Key Points
-
Primary liver cancers (PLCs), including the most common subtypes, hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (iCCA), are characterized by molecular and phenotypic heterogeneity, which impedes therapeutic progress.
-
Chronic inflammation of the hepatic microenvironment is a hallmark feature of the majority of PLCs.
-
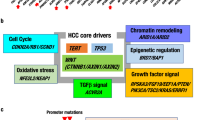
Although no clear oncogene addiction is recognized in PLCs, most common genetic alterations are detected in key cancer genes, such as TP53, WNT–CTNNB1 (which encodes β-catenin) and cell cycle-related genes. Other frequent changes are found in telomere maintenance (telomerase reverse transcriptase (TERT)), chromatin modifiers and inflammatory pathways.
-
Depending on the target cell of malignant transformation — that is, the cell of origin — a broad range of different phenotypes, from classic HCC and iCCA to mixed HCC–iCCA lesions, is observed.
-
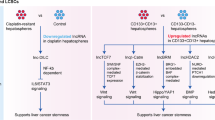
The putative cellular origin in PLCs is diverse and not clearly defined. The differentiation state of the cell (or cells) of origin might determine the tumour biology.
-
Tumours that have progenitor cell traits display activation of adverse signalling pathways and a poor overall outcome.
-
Crosstalk between cancer cells and the altered hepatic tumour microenvironment can promote disease progression and might be a promising new therapeutic target.
-
The identification of novel cellular and molecular targets requires integrative approaches. Precision medicine using next-generation technologies (whole-exome and RNA sequencing, and methylation profiling) and integration of individual tumour characteristics will be needed to improve the dismal outcome for patients with PLC.
Abstract
During the past decade, research on primary liver cancers has particularly highlighted the uncommon plasticity of differentiated parenchymal liver cells (that is, hepatocytes and cholangiocytes (also known as biliary epithelial cells)), the role of liver progenitor cells in malignant transformation, the importance of the tumour microenvironment and the molecular complexity of liver tumours. Whereas other reviews have focused on the landscape of genetic alterations that promote development and progression of primary liver cancers and the role of the tumour microenvironment, the crucial importance of the cellular origin of liver cancer has been much less explored. Therefore, in this Review, we emphasize the importance and complexity of the cellular origin in tumour initiation and progression, and attempt to integrate this aspect with recent discoveries in tumour genomics and the contribution of the disrupted hepatic microenvironment to liver carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lozano, R. et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2095–2128 (2012).
El-Serag, H. B. Hepatocellular carcinoma. N. Engl. J. Med. 365, 1118–1127 (2011).
Pawlotsky, J. M., Feld, J. J., Zeuzem, S. & Hoofnagle, J. H. From non-A, non-B hepatitis to hepatitis C virus cure. J. Hepatol. 62, S87–S99 (2015).
Marquardt, J. U., Galle, P. R. & Teufel, A. Molecular diagnosis and therapy of hepatocellular carcinoma (HCC): an emerging field for advanced technologies. J. Hepatol. 56, 267–275 (2012).
Razumilava, N. & Gores, G. J. Classification, diagnosis, and management of cholangiocarcinoma. Clin. Gastroenterol. Hepatol. 11, 13–21 (2013).
Bridgewater, J. et al. Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J. Hepatol. 60, 1268–1289 (2014). These are the first clinical practice guidelines for iCCA, summarizing epidemiological data and the current state of the art in diagnosis and therapy.
The International Consensus Group for Hepatocellular Neoplasia. Pathologic diagnosis of early hepatocellular carcinoma: a report of the international consensus group for hepatocellular neoplasia. Hepatology 49, 658–664 (2009); erratum 49, 1058 (2009)
Farazi, P. A. & DePinho, R. A. Hepatocellular carcinoma pathogenesis: from genes to environment. Nat. Rev. Cancer 6, 674–687 (2006). This is a landmark review summarizing the molecular pathomechansims of HCC.
Marquardt, J. U. et al. Sequential transcriptome analysis of human liver cancer indicates late stage acquisition of malignant traits. J. Hepatol. 60, 346–353 (2014).
Guichard, C. et al. Integrated analysis of somatic mutations and focal copy-number changes identifies key genes and pathways in hepatocellular carcinoma. Nat. Genet. 44, 694–698 (2012).
