Abstract
Brain circuitry processes information by rapidly and selectively engaging functional neuronal networks. The dynamic formation of networks is often evident in rhythmically synchronized neuronal activity and tightly correlates with perceptual, cognitive and motor performances. But how synchronized neuronal activity contributes to network formation and how it relates to the computation of behaviorally relevant information has remained difficult to discern. Here we structure recent empirical advances that link synchronized activity to the activation of so-called dynamic circuit motifs. These motifs explicitly relate (1) synaptic and cellular properties of circuits to (2) identified timescales of rhythmic activation and to (3) canonical circuit computations implemented by rhythmically synchronized circuits. We survey the ubiquitous evidence of specific cell and circuit properties underlying synchronized activity across theta, alpha, beta and gamma frequency bands and show that their activation likely implements gain control, context-dependent gating and state-specific integration of synaptic inputs. This evidence gives rise to the dynamic circuit motifs hypothesis of synchronized activation states, with its core assertion that activation states are linked to uniquely identifiable local circuit structures that are recruited during the formation of functional networks to perform specific computational operations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fell, J. & Axmacher, N. The role of phase synchronization in memory processes. Nat. Rev. Neurosci. 12, 105–118 (2011).
Siegel, M., Donner, T.H. & Engel, A.K. Spectral fingerprints of large-scale neuronal interactions. Nat. Rev. Neurosci. 13, 121–134 (2012).
Buzsáki, G. & Wang, X.J. Mechanisms of gamma oscillations. Annu. Rev. Neurosci. 35, 203–225 (2012).
Tiesinga, P., Fellous, J.M. & Sejnowski, T.J. Regulation of spike timing in visual cortical circuits. Nat. Rev. Neurosci. 9, 97–107 (2008).
Grillner, S., Markram, H., De Schutter, E., Silberberg, G. & LeBeau, F.E. Microcircuits in action–from CPGs to neocortex. Trends Neurosci. 28, 525–533 (2005).
Akam, T. & Kullmann, D.M. Oscillatory multiplexing of population codes for selective communication in the mammalian brain. Nat. Rev. Neurosci. 15, 111–122 (2014).
Silver, R.A. Neuronal arithmetic. Nat. Rev. Neurosci. 11, 474–489 (2010).
Karnani, M.M., Agetsuma, M. & Yuste, R. A blanket of inhibition: functional inferences from dense inhibitory connectivity. Curr. Opin. Neurobiol. 26C, 96–102 (2014).
Wang, X.J. Neurophysiological and computational principles of cortical rhythms in cognition. Physiol. Rev. 90, 1195–1268 (2010).
Kepecs, A. & Fishell, G. Interneuron cell types are fit to function. Nature 505, 318–326 (2014).
Bruno, R.M. Synchrony in sensation. Curr. Opin. Neurobiol. 21, 701–708 (2011).
Pouille, F., Marin-Burgin, A., Adesnik, H., Atallah, B.V. & Scanziani, M. Input normalization by global feedforward inhibition expands cortical dynamic range. Nat. Neurosci. 12, 1577–1585 (2009).
Kremkow, J., Aertsen, A. & Kumar, A. Gating of signal propagation in spiking neural networks by balanced and correlated excitation and inhibition. J. Neurosci. 30, 15760–15768 (2010).
Kremkow, J., Perrinet, L.U., Masson, G.S. & Aertsen, A. Functional consequences of correlated excitatory and inhibitory conductances in cortical networks. J. Comput. Neurosci. 28, 579–594 (2010).
Zemankovics, R., Veres, J.M., Oren, I. & Hajos, N. Feedforward inhibition underlies the propagation of cholinergically induced gamma oscillations from hippocampal CA3 to CA1. J. Neurosci. 33, 12337–12351 (2013).
Cruikshank, S.J., Urabe, H., Nurmikko, A.V. & Connors, B.W. Pathway-specific feedforward circuits between thalamus and neocortex revealed by selective optical stimulation of axons. Neuron 65, 230–245 (2010).
Thomson, A.M. & Bannister, A.P. Interlaminar connections in the neocortex. Cereb. Cortex 13, 5–14 (2003).
Kumar, A., Vlachos, I., Aertsen, A. & Boucsein, C. Challenges of understanding brain function by selective modulation of neuronal subpopulations. Trends Neurosci. 36, 579–586 (2013).
