Abstract
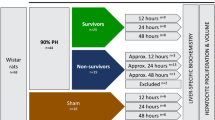
The ability of hepatocytes to enter the cell cycle and regenerate the liver after tissue loss provides an in vivo model to study the regulation of proliferation and organ regeneration. The extent of hepatocyte proliferation is directly proportional to the amount of resected liver tissue, and 2/3 partial hepatectomy (2/3 PH) leads to highly synchronized hepatocyte cell-cycle entry and progression. This surgical technique was first described in rats and requires modification for application in mice. Lack of standardization of 2/3 PH in mice has caused discrepancies in the results obtained in different laboratories. Here, we provide a protocol and a movie describing a straightforward surgical technique, which takes 15–20 min, to consistently remove two-thirds of the liver in mice. As this protocol is not associated with mortality and gives highly reproducible results, we hope that it will be widely used and serve to standardize 2/3 PH in mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
23 May 2014
Pain management during mouse experimentation has improved in the past several years, thereby requiring an update to this protocol. Updated information has been appended to the PDF version of this article.
References
Fausto, N., Campbell, J.S. & Riehle, K.J. Liver regeneration. Hepatology 43, S45–S53 (2006).
Higgins, G.M. & Anderson, R.M. Experimental pathology of the liver I. Restoration of the liver of the white rat following partial surgical removal. Arch. Pathol. 12, 186–202 (1931).
Wustefeld, T., Rakemann, T., Kubicka, S., Manns, M.P. & Trautwein, C. Hyperstimulation with interleukin 6 inhibits cell cycle progression after hepatectomy in mice. Hepatology 32, 514–522 (2000).
Borowiak, M. et al. Met provides essential signals for liver regeneration. Proc. Natl. Acad. Sci. USA 101, 10608–10613 (2004).
Greene, A.K. & Puder, M. Partial hepatectomy in the mouse: technique and perioperative management. J. Invest. Surg. 16, 99–102 (2003).
Martins, P.N., Theruvath, T.P. & Neuhaus, P. Rodent models of partial hepatectomies. Liver Int. 28, 3–11 (2008).
Bucher, N.L. Regeneration of mammalian liver. Int. Rev. Cytol. 15, 245–300 (1963).
Mitchell, C. et al. Heparin-binding epidermal growth factor-like growth factor links hepatocyte priming with cell cycle progression during liver regeneration. J. Biol. Chem. 280, 2562–2568 (2005).
Brooling, J.T., Campbell, J.S., Mitchell, C., Yeoh, G.C. & Fausto, N. Differential regulation of rodent hepatocyte and oval cell proliferation by interferon gamma. Hepatology 41, 906–915 (2005).
Latasa, M.U. et al. Delayed liver regeneration in mice lacking liver serum response factor. Am. J. Physiol. 292, G996–G1001 (2007).
Campbell, J.S. et al. Proinflammatory cytokine production in liver regeneration is Myd88-dependent, but independent of Cd14, Tlr2, and Tlr4. J. Immunol. 176, 2522–2528 (2006).
Skoulis, N.P., James, R.C., Harbison, R.D. & Roberts, S.M. Depression of hepatic glutathione by opioid analgesic drugs in mice. Toxicol. Appl. Pharmacol. 99, 139–147 (1989).
Berson, A. et al. Mechanisms for experimental buprenorphine hepatotoxicity: major role of mitochondrial dysfunction versus metabolic activation. J. Hepatol. 34, 261–269 (2001).
Matsuo, T. et al. Control mechanism of the circadian clock for timing of cell division in vivo. Science 302, 255–259 (2003).
Heijboer, A.C. et al. Sixteen hours of fasting differentially affects hepatic and muscle insulin sensitivity in mice. J. Lipid Res. 46, 582–588 (2005).
Selzner, M. & Clavien, P.A. Failure of regeneration of the steatotic rat liver: disruption at two different levels in the regeneration pathway. Hepatology 31, 35–42 (2000).
