Abstract
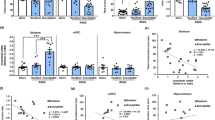
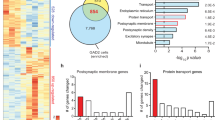
To better understand the molecular mechanisms of depression and antidepressant action, we administered chronic social defeat stress followed by chronic imipramine (a tricyclic antidepressant) to mice and studied adaptations at the levels of gene expression and chromatin remodeling of five brain-derived neurotrophic factor (Bdnf) splice variant mRNAs (I–V) and their unique promoters in the hippocampus. Defeat stress induced lasting downregulation of Bdnf transcripts III and IV and robustly increased repressive histone methylation at their corresponding promoters. Chronic imipramine reversed this downregulation and increased histone acetylation at these promoters. This hyperacetylation by chronic imipramine was associated with a selective downregulation of histone deacetylase (Hdac) 5. Furthermore, viral-mediated HDAC5 overexpression in the hippocampus blocked imipramine's ability to reverse depression-like behavior. These experiments underscore an important role for histone remodeling in the pathophysiology and treatment of depression and highlight the therapeutic potential for histone methylation and deacetylation inhibitors in depression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Nestler, E.J. et al. Neurobiology of depression. Neuron 34, 13–25 (2002).
Manji, H.K., Drevets, W.C. & Charney, D.S. The cellular neurobiology of depression. Nat. Med. 7, 541–547 (2001).
Sheline, Y.I., Gado, M.H. & Kraemer, H.C. Untreated depression and hippocampal volume loss. Am. J. Psychiatry 160, 1516–1518 (2003).
Duman, R.S. Depression: a case of neuronal life and death? Biol. Psychiatry 56, 140–145 (2004).
McEwen, B.S., Magarinos, A.M. & Reagan, L.P. Structural plasticity and tianeptine: cellular and molecular targets. Eur. Psychiatry 17 (suppl.), 318–330 (2002).
Sapolsky, R.M. Glucocorticoids and hippocampal atrophy in neuropsychiatric disorders. Arch. Gen. Psychiatry 57, 925–935 (2000).
Shirayama, Y., Chen, A.C., Nakagawa, S., Russell, D.S. & Duman, R.S. Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J. Neurosci. 22, 3251–3261 (2002).
Monteggia, L.M. et al. Essential role of brain-derived neurotrophic factor in adult hippocampal function. Proc. Natl. Acad. Sci. USA 101, 10827–10832 (2004).
Smith, M.A., Makino, S., Kvetnansky, R. & Post, R.M. Stress and glucocorticoids affect the expression of brain-derived neurotrophic factor and neurotrophin-3 mRNAs in the hippocampus. J. Neurosci. 15, 1768–1777 (1995).
Nibuya, M., Morinobu, S. & Duman, R.S. Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J. Neurosci. 15, 7539–7547 (1995).
Schaaf, M.J., de Jong, J., de Kloet, E.R. & Vreugdenhil, E. Downregulation of BDNF mRNA and protein in the rat hippocampus by corticosterone. Brain Res. 813, 112–120 (1998).
Pizarro, J.M. et al. Acute social defeat reduces neurotrophin expression in brain cortical and subcortical areas in mice. Brain Res. 1025, 10–20 (2004).
Russo-Neustadt, A., Beard, R.C. & Cotman, C.W. Exercise, antidepressant medications, and enhanced brain derived neurotrophic factor expression. Neuropsychopharmacology 21, 679–682 (1999).
Timmusk, T. et al. Multiple promoters direct tissue-specific expression of the rat BDNF gene. Neuron 10, 475–489 (1993).
Tapia-Arancibia, L., Rage, F., Givalois, L. & Arancibia, S. Physiology of BDNF: focus on hypothalamic function. Front. Neuroendocrinol. 25, 77–107 (2004).
Felsenfeld, G. & Groudine, M. Controlling the double helix. Nature 421, 448–453 (2003).
Jenuwein, T. & Allis, C.D. Translating the histone code. Science 293, 1074–1080 (2001).