Lee, A.T. et al. Pyramidal neurons in prefrontal cortex receive subtype-specific forms of excitation and inhibition. Neuron 81, 61–68 (2014).
Akam, T. & Kullmann, D.M. Oscillations and filtering networks support flexible routing of information. Neuron 67, 308–320 (2010).
Timofeev, I., Bazhenov, M., Seigneur, J. & Sejnowski, T. Neuronal synchronization and thalamocortical rhythms in sleep, wake and epilepsy. in Jasper's Basic Mechanisms of the Epilepsies (eds. Noebels, J.L., Avoli, M., Rogawski, M.A., Olsen, R.W. & Delgado-Escueta, A.V.) (National Center for Biotechnology Information, Bethesda, Maryland, USA, 2012).
Hutcheon, B. & Yarom, Y. Resonance, oscillation and the intrinsic frequency preferences of neurons. Trends Neurosci. 23, 216–222 (2000).
Otte, S., Hasenstaub, A. & Callaway, E.M. Cell type-specific control of neuronal responsiveness by gamma-band oscillatory inhibition. J. Neurosci. 30, 2150–2159 (2010).
Lawrence, J.J. Cholinergic control of GABA release: emerging parallels between neocortex and hippocampus. Trends Neurosci. 31, 317–327 (2008).
Fröhlich, F. & McCormick, D.A. Endogenous electric fields may guide neocortical network activity. Neuron 67, 129–143 (2010).
Moca, V.V., Nikolic, D., Singer, W. & Muresan, R.C. Membrane resonance enables stable and robust gamma oscillations. Cereb. Cortex 24, 119–142 (2014).
Sohal, V.S., Zhang, F., Yizhar, O. & Deisseroth, K. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459, 698–702 (2009).
Patel, M. & Joshi, B. Decoding synchronized oscillations within the brain: phase-delayed inhibition provides a robust mechanism for creating a sharp synchrony filter. J. Theor. Biol. 334, 13–25 (2013).
Bollimunta, A., Mo, J., Schroeder, C.E. & Ding, M. Neuronal mechanisms and attentional modulation of corticothalamic alpha oscillations. J. Neurosci. 31, 4935–4943 (2011).
Buffalo, E.A., Fries, P., Landman, R., Buschman, T.J. & Desimone, R. Laminar differences in gamma and alpha coherence in the ventral stream. Proc. Natl. Acad. Sci. USA 108, 11262–11267 (2011).
Saalmann, Y.B., Pinsk, M.A., Wang, L., Li, X. & Kastner, S. The pulvinar regulates information transmission between cortical areas based on attention demands. Science 337, 753–756 (2012).
Lorincz, M.L., Kekesi, K.A., Juhasz, G., Crunelli, V. & Hughes, S.W. Temporal framing of thalamic relay-mode firing by phasic inhibition during the alpha rhythm. Neuron 63, 683–696 (2009).
Constantinople, C.M. & Bruno, R.M. Deep cortical layers are activated directly by thalamus. Science 340, 1591–1594 (2013).
Hughes, S.W. et al. Thalamic gap junctions control local neuronal synchrony and influence macroscopic oscillation amplitude during EEG alpha rhythms. Front. Psychol. 2, 193 (2011).
Silva, L.R., Amitai, Y. & Connors, B.W. Intrinsic oscillations of neocortex generated by layer 5 pyramidal neurons. Science 251, 432–435 (1991).
Castro-Alamancos, M.A. & Rigas, P. Synchronized oscillations caused by disinhibition in rodent neocortex are generated by recurrent synaptic activity mediated by AMPA receptors. J. Physiol. (Lond.) 542, 567–581 (2002).
Olsen, S.R., Bortone, D.S., Adesnik, H. & Scanziani, M. Gain control by layer six in cortical circuits of vision. Nature 483, 47–52 (2012).
Castro-Alamancos, M.A. & Oldford, E. Cortical sensory suppression during arousal is due to the activity-dependent depression of thalamocortical synapses. J. Physiol. (Lond.) 541, 319–331 (2002).
Florez, C.M. et al. In vitro recordings of human neocortical oscillations. Cereb. Cortex doi:10.1093/cercor/bht235 17 September (2013).
Jensen, O. & Mazaheri, A. Shaping functional architecture by oscillatory alpha activity: gating by inhibition. Front. Hum. Neurosci. 4, 186 (2010).