Yang, S.Q., Lin, H.Z., Mandal, A.K., Huang, J. & Diehl, A.M. Disrupted signaling and inhibited regeneration in obese mice with fatty livers: implications for nonalcoholic fatty liver disease pathophysiology. Hepatology 34, 694–706 (2001).
DeAngelis, R.A., Markiewski, M.M., Taub, R. & Lambris, J.D. A high-fat diet impairs liver regeneration in C57BL/6 mice through overexpression of the NF-kappaB inhibitor, IkappaBalpha. Hepatology 42, 1148–1157 (2005).
Shteyer, E., Liao, Y., Muglia, L.J., Hruz, P.W. & Rudnick, D.A. Disruption of hepatic adipogenesis is associated with impaired liver regeneration in mice. Hepatology 40, 1322–1332 (2004).
Naugler, W.E. et al. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science 317, 121–124 (2007).
Desbois-Mouthon, C. et al. Hepatocyte proliferation during liver regeneration is impaired in mice with liver-specific IGF-1R knockout. FASEB J. 20, 773–775 (2006).
Iakova, P., Awad, S.S. & Timchenko, N.A. Aging reduces proliferative capacities of liver by switching pathways of C/EBPalpha growth arrest. Cell 113, 495–506 (2003).
Leclercq, I.A. et al. Defective hepatic regeneration after partial hepatectomy in leptin-deficient mice is not rescued by exogenous leptin. Lab. Invest. 86, 1161–1171 (2006).
Alwayn, I.P. et al. A critical role for matrix metalloproteinases in liver regeneration. J. Surg. Res. 145, 192–198 (2008).
Acknowledgements
H.W. and C.M. thank B. Rain Bennett (Flying Flounder Productions) and Dr. Susan Sunnarborg for helping with the production of the video. C.M. thanks Dr. Jose Luiz de Godoy for introducing her to the art of surgery and Dr. Nelson Fausto for his mentorship in the field of liver regeneration. H.W. is a Liver Scholar of the American Liver Foundation/American Society of Transplantation.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Video 1
Step 3. Opening. (MOV 2853 kb)
Supplementary Video 2
Step 4. Liver exposure with rubber band. (MOV 1013 kb)
Supplementary Video 3
Step 5. Retractors. (MOV 1738 kb)
Supplementary Video 4
Step 6. Cutting the falciform ligament. (MOV 2023 kb)
Supplementary Video 5
Step 7. Cutting membrane between caudate and left lateral lobes. (MOV 3463 kb)
Supplementary Video 6
Step 8. Placing 1st knot. (MOV 7625 kb)
Supplementary Video 7
Step 9. Tying 1st knot. (MOV 6002 kb)
Supplementary Video 8
Step 10. Resection of 1st lobe. (MOV 10964 kb)
Supplementary Video 9
Step 11. Placing 2nd knot. (MOV 9684 kb)
Supplementary Video 10
Step 12. Tying and resecting 2nd lobe. (MOV 23422 kb)
Supplementary Video 11
Step 13. Closure. (MOV 10275 kb)
Rights and permissions
About this article
Cite this article
Mitchell, C., Willenbring, H. A reproducible and well-tolerated method for 2/3 partial hepatectomy in mice. Nat Protoc 3, 1167–1170 (2008). https://doi.org/10.1038/nprot.2008.80
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.80
This article is cited by
-
Identification of myeloid-derived growth factor as a mechanically-induced, growth-promoting angiocrine signal for human hepatocytes
Nature Communications (2024)
-
Transcriptome sequencing and metabolome analysis reveal the metabolic reprogramming of partial hepatectomy and extended hepatectomy
BMC Genomics (2023)
-
A highly sensitive strategy for monitoring real-time proliferation of targeted cell types in vivo
Nature Communications (2023)
-
Mfap4: a promising target for enhanced liver regeneration and chronic liver disease treatment
npj Regenerative Medicine (2023)
-
tRF-Gln-CTG-026 ameliorates liver injury by alleviating global protein synthesis
Signal Transduction and Targeted Therapy (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.