Abstract
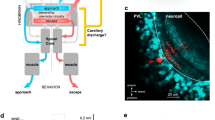
A basic question in the field of motor control is how different actions are represented by activity in spinal projection neurons. We used a new behavioral assay to identify visual stimuli that specifically drive basic motor patterns in zebrafish. These stimuli evoked consistent patterns of neural activity in the neurons projecting to the spinal cord, which we could map throughout the entire population using in vivo two-photon calcium imaging. We found that stimuli that drive distinct behaviors activated distinct subsets of projection neurons, consisting, in some cases, of just a few cells. This stands in contrast to the distributed activation seen for more complex behaviors. Furthermore, targeted cell by cell ablations of the neurons associated with evoked turns abolished the corresponding behavioral response. This description of the functional organization of the zebrafish motor system provides a framework for identifying the complete circuit underlying a vertebrate behavior.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Orlovsky, G.N., Deliagina, T.G. & Grillner, S. Neuronal Control of Locomotion (Oxford University Press, New York, 1999).
Rossignol, S., Dubuc, R.J. & Gossard, J.P. Dynamic sensorimotor interactions in locomotion. Physiol. Rev. 86, 89–154 (2006).
Zelenin, P.V. Activity of individual reticulospinal neurons during different forms of locomotion in the lamprey. Eur. J. Neurosci. 22, 2271–2282 (2005).
Deliagina, T.G., Zelenin, P.V. & Orlovsky, G.N. Encoding and decoding of reticulospinal commands. Brain Res. Brain Res. Rev. 40, 166–177 (2002).
Korn, H. & Faber, D.S. The Mauthner cell half a century later: a neurobiological model for decision-making? Neuron 47, 13–28 (2005).
Gahtan, E. & Baier, H. Of lasers, mutants and see-through brains: functional neuroanatomy in zebrafish. J. Neurobiol. 59, 147–161 (2004).
Saint-Amant, L. & Drapeau, P. Time course of the development of motor behaviors in the zebrafish embryo. J. Neurobiol. 37, 622–632 (1998).
Budick, S.A. & O'Malley, D.M. Locomotor repertoire of the larval zebrafish: swimming, turning and prey capture. J. Exp. Biol. 203, 2565–2579 (2000).
Burgess, H.A. & Granato, M. Modulation of locomotor activity in larval zebrafish during light adaptation. J. Exp. Biol. 210, 2526–2539 (2007).
Kimmel, C.B., Powell, S.L. & Metcalfe, W.K. Brain neurons which project to the spinal cord in young larvae of the zebrafish. J. Comp. Neurol. 205, 112–127 (1982).
Lee, R.K. & Eaton, R.C. Identifiable reticulospinal neurons of the adult zebrafish, Brachydanio rerio. J. Comp. Neurol. 304, 34–52 (1991).
O'Malley, D.M., Kao, Y.H. & Fetcho, J.R. Imaging the functional organization of zebrafish hindbrain segments during escape behaviors. Neuron 17, 1145–1155 (1996).
Metcalfe, W.K., Mendelson, B. & Kimmel, C.B. Segmental homologies among reticulospinal neurons in the hindbrain of the zebrafish larva. J. Comp. Neurol. 251, 147–159 (1986).
Gahtan, E. & O'Malley, D.M. Visually guided injection of identified reticulospinal neurons in zebrafish: a survey of spinal arborization patterns. J. Comp. Neurol. 459, 186–200 (2003).
Nissanov, J., Eaton, R.C. & DiDomenico, R. The motor output of the Mauthner cell, a reticulospinal command neuron. Brain Res. 517, 88–98 (1990).
Liu, K.S. & Fetcho, J.R. Laser ablations reveal functional relationships of segmental hindbrain neurons in zebrafish. Neuron 23, 325–335 (1999).
Burgess, H.A. & Granato, M. Sensorimotor gating in larval zebrafish. J. Neurosci. 27, 4984–4994 (2007).
Gahtan, E., Sankrithi, N., Campos, J.B. & O'Malley, D.M. Evidence for a widespread brain stem escape network in larval zebrafish. J. Neurophysiol. 87, 608–614 (2002).
Briggman, K.L., Abarbanel, H.D. & Kristan, W.B., Jr. Optical imaging of neuronal populations during decision-making. Science 307, 896–901 (2005).
Wu, J.Y., Cohen, L.B. & Falk, C.X. Neuronal activity during different behaviors in Aplysia: a distributed organization? Science 263, 820–823 (1994).
Bosch, T.J., Maslam, S. & Roberts, B.L. Fos-like immunohistochemical identification of neurons active during the startle response of the rainbow trout. J. Comp Neurol. 439, 306–314 (2001).
Zelenin, P.V., Orlovsky, G.N. & Deliagina, T.G. Sensory-motor transformation by individual command neurons. J. Neurosci. 27, 1024–1032 (2007).
Wiersma, C.A. & Ikeda, K. Interneurons commanding swimmeret movements in the crayfish, Procambarus clarkii (Girard). Comp. Biochem. Physiol. 12, 509–525 (1964).
Pearson, K.G. Common principles of motor control in vertebrates and invertebrates. Annu. Rev. Neurosci. 16, 265–297 (1993).
Orger, M.B., Smear, M.C., Anstis, S.M. & Baier, H. Perception of fourier and non-fourier motion by larval zebrafish. Nat. Neurosci. 3, 1128–1133 (2000).
Borla, M.A., Palecek, B., Budick, S. & O'Malley, D.M. Prey capture by larval zebrafish: evidence for fine axial motor control. Brain Behav. Evol. 60, 207–229 (2002).
Euler, T., Detwiler, P.B. & Denk, W. Directionally selective calcium signals in dendrites of starburst amacrine cells. Nature 418, 845–852 (2002).
