Abstract
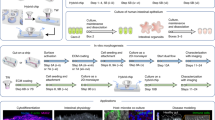
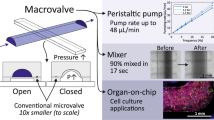
'Organs-on-chips' are microengineered biomimetic systems containing microfluidic channels lined by living human cells, which replicate key functional units of living organs to reconstitute integrated human organ-level pathophysiology in vitro. These microdevices can be used to test efficacy and toxicity of drugs and chemicals, and to create in vitro models of human disease. Thus, they potentially represent low-cost alternatives to conventional animal models for pharmaceutical, chemical and environmental applications. Here we describe a protocol for the fabrication, microengineering and operation of these microfluidic organ-on-chip systems. First, microengineering is used to fabricate a multilayered microfluidic device that contains two parallel elastomeric microchannels separated by a thin porous flexible membrane, along with two full-height, hollow vacuum chambers on either side; this requires ∼3.5 d to complete. To create a 'breathing' lung-on-a-chip that mimics the mechanically active alveolar-capillary interface of the living human lung, human alveolar epithelial cells and microvascular endothelial cells are cultured in the microdevice with physiological flow and cyclic suction applied to the side chambers to reproduce rhythmic breathing movements. We describe how this protocol can be easily adapted to develop other human organ chips, such as a gut-on-a-chip lined by human intestinal epithelial cells that experiences peristalsis-like motions and trickling fluid flow. Also, we discuss experimental techniques that can be used to analyze the cells in these organ-on-chip devices.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Huh, D., Hamilton, G.A. & Ingber, D.E. From 3D cell culture to organs-on-chips. Trends Cell Biol. 21, 745–754 (2011).
Jang, K., Sato, K., Igawa, K., Chung, U.I. & Kitamori, T. Development of an osteoblast-based 3D continuous-perfusion microfluidic system for drug screening. Anal. Bioanal. Chem. 390, 825–832 (2008).
Leclerc, E. et al. Study of osteoblastic cells in a microfluidic environment. Biomaterials 27, 586–595 (2006).
O'Neill, A.T., Monteiro-Riviere, N.A. & Walker, G.M. Characterization of microfluidic human epidermal keratinocyte culture. Cytotechnology 56, 197–207 (2008).
Chao, P.G. et al. Dynamic osmotic loading of chondrocytes using a novel microfluidic device. J. Biomech 38, 1273–1281 (2005).
Baudoin, R., Griscom, L., Monge, M., Legallais, C. & Leclerc, E. Development of a renal microchip for in vitro distal tubule models. Biotechnol. Prog. 23, 1245–1253 (2007).
Leclerc, E., Sakai, Y. & Fujii, T. Microfluidic PDMS (polydimethylsiloxane) bioreactor for large-scale culture of hepatocytes. Biotechnol. Prog. 20, 750–755 (2004).
Powers, M.J. et al. A microfabricated array bioreactor for perfused 3D liver culture. Biotechnol. Bioeng. 78, 257–269 (2002).
Tilles, A.W., Baskaran, H., Roy, P., Yarmush, M.L. & Toner, M. Effects of oxygenation and flow on the viability and function of rat hepatocytes cocultured in a microchannel flat-plate bioreactor. Biotechnol. Bioeng. 73, 379–389 (2001).
Shin, M. et al. Endothelialized networks with a vascular geometry in microfabricated poly(dimethyl siloxane). Biomed. Microdevices 6, 269–278 (2004).
Huh, D., Torisawa, Y.S., Hamilton, G.A., Kim, H.J. & Ingber, D.E. Microengineered physiological biomimicry: organs-on-chips. Lab Chip 12, 2156–2164 (2012).
van der Meer, A.D. & van den Berg, A. Organs-on-chips: breaking the in vitro impasse. Integr. Biol. 4, 461–470 (2012).
Sung, J.H. et al. Microfabricated mammalian organ systems and their integration into models of whole animals and humans. Lab Chip 13, 1201–1212 (2013).