Kan, Z. et al. Whole-genome sequencing identifies recurrent mutations in hepatocellular carcinoma. Genome Res. 23, 1422–1433 (2013).
Nault, J. C. et al. High frequency of telomerase reverse-transcriptase promoter somatic mutations in hepatocellular carcinoma and preneoplastic lesions. Nat. Commun. 4, 2218 (2013).
Raggi, C., Invernizzi, P. & Andersen, J. B. Impact of microenvironment and stem-like plasticity in cholangiocarcinoma: molecular networks and biological concepts. J. Hepatol. 62, 198–207 (2015).
Li, M. et al. Inactivating mutations of the chromatin remodeling gene ARID2 in hepatocellular carcinoma. Nat. Genet. 43, 828–829 (2011).
Totoki, Y. et al. High-resolution characterization of a hepatocellular carcinoma genome. Nat. Genet. 43, 464–469 (2011). This is the first landmark study to use next-generation sequencing in HCC on 12 individual Japanese tumours.
Fujimoto, A. et al. Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat. Genet. 44, 760–764 (2012).
Andersen, J. B. et al. Genomic and genetic characterization of cholangiocarcinoma identifies therapeutic targets for tyrosine kinase inhibitors. Gastroenterology 142, 1021–1031.e15 (2012). This is a seminal study on the CCA transcriptome. The study revealed different prognostic subtypes in CCA with distinct genetic alterations that could be used for therapeutic interventions.
Oishi, N. et al. Transcriptomic profiling reveals hepatic stem-like gene signatures and interplay of miR-200c and epithelial-mesenchymal transition in intrahepatic cholangiocarcinoma. Hepatology 56, 1792–1803 (2012).
Sia, D. et al. Integrative molecular analysis of intrahepatic cholangiocarcinoma reveals 2 classes that have different outcomes. Gastroenterology 144, 829–840 (2013).
Seok, J. Y. et al. A fibrous stromal component in hepatocellular carcinoma reveals a cholangiocarcinoma-like gene expression trait and epithelial-mesenchymal transition. Hepatology 55, 1776–1786 (2012).
Woo, H. G. et al. Identification of a cholangiocarcinoma-like gene expression trait in hepatocellular carcinoma. Cancer Res. 70, 3034–3041 (2010).
Cazals-Hatem, D. et al. Clinical and molecular analysis of combined hepatocellular-cholangiocarcinomas. J. Hepatol. 41, 292–298 (2004).
Fujii, H. et al. Genetic classification of combined hepatocellular-cholangiocarcinoma. Hum. Pathol. 31, 1011–1017 (2000).
Schulze, K. et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat. Genet. 47, 505–511 (2015).
Jiang, J. H. et al. Clinical significance of the ubiquitin ligase UBE3C in hepatocellular carcinoma revealed by exome sequencing. Hepatology 59, 2216–2227 (2014).
Kaposi-Novak, P. et al. Central role of c-Myc during malignant conversion in human hepatocarcinogenesis. Cancer Res. 69, 2775–2782 (2009).
Holczbauer, A. et al. Modeling pathogenesis of primary liver cancer in lineage-specific mouse cell types. Gastroenterology 145, 221–231 (2013). This study demonstrates that induction of malignant transformation by selected oncogenes can induce liver cancer development regardless of the putative cell of origin in the hepatic lineage.
Nault, J. C. et al. Telomerase reverse transcriptase promoter mutation is an early somatic genetic alteration in the transformation of premalignant nodules in hepatocellular carcinoma on cirrhosis. Hepatology 60, 1983–1992 (2014).
Tovar, V. et al. IGF activation in a molecular subclass of hepatocellular carcinoma and pre-clinical efficacy of IGF-1R blockage. J. Hepatol. 52, 550–559 (2010).
Woo, H. G. et al. Profiling of exome mutations associated with progression of HBV-related hepatocellular carcinoma. PLoS ONE 9, e115152 (2014).
Sung, W. K. et al. Genome-wide survey of recurrent HBV integration in hepatocellular carcinoma. Nat. Genet. 44, 765–769 (2012).
Alexandrov, L. B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Totoki, Y. et al. Trans-ancestry mutational landscape of hepatocellular carcinoma genomes. Nat. Genet. 46, 1267–1273 (2014). This is a large next-generation sequencing study on PLC performed on data from 488 HCCs from different populations. The study revealed 30 candidate driver genes and 11 core pathway modules.