Chung, S.H., Clark, D.A., Gabel, C.V., Mazur, E. & Samuel, A.D. The role of the AFD neuron in C. elegans thermotaxis analyzed using femtosecond laser ablation. BMC Neurosci. 7, 30 (2006).
Vogel, A. & Venugopalan, V. Mechanisms of pulsed laser ablation of biological tissues. Chem. Rev. 103, 577–644 (2003).
Leonardo, A. & Fee, M.S. Ensemble coding of vocal control in birdsong. J. Neurosci. 25, 652–661 (2005).
d'Avella, A., Saltiel, P. & Bizzi, E. Combinations of muscle synergies in the construction of a natural motor behavior. Nat. Neurosci. 6, 300–308 (2003).
Bizzi, E., d'Avella, A., Saltiel, P. & Tresch, M. Modular organization of spinal motor systems. Neuroscientist 8, 437–442 (2002).
Gahtan, E. & O'Malley, D.M. Rapid lesioning of large numbers of identified vertebrate neurons: applications in zebrafish. J. Neurosci. Methods 108, 97–110 (2001).
Zottoli, S.J., Newman, B.C., Rieff, H.I. & Winters, D.C. Decrease in occurrence of fast startle responses after selective Mauthner cell ablation in goldfish (Carassius auratus). J. Comp. Physiol. [A] 184, 207–218 (1999).
Gahtan, E., Tanger, P. & Baier, H. Visual prey capture in larval zebrafish is controlled by identified reticulospinal neurons downstream of the tectum. J. Neurosci. 25, 9294–9303 (2005).
Mendelson, B. Development of reticulospinal neurons of the zebrafish. II. Early axonal outgrowth and cell body position. J. Comp. Neurol. 251, 172–184 (1986).
Kimura, Y., Okamura, Y. & Higashijima, S. alx, a zebrafish homolog of Chx10, marks ipsilateral descending excitatory interneurons that participate in the regulation of spinal locomotor circuits. J. Neurosci. 26, 5684–5697 (2006).
Bhatt, D.H., McLean, D.L., Hale, M.E. & Fetcho, J.R. Grading movement strength by changes in firing intensity versus recruitment of spinal interneurons. Neuron 53, 91–102 (2007).
McLean, D.L., Fan, J., Higashijima, S., Hale, M.E. & Fetcho, J.R. A topographic map of recruitment in spinal cord. Nature 446, 71–75 (2007).
Chong, M. & Drapeau, P. Interaction between hindbrain and spinal networks during the development of locomotion in zebrafish. Dev. Neurobiol. 67, 933–947 (2007).
Niell, C.M. & Smith, S.J. Functional imaging reveals rapid development of visual response properties in the zebrafish tectum. Neuron 45, 941–951 (2005).
Sato, T., Hamaoka, T., Aizawa, H., Hosoya, T. & Okamoto, H. Genetic single-cell mosaic analysis implicates ephrinB2 reverse signaling in projections from the posterior tectum to the hindbrain in zebrafish. J. Neurosci. 27, 5271–5279 (2007).
Higashijima, S., Masino, M.A., Mandel, G. & Fetcho, J.R. Imaging neuronal activity during zebrafish behavior with a genetically encoded calcium indicator. J. Neurophysiol. 90, 3986–3997 (2003).
Lister, J.A., Robertson, C.P., Lepage, T., Johnson, S.L. & Raible, D.W. nacre encodes a zebrafish microphthalmia-related protein that regulates neural crest–derived pigment cell fate. Development 126, 3757–3767 (1999).
Denk, W., Strickler, J.H. & Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990).
Acknowledgements
The authors would like to thank M. Meister, T. Bonhoeffer, J.R. Sanes, B.P. Olveczky, M.C. Smear and J.E. Dowling for valuable comments on the manuscript, D.M. O'Malley for advice on experimental techniques, A.F. Schier for generous help with fish rearing and O.C. Orger for assistance with data analysis. This work was supported by postdoctoral fellowships from the Helen Hay Whitney Foundation (M.B.O.) and Human Frontier Science Program (J.H.B.), and US National Institutes of Health grant R01 EY014429-01A2 and funding from the McKnight and Dana Foundations (F.E.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 243 kb)
Supplementary Movie 1
Phenotype of Mauthner array ablation. The left Mauthner cell and its segmental homologs have been ablated in a 6-d-old zebrafish. In response to a pressure pulse delivered by a picospritzer to the right of the body, the fish responds with a leftward tail bend with a latency of 8 ms. In contrast, the rightward bend elicited by the same stimulus delivered to the left side of the body has a latency of 35 ms. Flashed rectangle indicates the onset of the pulse. (AVI 4691 kb)
Rights and permissions
About this article
Cite this article
Orger, M., Kampff, A., Severi, K. et al. Control of visually guided behavior by distinct populations of spinal projection neurons. Nat Neurosci 11, 327–333 (2008). https://doi.org/10.1038/nn2048
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn2048
This article is cited by
-
Functional neuronal circuits emerge in the absence of developmental activity
Nature Communications (2024)
-
Biomechanics and neural circuits for vestibular-induced fine postural control in larval zebrafish
Nature Communications (2023)
-
The mesencephalic locomotor region recruits V2a reticulospinal neurons to drive forward locomotion in larval zebrafish
Nature Neuroscience (2023)
-
Cognitive behavioral therapy (CBT), acceptance and commitment therapy (ACT), and Morita therapy (MT); comparison of three established psychotherapies and possible common neural mechanisms of psychotherapies
Journal of Neural Transmission (2022)
-
In vivo imaging reveals mature Oligodendrocyte division in adult Zebrafish
Cell Regeneration (2021)