Fischer, I. et al. Zero-lag long-range synchronization via dynamical relaying. Phys. Rev. Lett. 97, 123902 (2006).
Helfrich, R.F. et al. Entrainment of brain oscillations by transcranial alternating current stimulation. Curr. Biol. 24, 333–339 (2014).
Saalmann, Y.B. & Kastner, S. Cognitive and perceptual functions of the visual thalamus. Neuron 71, 209–223 (2011).
Sherman, S.M. & Guillery, R.W. Exploring the Thalamus and its Role in Cortical Function (MIT Press, 2006).
Petersen, C.C. & Crochet, S. Synaptic computation and sensory processing in neocortical layer 2/3. Neuron 78, 28–48 (2013).
Vinck, M., Womelsdorf, T., Buffalo, E.A., Desimone, R. & Fries, P. Attentional modulation of cell-class-specific gamma-band synchronization in awake monkey area v4. Neuron 80, 1077–1089 (2013).
Gregoriou, G.G., Gotts, S.J. & Desimone, R. Cell-type-specific synchronization of neural activity in FEF with V4 during attention. Neuron 73, 581–594 (2012).
Spaak, E., Bonnefond, M., Maier, A., Leopold, D.A. & Jensen, O. Layer-specific entrainment of gamma-band neural activity by the alpha rhythm in monkey visual cortex. Curr. Biol. 22, 2313–2318 (2012).
Thut, G., Miniussi, C. & Gross, J. The functional importance of rhythmic activity in the brain. Curr. Biol. 22, R658–R663 (2012).
de Almeida, L., Idiart, M. & Lisman, J.E. The input-output transformation of the hippocampal granule cells: from grid cells to place fields. J. Neurosci. 29, 7504–7512 (2009).
Tiesinga, P. & Sejnowski, T.J. Cortical enlightenment: are attentional gamma oscillations driven by ING or PING? Neuron 63, 727–732 (2009).
Börgers, C., Epstein, S. & Kopell, N.J. Gamma oscillations mediate stimulus competition and attentional selection in a cortical network model. Proc. Natl. Acad. Sci. USA 105, 18023–18028 (2008).
Cardin, J.A. et al. Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459, 663–667 (2009).
Womelsdorf, T. et al. Orientation selectivity and noise correlation in awake monkey area V1 are modulated by the gamma cycle. Proc. Natl. Acad. Sci. USA 109, 4302–4307 (2012).
Vinck, M. et al. Gamma-phase shifting in awake monkey visual cortex. J. Neurosci. 30, 1250–1257 (2010).
Tiesinga, P.H. & Sejnowski, T.J. Mechanisms for phase shifting in cortical networks and their role in communication through coherence. Front. Hum. Neurosci. 4, 196 (2010).
Knoblich, U., Siegle, J.H., Pritchett, D.L. & Moore, C.I. What do we gain from gamma? Local dynamic gain modulation drives enhanced efficacy and efficiency of signal transmission. Front. Hum. Neurosci. 4, 185 (2010).
Azouz, R. & Gray, C.M. Adaptive coincidence detection and dynamic gain control in visual cortical neurons in vivo. Neuron 37, 513–523 (2003).
Bosman, C.A. et al. Attentional stimulus selection through selective synchronization between monkey visual areas. Neuron 75, 875–888 (2012).
Womelsdorf, T. et al. Modulation of neuronal interactions through neuronal synchronization. Science 316, 1609–1612 (2007).
Womelsdorf, T., Fries, P., Mitra, P.P. & Desimone, R. Gamma-band synchronization in visual cortex predicts speed of change detection. Nature 439, 733–736 (2006).
Sippy, T. & Yuste, R. Decorrelating action of inhibition in neocortical networks. J. Neurosci. 33, 9813–9830 (2013).
Tetzlaff, T., Helias, M., Einevoll, G.T. & Diesmann, M. Decorrelation of neural-network activity by inhibitory feedback. PLoS Comput. Biol. 8, e1002596 (2012).
Yizhar, O. et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature 477, 171–178 (2011).
Wilson, N.R., Runyan, C.A., Wang, F.L. & Sur, M. Division and subtraction by distinct cortical inhibitory networks in vivo. Nature 488, 343–348 (2012).