Chen, Y., Wang, L., Xu, H., Liu, X. & Zhao, Y. Exome capture sequencing reveals new insights into hepatitis B virus-induced hepatocellular carcinoma at the early stage of tumorigenesis. Oncol. Rep. 30, 1906–1912 (2013).
Cleary, S. P. et al. Identification of driver genes in hepatocellular carcinoma by exome sequencing. Hepatology 58, 1693–1702 (2013).
Huang, J. et al. Exome sequencing of hepatitis B virus-associated hepatocellular carcinoma. Nat. Genet. 44, 1117–1121 (2012).
Liu, Y. X. et al. Whole-exome sequencing identifies mutated PCK2 and HUWE1 associated with carcinoma cell proliferation in a hepatocellular carcinoma patient. Oncol. Lett. 4, 847–851 (2012).
Chen, Y. L. et al. TERT promoter mutation in resectable hepatocellular carcinomas: a strong association with hepatitis C infection and absence of hepatitis B infection. Int. J. Surg. 12, 659–665 (2014).
Ding, D. et al. Recurrent targeted genes of hepatitis B virus in the liver cancer genomes identified by a next-generation sequencing-based approach. PLoS Genet. 8, e1003065 (2012).
Jhunjhunwala, S. et al. Diverse modes of genomic alteration in hepatocellular carcinoma. Genome Biol. 15, 436 (2014).
Shiraishi, Y. et al. Integrated analysis of whole genome and transcriptome sequencing reveals diverse transcriptomic aberrations driven by somatic genomic changes in liver cancers. PLoS ONE 9, e114263 (2014).
Zhong, R. et al. Genetic variant in SWI/SNF complexes influences hepatocellular carcinoma risk: a new clue for the contribution of chromatin remodeling in carcinogenesis. Sci. Rep. 4, 4147 (2014).
Chan, T. H. et al. A disrupted RNA editing balance mediated by ADARs (Adenosine DeAminases that act on RNA) in human hepatocellular carcinoma. Gut 63, 832–843 (2014).
Chen, L. et al. Recoding RNA editing of AZIN1 predisposes to hepatocellular carcinoma. Nat. Med. 19, 209–216 (2013).
Kang, L. et al. Genome-wide identification of RNA editing in hepatocellular carcinoma. Genomics 105, 76–82 (2015).
Andersen, J. B. Molecular pathogenesis of intrahepatic cholangiocarcinoma. J. Hepatobiliary Pancreat. Sci. 22, 101–113 (2015).
Wang, P. et al. Mutations in isocitrate dehydrogenase 1 and 2 occur frequently in intrahepatic cholangiocarcinomas and share hypermethylation targets with glioblastomas. Oncogene 32, 3091–3100 (2013).
Chan-On, W. et al. Exome sequencing identifies distinct mutational patterns in liver fluke-related and non-infection-related bile duct cancers. Nat. Genet. 45, 1474–1478 (2013).
Gao, Q. et al. Activating mutations in PTPN3 promote cholangiocarcinoma cell proliferation and migration and are associated with tumor recurrence in patients. Gastroenterology 146, 1397–1407 (2014).
Jiao, Y. et al. Exome sequencing identifies frequent inactivating mutations in BAP1, ARID1A and PBRM1 in intrahepatic cholangiocarcinomas. Nat. Genet. 45, 1470–1473 (2013).
Ong, C. K. et al. Exome sequencing of liver fluke-associated cholangiocarcinoma. Nat. Genet. 44, 690–693 (2012).
Sia, D. et al. Massive parallel sequencing uncovers actionable FGFR2–PPHLN1 fusion and ARAF mutations in intrahepatic cholangiocarcinoma. Nat. Commun. 6, 6087 (2015).
Zou, S. et al. Mutational landscape of intrahepatic cholangiocarcinoma. Nat. Commun. 5, 5696 (2014). This is currently the largest next-generation sequencing study on iCCA. The study identified mutational signatures associated with liver inflammation, fibrosis and cirrhosis. Twenty-five genes were substantially mutated, including eight potential driver genes.
Marquardt, J. U. & Andersen, J. B. Liver cancer oncogenomics: opportunities and dilemmas for clinical applications. Hepat. Oncol. 2, 79–93 (2015).