Lachner, M., O'Sullivan, R.J. & Jenuwein, T. An epigenetic road map for histone lysine methylation. J. Cell Sci. 116, 2117–2124 (2003).
Lachner, M. & Jenuwein, T. The many faces of histone lysine methylation. Curr. Opin. Cell Biol. 14, 286–298 (2002).
Lunyak, V.V. et al. Corepressor-dependent silencing of chromosomal regions encoding neuronal genes. Science 298, 1747–1752 (2002).
Hsieh, J. & Gage, F.H. Epigenetic control of neural stem cell fate. Curr. Opin. Genet. Dev. 14, 461–469 (2004).
Steffan, J.S. et al. Histone deacetylase inhibitors arrest polyglutamine-dependent neurodegeneration in Drosophila. Nature 413, 739–743 (2001).
Hockly, E. et al. Suberoylanilide hydroxamic acid, a histone deacetylase inhibitor, ameliorates motor deficits in a mouse model of Huntington's disease. Proc. Natl. Acad. Sci. USA 100, 2041–2046 (2003).
Hoshino, M. et al. Histone deacetylase activity is retained in primary neurons expressing mutant huntingtin protein. J. Neurochem. 87, 257–267 (2003).
Etchegaray, J.P., Lee, C., Wade, P.A. & Reppert, S.M. Rhythmic histone acetylation underlies transcription in the mammalian circadian clock. Nature 421, 177–182 (2003).
Huang, Y., Doherty, J.J. & Dingledine, R. Altered histone acetylation at glutamate receptor 2 and brain-derived neurotrophic factor genes is an early event triggered by status epilepticus. J. Neurosci. 22, 8422–8428 (2002).
Tsankova, N.M., Kumar, A. & Nestler, E.J. Histone modifications at gene promoter regions in rat hippocampus after acute and chronic electroconvulsive seizures. J. Neurosci. 24, 5603–5610 (2004).
Yeh, S.H., Lin, C.H. & Gean, P.W. Acetylation of nuclear factor-kappaB in rat amygdala improves long-term but not short-term retention of fear memory. Mol. Pharmacol. 65, 1286–1292 (2004).
Levenson, J.M. et al. Regulation of histone acetylation during memory formation in the hippocampus. J. Biol. Chem. 279, 40545–40559 (2004).
Korzus, E., Rosenfeld, M.G. & Mayford, M. CBP histone acetyltransferase activity is a critical component of memory consolidation. Neuron 42, 961–972 (2004).
Levenson, J.M. & Sweatt, J.D. Epigenetic mechanisms in memory formation. Nat. Rev. Neurosci. 6, 108–118 (2005).
Guan, Z. et al. Integration of long-term-memory-related synaptic plasticity involves bidirectional regulation of gene expression and chromatin structure. Cell 111, 483–493 (2002).
Kumar, A. et al. Chromatin remodeling is a key mechanism underlying cocaine-induced plasticity in striatum. Neuron 48, 303–314 (2005).
Buwalda, B. et al. Long-term effects of social stress on brain and behavior: a focus on hippocampal functioning. Neurosci. Biobehav. Rev. 29, 83–97 (2005).
Koolhaas, J.M., De Boer, S.F., De Rutter, A.J., Meerlo, P. & Sgoifo, A. Social stress in rats and mice. Acta Physiol. Scand. Suppl. 640, 69–72 (1997).
Fuchs, E. & Flugge, G. Chronic social stress: effects on limbic brain structures. Physiol. Behav. 79, 417–427 (2003).
Miczek, K.A., Covington, H.E. III, Nikulina, E.M. Jr. & Hammer, R.P. Aggression and defeat: persistent effects on cocaine self-administration and gene expression in peptidergic and aminergic mesocorticolimbic circuits. Neurosci. Biobehav. Rev. 27, 787–802 (2004).
Fuchs, E. & Flugge, G. Social stress in tree shrews: effects on physiology, brain function, and behavior of subordinate individuals. Pharmacol. Biochem. Behav. 73, 247–258 (2002).