Lee, S.H., Kwan, A.C. & Dan, Y. Interneuron subtypes and orientation tuning. Nature 508, E1–E2 (2014).
Kopell, N., Whittington, M.A. & Kramer, M.A. Neuronal assembly dynamics in the beta1 frequency range permits short-term memory. Proc. Natl. Acad. Sci. USA 108, 3779–3784 (2011).
Kramer, M.A. et al. Rhythm generation through period concatenation in rat somatosensory cortex. PLoS Comput. Biol. 4, e1000169 (2008).
Roopun, A.K. et al. Period concatenation underlies interactions between gamma and beta rhythms in neocortex. Front. Cell. Neurosci. 2, 1 (2008).
Salazar, R.F., Dotson, N.M., Bressler, S.L. & Gray, C.M. Content-specific fronto-parietal synchronization during visual working memory. Science 338, 1097–1100 (2012).
Lipsman, N. et al. Beta coherence within human ventromedial prefrontal cortex precedes affective value choices. Neuroimage 85, 769–778 (2014).
Larkum, M. A cellular mechanism for cortical associations: an organizing principle for the cerebral cortex. Trends Neurosci. 36, 141–151 (2013).
Lovett-Barron, M. et al. Regulation of neuronal input transformations by tunable dendritic inhibition. Nat. Neurosci. 15, 423–430 (2012).
Palmer, L., Murayama, M. & Larkum, M. Inhibitory regulation of dendritic activity in vivo. Front. Neural Circuits 6, 26 (2012).
Wang, X.J., Tegner, J., Constantinidis, C. & Goldman-Rakic, P.S. Division of labor among distinct subtypes of inhibitory neurons in a cortical microcircuit of working memory. Proc. Natl. Acad. Sci. USA 101, 1368–1373 (2004).
Leão, R.N. et al. OLM interneurons differentially modulate CA3 and entorhinal inputs to hippocampal CA1 neurons. Nat. Neurosci. 15, 1524–1530 (2012).
Gloveli, T. et al. Differential involvement of oriens/pyramidale interneurones in hippocampal network oscillations in vitro. J. Physiol. (Lond.) 562, 131–147 (2005).
Varga, C., Golshani, P. & Soltesz, I. Frequency-invariant temporal ordering of interneuronal discharges during hippocampal oscillations in awake mice. Proc. Natl. Acad. Sci. USA 109, E2726–E2734 (2012).
Colgin, L.L. et al. Frequency of gamma oscillations routes flow of information in the hippocampus. Nature 462, 353–357 (2009).
Hasselmo, M.E., Bodelon, C. & Wyble, B.P. A proposed function for hippocampal theta rhythm: separate phases of encoding and retrieval enhance reversal of prior learning. Neural Comput. 14, 793–817 (2002).
Li, X., Morita, K., Robinson, H.P. & Small, M. Control of layer 5 pyramidal cell spiking by oscillatory inhibition in the distal apical dendrites: a computational modeling study. J. Neurophysiol. 109, 2739–2756 (2013).
Mancilla, J.G., Lewis, T.J., Pinto, D.J., Rinzel, J. & Connors, B.W. Synchronization of electrically coupled pairs of inhibitory interneurons in neocortex. J. Neurosci. 27, 2058–2073 (2007).
Jiang, X., Wang, G., Lee, A.J., Stornetta, R.L. & Zhu, J.J. The organization of two new cortical interneuronal circuits. Nat. Neurosci. 16, 210–218 (2013).
Metherate, R. & Cruikshank, S.J. Thalamocortical inputs trigger a propagating envelope of gamma-band activity in auditory cortex in vitro. Exp. Brain Res. 126, 160–174 (1999).
Robbe, D. & Buzsaki, G. Alteration of theta timescale dynamics of hippocampal place cells by a cannabinoid is associated with memory impairment. J. Neurosci. 29, 12597–12605 (2009).
Buzsáki, G., Logothetis, N. & Singer, W. Scaling brain size, keeping timing: evolutionary preservation of brain rhythms. Neuron 80, 751–764 (2013).
Drion, G., Massotte, L., Sepulchre, R. & Seutin, V. How modeling can reconcile apparently discrepant experimental results: the case of pacemaking in dopaminergic neurons. PLoS Comput. Biol. 7, e1002050 (2011).