Moraes, C., Mehta, G., Lesher-Perez, S.C. & Takayama, S. Organs-on-a-chip: a focus on compartmentalized microdevices. Ann. Biomed. Eng. 40, 1211–1227 (2012).
Ghaemmaghami, A.M., Hancock, M.J., Harrington, H., Kaji, H. & Khademhosseini, A. Biomimetic tissues on a chip for drug discovery. Drug Discov. Today 17, 173–181 (2012).
Esch, M.B., King, T.L. & Shuler, M.L. The role of body-on-a-chip devices in drug and toxicity studies. Annu. Rev. Biomed. Eng. 13, 55–72 (2011).
Jang, K.J. et al. Fluid-shear-stress-induced translocation of aquaporin-2 and reorganization of actin cytoskeleton in renal tubular epithelial cells. Integr. Biol. 3, 134–141 (2011).
Jang, K.J. & Suh, K.Y. A multi-layer microfluidic device for efficient culture and analysis of renal tubular cells. Lab Chip 10, 36–42 (2010).
Jang, K.J. et al. Human kidney proximal tubule-on-a-chip for drug transport and nephrotoxicity assessment. Integr. Biol. 5, 1119–1129 (2013)
Carraro, A. et al. In vitro analysis of a hepatic device with intrinsic microvascular-based channels. Biomed. Microdevices 10, 795–805 (2008).
Kane, B.J., Zinner, M.J., Yarmush, M.L. & Toner, M. Liver-specific functional studies in a microfluidic array of primary mammalian hepatocytes. Anal. Chem. 78, 4291–4298 (2006).
Khetani, S.R. & Bhatia, S.N. Microscale culture of human liver cells for drug development. Nat. Biotechnol. 26, 120–126 (2008).
Lee, P.J., Hung, P.J. & Lee, L.P. An artificial liver sinusoid with a microfluidic endothelial-like barrier for primary hepatocyte culture. Biotechnol. Bioeng. 97, 1340–1346 (2007).
Nakao, Y., Kimura, H., Sakai, Y. & Fujii, T. Bile canaliculi formation by aligning rat primary hepatocytes in a microfluidic device. Biomicrofluidics 5, 22212 (2011).
Griep, L.M. et al. BBB ON CHIP: microfluidic platform to mechanically and biochemically modulate blood-brain barrier function. Biomed. Microdevices 15, 145–150 (2013).
Khademhosseini, A. et al. Microfluidic patterning for fabrication of contractile cardiac organoids. Biomed. Microdevices 9, 149–157 (2007).
Grosberg, A., Alford, P.W., McCain, M.L. & Parker, K.K. Ensembles of engineered cardiac tissues for physiological and pharmacological study: heart on a chip. Lab Chip 11, 4165–4173 (2011).
Feinberg, A.W. et al. Muscular thin films for building actuators and powering devices. Science 317, 1366–1370 (2007).
Nawroth, J.C. et al. A tissue-engineered jellyfish with biomimetic propulsion. Nat. Biotechnol. 30, 792–797 (2012).
McCain, M.L., Sheehy, S.P., Grosberg, A., Goss, J.A. & Parker, K.K. Recapitulating maladaptive, multiscale remodeling of failing myocardium on a chip. Proc. Natl. Acad. Sci. USA 110, 9770–9775 (2013).
Giridharan, G.A. et al. Microfluidic cardiac cell culture model (muCCCM). Anal. Chem. 82, 7581–7587 (2010).
Wilson, K., Das, M., Wahl, K.J., Colton, R.J. & Hickman, J. Measurement of contractile stress generated by cultured rat muscle on silicon cantilevers for toxin detection and muscle performance enhancement. PLoS ONE 5, e11042 (2010).
Esch, M.B. et al. On chip porous polymer membranes for integration of gastrointestinal tract epithelium with microfluidic 'body-on-a-chip' devices. Biomed. Microdevices 14, 895–906 (2012).
