger, R.M., Park, T.J., Klug, A. & Bauer, E.E. Roles of inhibition for transforming binaural properties in the brainstem auditory system. Hear. Res. 168, 60–78 (2002).
Oliver, D.L. Neuronal organization of the inferior colliculus. in The Inferior Colliculus (eds Winer, J.A. & Schreiner, C.E.) 69–114 (Springer, New York, 2005).
Polley, D.B., Steinberg, E.E. & Merzenich, M.M. Perceptual learning directs auditory cortical map reorganization through top-down influences. J. Neurosci. 26, 4970–4982 (2006).
Kilgard, M.P. & Merzenich, M.M. Cortical map reorganization enabled by nucleus basalis activity. Science 279, 1714–1718 (1998).
Weinberger, N.M. Specific long-term memory traces in primary auditory cortex. Nat. Rev. Neurosci. 5, 279–290 (2004).
Acknowledgements
We are grateful to J.D. Macklis and R. Fricker-Gates for helping us to set up the chromophore-targeted laser photolysis technique and to B. Willmore for statistical advice. K. Allen and A. Fieger assisted with the early stages of the project, and J. Bizley, R. Campbell, D. Kumpik and S. Spires contributed to the behavioral testing and provided valuable discussion. This work was supported by the Wellcome Trust through a Principal Research Fellowship to A.J.K. (WT076508AIA) and a project grant to A.J.K. and D.R.M. (WT069600/Z/02/Z).
Author information
Authors and Affiliations
Contributions
This study was conceived by V.M.B., A.J.K. and D.R.M. and designed by V.M.B. and A.J.K. The behavioral experiments were performed by V.M.B., F.R.N. and A.J.K. The anatomical studies were carried out by V.M.B., who jointly analyzed all of the data with F.R.N. A.J.K., V.M.B. and F.R.N. wrote the paper with assistance from D.R.M.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Tables 1–3 (PDF 685 kb)
Rights and permissions
About this article
Cite this article
Bajo, V., Nodal, F., Moore, D. et al. The descending corticocollicular pathway mediates learning-induced auditory plasticity. Nat Neurosci 13, 253–260 (2010). https://doi.org/10.1038/nn.2466
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2466
This article is cited by
-
Descending projections to the auditory midbrain: evolutionary considerations
Journal of Comparative Physiology A (2023)
-
Individual differences in the attentional modulation of the human auditory brainstem response to speech inform on speech-in-noise deficits
Scientific Reports (2019)
-
Silencing cortical activity during sound-localization training impairs auditory perceptual learning
Nature Communications (2019)
-
Sensory overamplification in layer 5 auditory corticofugal projection neurons following cochlear nerve synaptic damage
Nature Communications (2018)
-
Control of synaptic plasticity in deep cortical networks
Nature Reviews Neuroscience (2018)