Hentschke, H. et al. Altered GABAA,slow inhibition and network oscillations in mice lacking the GABAA receptor β3 subunit. J. Neurophysiol. 102, 3643–3655 (2009).
White, J.A., Banks, M.I., Pearce, R.A. & Kopell, N.J. Networks of interneurons with fast and slow gamma-aminobutyric acid type A (GABAA) kinetics provide substrate for mixed gamma-theta rhythm. Proc. Natl. Acad. Sci. USA 97, 8128–8133 (2000).
Capogna, M. & Pearce, R.A. GABAA,slow: causes and consequences. Trends Neurosci. 34, 101–112 (2011).
English, D.F. et al. GABAergic circuits mediate the reinforcement-related signals of striatal cholinergic interneurons. Nat. Neurosci. 15, 123–130 (2012).
Molyneaux, B.J. & Hasselmo, M.E. GABAB presynaptic inhibition has an in vivo time constant sufficiently rapid to allow modulation at theta frequency. J. Neurophysiol. 87, 1196–1205 (2002).
Blatow, M. et al. A novel network of multipolar bursting interneurons generates theta frequency oscillations in neocortex. Neuron 38, 805–817 (2003).
Chapman, C.A. & Lacaille, J.C. Intrinsic theta-frequency membrane potential oscillations in hippocampal CA1 interneurons of stratum lacunosum-moleculare. J. Neurophysiol. 81, 1296–1307 (1999).
Glasgow, S.D. & Chapman, C.A. Conductances mediating intrinsic theta-frequency membrane potential oscillations in layer II parasubicular neurons. J. Neurophysiol. 100, 2746–2756 (2008).
Boehlen, A., Henneberger, C., Heinemann, U. & Erchova, I. Contribution of near-threshold currents to intrinsic oscillatory activity in rat medial entorhinal cortex layer II stellate cells. J. Neurophysiol. 109, 445–463 (2013).
Beierlein, M., Gibson, J.R. & Connors, B.W. A network of electrically coupled interneurons drives synchronized inhibition in neocortex. Nat. Neurosci. 3, 904–910 (2000).
Stark, E. et al. Inhibition-induced theta resonance in cortical circuits. Neuron 80, 1263–1276 (2013).
Fanselow, E.E., Richardson, K.A. & Connors, B.W. Selective, state-dependent activation of somatostatin-expressing inhibitory interneurons in mouse neocortex. J. Neurophysiol. 100, 2640–2652 (2008).
Hughes, S.W. et al. Synchronized oscillations at alpha and theta frequencies in the lateral geniculate nucleus. Neuron 42, 253–268 (2004).
Karameh, F.N., Dahleh, M.A., Brown, E.N. & Massaquoi, S.G. Modeling the contribution of lamina 5 neuronal and network dynamics to low frequency EEG phenomena. Biol. Cybern. 95, 289–310 (2006).
Castro-Alamancos, M.A. The motor cortex: a network tuned to 7–14 Hz. Front. Neural Circuits 7, 21 (2013).
Castro-Alamancos, M.A., Rigas, P. & Tawara-Hirata, Y. Resonance (approximately 10 Hz) of excitatory networks in motor cortex: effects of voltage-dependent ion channel blockers. J. Physiol. (Lond.) 578, 173–191 (2007).
Roopun, A.K. et al. A beta2-frequency (20–30 Hz) oscillation in nonsynaptic networks of somatosensory cortex. Proc. Natl. Acad. Sci. USA 103, 15646–15650 (2006).
Brumberg, J.C., Nowak, L.G. & McCormick, D.A. Ionic mechanisms underlying repetitive high-frequency burst firing in supragranular cortical neurons. J. Neurosci. 20, 4829–4843 (2000).
Cunningham, M.O. et al. A role for fast rhythmic bursting neurons in cortical gamma oscillations in vitro. Proc. Natl. Acad. Sci. USA 101, 7152–7157 (2004).
Cardin, J.A., Palmer, L.A. & Contreras, D. Stimulus-dependent gamma (30–50 Hz) oscillations in simple and complex fast rhythmic bursting cells in primary visual cortex. J. Neurosci. 25, 5339–5350 (2005).
Wang, X.J. Fast burst firing and short-term synaptic plasticity: a model of neocortical chattering neurons. Neuroscience 89, 347–362 (1999).
Hughes, S.W. et al. Novel modes of rhythmic burst firing at cognitively-relevant frequencies in thalamocortical neurons. Brain Res. 1235, 12–20 (2008).