Imura, Y., Asano, Y., Sato, K. & Yoshimura, E. A microfluidic system to evaluate intestinal absorption. Anal. Sci. 25, 1403–1407 (2009).
Kimura, H., Yamamoto, T., Sakai, H., Sakai, Y. & Fujii, T. An integrated microfluidic system for long-term perfusion culture and on-line monitoring of intestinal tissue models. Lab Chip 8, 741–746 (2008).
Mahler, G.J., Esch, M.B., Glahn, R.P. & Shuler, M.L. Characterization of a gastrointestinal tract microscale cell culture analog used to predict drug toxicity. Biotechnol. Bioeng. 104, 193–205 (2009).
Sung, J.H., Yu, J., Luo, D., Shuler, M.L. & March, J.C. Microscale 3-D hydrogel scaffold for biomimetic gastrointestinal (GI) tract model. Lab Chip 11, 389–392 (2011).
Wang, L., Murthy, S.K., Barabino, G.A. & Carrier, R.L. Synergic effects of crypt-like topography and ECM proteins on intestinal cell behavior in collagen based membranes. Biomaterials 31, 7586–7598 (2010).
Puleo, C.M., McIntosh Ambrose, W., Takezawa, T., Elisseeff, J. & Wang, T.H. Integration and application of vitrified collagen in multilayered microfluidic devices for corneal microtissue culture. Lab Chip 9, 3221–3227 (2009).
Chung, S. et al. Cell migration into scaffolds under co-culture conditions in a microfluidic platform. Lab Chip 9, 269–275 (2009).
Shin, Y. et al. Microfluidic assay for simultaneous culture of multiple cell types on surfaces or within hydrogels. Nat. Protoc. 7, 1247–1259 (2012).
Sung, K.E. et al. Control of 3-dimensional collagen matrix polymerization for reproducible human mammary fibroblast cell culture in microfluidic devices. Biomaterials 30, 4833–4841 (2009).
Sung, K.E. et al. Transition to invasion in breast cancer: a microfluidic in vitro model enables examination of spatial and temporal effects. Integr. Biol. 3, 439–450 (2011).
Zervantonakis, I.K., Kothapalli, C.R., Chung, S., Sudo, R. & Kamm, R.D. Microfluidic devices for studying heterotypic cell-cell interactions and tissue specimen cultures under controlled microenvironments. Biomicrofluidics 5, 13406 (2011).
Achyuta, A.K. et al. A modular approach to create a neurovascular unit-on-a-chip. Lab Chip 13, 542–553 (2013).
Park, J., Koito, H., Li, J. & Han, A. Multi-compartment neuron-glia co-culture platform for localized CNS axon-glia interaction study. Lab Chip 12, 3296–3304 (2012).
Prabhakarpandian, B. et al. SyM-BBB: a microfluidic Blood Brain Barrier model. Lab Chip 13, 1093–1101 (2013).
Ma, S.H., Lepak, L.A., Hussain, R.J., Shain, W. & Shuler, M.L. An endothelial and astrocyte co-culture model of the blood-brain barrier utilizing an ultra-thin, nanofabricated silicon nitride membrane. Lab Chip 5, 74–85 (2005).
Allen, J.W. & Bhatia, S.N. Formation of steady-state oxygen gradients in vitro: application to liver zonation. Biotechnol. Bioeng. 82, 253–262 (2003).
Kim, S.H., Kang, J.H., Chung, I.Y. & Chung, B.G. Mucin (MUC5AC) expression by lung epithelial cells cultured in a microfluidic gradient device. Electrophoresis 32, 254–260 (2011).
Torisawa, Y.S. et al. Microfluidic platform for chemotaxis in gradients formed by CXCL12 source-sink cells. Integr. Biol. 2, 680–686 (2010).
Douville, N.J. et al. Combination of fluid and solid mechanical stresses contribute to cell death and detachment in a microfluidic alveolar model. Lab Chip 11, 609–619 (2011).
Huh, D. et al. Acoustically detectable cellular-level lung injury induced by fluid mechanical stresses in microfluidic airway systems. Proc. Natl. Acad. Sci. USA 104, 18886–18891 (2007).