Berton, O. et al. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science 311, 864–868 (2006).
Dias, B.G., Banerjee, S.B., Duman, R.S. & Vaidya, V.A. Differential regulation of brain derived neurotrophic factor transcripts by antidepressant treatments in the adult rat brain. Neuropharmacology 45, 553–563 (2003).
Turner, B.M. Cellular memory and the histone code. Cell 111, 285–291 (2002).
Clark, M.S. et al. Overexpression of 5–HT1B receptor in dorsal raphe nucleus using Herpes Simplex Virus gene transfer increases anxiety behavior after inescapable stress. J. Neurosci. 22, 4550–4562 (2002).
Barrot, M. et al. CREB activity in the nucleus accumbens shell controls gating of behavioral responses to emotional stimuli. Proc. Natl. Acad. Sci. USA 99, 11435–11440 (2002).
Lucki, I. The forced swimming test as a model for core and component behavioral effects of antidepressant drugs. Behav. Pharmacol. 8, 523–532 (1997).
Kubicek, S. & Jenuwein, T. A crack in histone lysine methylation. Cell 119, 903–906 (2004).
Chen, J. et al. Activity-induced expression of common reference genes in individual cns neurons. Lab. Invest. 81, 913–916 (2001).
Newton, S.S. et al. Gene profile of electroconvulsive seizures: induction of neurotrophic and angiogenic factors. J. Neurosci. 23, 10841–10851 (2003).
Weaver, I.C. et al. Epigenetic programming by maternal behavior. Nat. Neurosci. 7, 847–854 (2004).
Clark, S.J., Harrison, J., Paul, C.L. & Frommer, M. High sensitivity mapping of methylated cytosines. Nucleic Acids Res. 22, 2990–2997 (1994).
Dennis, K. & Levitt, P. Expression of brain derived neurotrophic factor (BDNF) is correlated with dynamic patterns of promoter methylation in the developing mouse forebrain. Brain Res. Mol. Brain Res. 140, 1–9 (2005).
Acknowledgements
We thank D. Theobald and T. Sasaki for helping with some of the mouse injections; K. Dennis (Vanderbilt University) for supplying the sodium bisulfite protocol for DNA methylation; and G. Petrov for graphic design expertise. This work was supported by grants from the US National Institute of Mental Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Structure of the Bdnf gene. (PDF 953 kb)
Supplementary Fig. 2
Changes in the H3-K27 di-methylation are specific for the Bdnf P3 and P4 promoters. (PDF 322 kb)
Supplementary Fig. 3
Changes in the H3 hyperacetylation and H3-K4 di-methylation are specific for the Bdnf P3 and P4 promoters. (PDF 451 kb)
Supplementary Fig. 4
H4 acetylation levels at the Bdnf P3 promoter are not changed after chronic defeat stress and imipramine treaments. (PDF 192 kb)
Supplementary Table 1
Locomotor activity in social defeat and control mice. (PDF 59 kb)
Supplementary Table 2
List of all primer sequences used. (PDF 57 kb)
Rights and permissions
About this article
Cite this article
Tsankova, N., Berton, O., Renthal, W. et al. Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nat Neurosci 9, 519–525 (2006). https://doi.org/10.1038/nn1659
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1659
This article is cited by
-
Contribution of changes in the orexin system and energy sensors in the brain in depressive disorder - a study in an animal model
Pharmacological Reports (2024)
-
Fatty Acid Amide Hydrolase and Cannabinoid Receptor Type 1 Genes Regulation is Modulated by Social Isolation in Rats
Neurochemical Research (2024)
-
Post-stroke depression: epigenetic and epitranscriptomic modifications and their interplay with gut microbiota
Molecular Psychiatry (2023)
-
Automatically annotated motion tracking identifies a distinct social behavioral profile following chronic social defeat stress
Nature Communications (2023)
-
Epigenetic and epitranscriptomic regulation of axon regeneration
Molecular Psychiatry (2023)