Whittington, M.A., Traub, R.D. & Jefferys, J.G. Synchronized oscillations in interneuron networks driven by metabotropic glutamate receptor activation. Nature 373, 612–615 (1995).
Hormuzdi, S.G. et al. Impaired electrical signaling disrupts gamma frequency oscillations in connexin 36-deficient mice. Neuron 31, 487–495 (2001).
Xu, H., Jeong, H.Y., Tremblay, R. & Rudy, B. Neocortical somatostatin-expressing GABAergic interneurons disinhibit the thalamorecipient layer 4. Neuron 77, 155–167 (2013).
Pfeffer, C.K., Xue, M., He, M., Huang, Z.J. & Scanziani, M. Inhibition of inhibition in visual cortex: the logic of connections between molecularly distinct interneurons. Nat. Neurosci. 16, 1068–1076 (2013).
Gibson, J.R., Beierlein, M. & Connors, B.W. Two networks of electrically coupled inhibitory neurons in neocortex. Nature 402, 75–79 (1999).
Swadlow, H.A. Fast-spike interneurons and feedforward inhibition in awake sensory neocortex. Cereb. Cortex 13, 25–32 (2003).
Hu, H., Ma, Y. & Agmon, A. Submillisecond firing synchrony between different subtypes of cortical interneurons connected chemically but not electrically. J. Neurosci. 31, 3351–3361 (2011).
Berger, T.K., Silberberg, G., Perin, R. & Markram, H. Brief bursts self-inhibit and correlate the pyramidal network. PLoS Biol. 8, e1000473 (2010).
Flint, A.C. & Connors, B.W. Two types of network oscillations in neocortex mediated by distinct glutamate receptor subtypes and neuronal populations. J. Neurophysiol. 75, 951–957 (1996).
Murayama, M. et al. Dendritic encoding of sensory stimuli controlled by deep cortical interneurons. Nature 457, 1137–1141 (2009).
Carracedo, L.M. et al. A neocortical delta rhythm facilitates reciprocal interlaminar interactions via nested theta rhythms. J. Neurosci. 33, 10750–10761 (2013).
Acknowledgements
We are grateful to the speakers of two workshops held at the Computational and Systems Neuroscience (cosyne.org) meetings on The Consequences of Brain Rhythms in the Organization of Neuronal Computation (2009) and Developing Simplified Algebras to Describe Large-Scale Brain Dynamics (2011) for numerous discussions. We thank T. Donner, C. Eliasmith, B. Hansen and M. Vinck for discussions and comments on an earlier version of the manuscript. T.W. was supported by grants from the Canadian Institutes of Health Research (CIHR), the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Ontario Ministry of Economic Development and Innovation (MEDI). T.A.V. was supported by grants from the CIHR. N.T.S. was supported by US National Institutes of Health (NIH) R01 grants NS18741, NS44623 and grant HD 18381, NIH Institutional Training Grant T32 MH070328, and the US National Center for Research Resources (P41 RR14075). P.T. was supported by a grant from the Netherlands Organization for Scientific Research (NWO) Computational Lifesciences program and by Neuroseeker (FP7 grant agreement 600925).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Thalamo-cortical alpha rhythmic motif.
(a) The structural connectivity (top) shows that the alpha rhythmic motif depends on rhythmically bursting, high threshold (HT) cells entraining inhibitory neurons (IN), which in turn impose rhythmic suppression onto relay-mode, regular spiking projection neurons. The bursting frequency of the HT cells depends on intrinsic currents, whereas the synchrony derives from coupling by gap junctions. Depending on whether the IN cells fire single spikes or burst, the relay cells are either in-phase suppressed or anti-phase suppressed. The bottom panels shows the cross correlograms illustrating the rhythmic activation of the interneurons (Int) by bursting HT cells (left) and the alpha rhythmic inhibition of relay mode cells by an interneurons (right). (b) Example activation traces showing the relation of interneuron IPSPs (bottom) on relay mode neuron spike generation (middle) and the extracellular recorded filtered LFP (top). The phase-of-firing histogram (right) shows that during alpha rhythmic pulsed inhibition relay neuron firing is facilitated particularly in the third quarter of the oscillation cycle. Adapted from Lorincz, M.L., Kekesi, K.A., Juhasz, G., Crunelli, V. & Hughes, S.W. Temporal framing of thalamic relay-mode firing by phasic inhibition during the alpha rhythm. Neuron 63, 683-696, copyright Elsevier (2009).