Huh, D. et al. Reconstituting organ-level lung functions on a chip. Science 328, 1662–1668 (2010).
Huh, D. et al. A human disease model of drug toxicity-induced pulmonary edema in a lung-on-a-chip microdevice. Sci. Transl. Med. 4, 159ra147 (2012).
Kim, H.J., Huh, D., Hamilton, G. & Ingber, D.E. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab Chip 12, 2165–2174 (2012).
Kim, H.J. & Ingber, D.E. Gut-on-a-Chip microenvironment induces human intestinal cells to undergo villus differentiation. Integr. Biol. 5, 1130–1140 (2013).
Ochs, M. et al. The number of alveoli in the human lung. Am. J. Respir. Crit. Care Med. 169, 120–124 (2004).
Toepke, M.W. & Beebe, D.J. PDMS absorption of small molecules and consequences in microfluidic applications. Lab Chip 6, 1484–1486 (2006).
Wang, J.D., Douville, N.J., Takayama, S. & ElSayed, M. Quantitative analysis of molecular absorption into PDMS microfluidic channels. Ann. Biomed. Eng. 40, 1862–1873 (2012).
Gomez-Sjoberg, R., Leyrat, A.A., Houseman, B.T., Shokat, K. & Quake, S.R. Biocompatibility and reduced drug absorption of sol-gel-treated poly(dimethyl siloxane) for microfluidic cell culture applications. Anal. Chem. 82, 8954–8960 (2010).
Borysiak, M.D. et al. Simple replica micromolding of biocompatible styrenic elastomers. Lab Chip 13, 2773–2784 (2013).
Domansky, K. et al. Clear castable polyurethane elastomer for fabrication of microfluidic devices. Lab Chip 13, 3956–3964 (2013).
Acknowledgements
We thank D. Levner and C. Hinojosa for their assistance in preparing the protocols for device fabrication. This work was supported by the Wyss Institute for Biologically Inspired Engineering at Harvard University and grants from US National Institutes of Health (NIH) NIEHS (ES016665-01A1) and the NIH Common Fund (U01 NS073474) through the Division of Program Coordination, Planning, and Strategic Initiatives (DPCPSI), Office of the Director, NIH and the US Food and Drug Administration (FDA). Additional funds were provided by the Defense Advanced Research Projects Agency (DARPA) under Cooperative Agreement Number W911NF-12-2-0036, and FDA contract HHSF223201310079C. The content of the information does not necessarily reflect the position or the policy of DARPA or the US Government, and no official endorsement should be inferred.
Author information
Authors and Affiliations
Contributions
D.H. led development of the lung-on-a-chip, performed experiments, analyzed data and prepared the manuscript. H.J.K. led development of the gut-on-a-chip, performed experiments, analyzed data and contributed to preparation of the manuscript. J.P.F., D.E.S., M.K., A.B. and G.A.H. provided assistance in experiments, data analysis and manuscript preparation. D.E.I. led the organ-on-chip effort, assisted in experimental design and analysis and helped in writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Huh, D., Kim, H., Fraser, J. et al. Microfabrication of human organs-on-chips. Nat Protoc 8, 2135–2157 (2013). https://doi.org/10.1038/nprot.2013.137
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.137
This article is cited by
-
Engineered platforms for mimicking cardiac development and drug screening
Cellular and Molecular Life Sciences (2024)
-
Polymeric and biological membranes for organ-on-a-chip devices
Microsystems & Nanoengineering (2023)
-
Enabling direct microcalorimetric measurement of metabolic activity and exothermic reactions onto microfluidic platforms via heat flux sensor integration
Microsystems & Nanoengineering (2023)
-
The feasible application of microfluidic tissue/organ-on-a-chip as an impersonator of oral tissues and organs: a direction for future research
Bio-Design and Manufacturing (2023)
-
Recent developments in organ-on-a-chip technology for cardiovascular disease research
Analytical and Bioanalytical Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.