Supplementary Figure 2 Selective gamma coherence during selective visual processing.
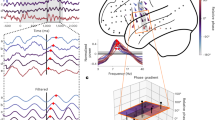
(a) The spatial coverage of ECoG electrode locations (dots) projected on the rendered cortical surface of a macaque brain. Dots indicate the 252 electrodes of a high density ECoG grid. Green and red dots indicate electrodes recording activity over V1 and V4, respectively. ‘V1a’ and ‘V1b’ denote two V1 electrode locations recording non-overlapping receptive fields. (b) Stimulus arrangement for the two attention conditions was identical. In one condition attention was directed to the stimulus that activated V4 and the V1a site (stimulus circled in red for illustration purposes). In the other condition attention was directed to the stimulus that activated V4 and the V1b site (stimulus circled in blue for illustration purposes). (c) Spectral power change relative to pre-stimulus baseline (upper panel) in V1 in the two attention conditions (red and blue). The bottom panel shows the coherence spectra of the V4 to V1a and V1b (red and blue) when the attended stimulus overlaid V1a (red) and V1b (blue). (d) Illustration of the attention condition (left). Bottom-Up Granger causal influence for the V1a to V4 (red) and the V1b to V4 (blue) connections when the attended stimulus overlaid with V4 and V1a. The rightmost panel shows the respective Top-Down Granger causal influence. (e) Same format as (d) but for the condition with attention on the stimulus that activated the V4 and the V1b recording site. Gray bars in (c,d,e) indicate the frequencies with a significant effect (p<0.05, corrected for multiple comparisons across frequencies, non-parametric randomization across site pairs). Adapted from Bosman, C.A., et al. Attentional Stimulus Selection through Selective Synchronization between Monkey Visual Areas. Neuron 75, 875-888, copyright Elsevier (2012).
Supplementary Figure 3 Visual attention and selective ‘push-pull’ gating in visual cortex at gamma band frequencies mediated by putative interneurons.
(a) Normalized action potential waveforms of single cells recorded in area V4. (b) Distribution of peak-to-trough durations of action potentials across neurons reveals a bimodal distribution that separate narrow spiking from broad spiking cells. (c) Spike-LFP phase locking (measured as pairwise phase consistency) across cells in V4 for narrow and broad spiking cells shows significant gamma band synchronization for NS and BS cells, and significant alpha band synchronization for NS cells. (d,e) Attention (attend inside, PPCin, versus outside, the visual receptive field of the cells, PPCout) decreases gamma band phase locking for NS and BS cells that show low firing rates (d), and increases phase locking for cells with higher firing rates (e). The respective gamma bands are highlighted with grey bars in the top of the panels. This finding suggests a push-pull mechanism of attention that implements the up- and down-modulation of cells’ synchronization depending on their overall rate in response to visual stimuli. Low/High firing rate cells were median split. Adapted from Vinck, M., Womelsdorf, T., Buffalo, E.A., Desimone, R. & Fries, P. Attentional modulation of cell-class-specific gamma-band synchronization in awake monkey area v4. Neuron 80, 1077-1089, copyright Elsevier (2013).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 (PDF 5557 kb)
Rights and permissions
About this article
Cite this article
Womelsdorf, T., Valiante, T., Sahin, N. et al. Dynamic circuit motifs underlying rhythmic gain control, gating and integration. Nat Neurosci 17, 1031–1039 (2014). https://doi.org/10.1038/nn.3764
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3764
This article is cited by
-
Deep psychophysiological phenotyping of adolescents and adults with 22q11.2 deletion syndrome: a multilevel approach to defining core disease processes
BMC Psychiatry (2023)
-
Cell-type-specific plasticity of inhibitory interneurons in the rehabilitation of auditory cortex after peripheral damage
Nature Communications (2023)
-
Exploratory evidence for differences in GABAergic regulation of auditory processing in autism spectrum disorder
Translational Psychiatry (2023)
-
Neural attentional-filter mechanisms of listening success in middle-aged and older individuals
Nature Communications (2021)
-
Electrophysiological biomarkers of behavioral dimensions from cross-species paradigms
Translational Psychiatry (2021)