Muisuk, K. et al. Novel mutations in cholangiocarcinoma with low frequencies revealed by whole mitochondrial genome sequencing. Asian Pac. J. Cancer Prev. 16, 1737–1742 (2015).
Wu, Y. M. et al. Identification of targetable FGFR gene fusions in diverse cancers. Cancer Discov. 3, 636–647 (2013).
Ross, J. S. et al. New routes to targeted therapy of intrahepatic cholangiocarcinomas revealed by next-generation sequencing. Oncologist 19, 235–242 (2014).
Arai, Y. et al. Fibroblast growth factor receptor 2 tyrosine kinase fusions define a unique molecular subtype of cholangiocarcinoma. Hepatology 59, 1427–1434 (2014).
Borad, M. J. et al. Integrated genomic characterization reveals novel, therapeutically relevant drug targets in FGFR and EGFR pathways in sporadic intrahepatic cholangiocarcinoma. PLoS Genet. 10, e1004135 (2014).
Graham, R. P. et al. Fibroblast growth factor receptor 2 translocations in intrahepatic cholangiocarcinoma. Hum. Pathol. 45, 1630–1638 (2014).
Klein, W. M. et al. Primary liver carcinoma arising in people younger than 30 years. Am. J. Clin. Pathol. 124, 512–518 (2005).
Pilati, C. et al. Genomic profiling of hepatocellular adenomas reveals recurrent FRK-activating mutations and the mechanisms of malignant transformation. Cancer Cell 25, 428–441 (2014). This is the first study to perform large-scale analyses on HPAs. The study identified recurrent FRK -activating mutations and other genetic predictors of malignant transformation, including TERT alterations.
Nault, J. C., Bioulac-Sage, P. & Zucman-Rossi, J. Hepatocellular benign tumors — from molecular classification to personalized clinical care. Gastroenterology 144, 888–902 (2013).
Marquardt, J. U. & Thorgeirsson, S. S. Next-generation genomic profiling of hepatocellular adenomas: a new era of individualized patient care. Cancer Cell 25, 409–411 (2014).
Eichenmuller, M. et al. The genomic landscape of hepatoblastoma and their progenies with HCC-like features. J. Hepatol. 61, 1312–1320 (2014).
Jia, D. et al. Exome sequencing of hepatoblastoma reveals novel mutations and cancer genes in the Wnt pathway and ubiquitin ligase complex. Hepatology 60, 1686–1696 (2014). This is the first study to perform whole-exome sequencing of six paired hepatoblastoma tumours. It provides a detailed description of the landscape of genetic alterations in hepatoblastoma, highlighting the importance of WNT and ubiquitin pathways.
Eggert, T. et al. Fibrolamellar hepatocellular carcinoma in the USA, 2000–2010: a detailed report on frequency, treatment and outcome based on the Surveillance, Epidemiology, and End Results database. United European Gastroenterol. J. 1, 351–357 (2013).
Honeyman, J. N. et al. Detection of a recurrent DNAJB1–PRKACA chimeric transcript in fibrolamellar hepatocellular carcinoma. Science 343, 1010–1014 (2014).
Cornella, H. et al. Unique genomic profile of fibrolamellar hepatocellular carcinoma. Gastroenterology 148, 806–818 (2014). This study provides integrative analyses of 78 FLCs. The study identified three subtypes with distinct genetic alterations and survival rates.
Darcy, D. G. et al. The genomic landscape of fibrolamellar hepatocellular carcinoma: whole genome sequencing of ten patients. Oncotarget 6, 755–770 (2014).
Yamashita, T. & Wang, X. W. Cancer stem cells in the development of liver cancer. J. Clin. Invest. 123, 1911–1918 (2013).
Llovet, J. M. & Bruix, J. Molecular targeted therapies in hepatocellular carcinoma. Hepatology 48, 1312–1327 (2008).
Marquardt, J. U. & Thorgeirsson, S. S. Stem cells in hepatocarcinogenesis: evidence from genomic data. Semin. Liver Dis. 30, 26–34 (2010).
Duncan, A. W., Dorrell, C. & Grompe, M. Stem cells and liver regeneration. Gastroenterology 137, 466–481 (2009).
Overturf, K., al-Dhalimy, M., Ou, C. N., Finegold, M. & Grompe, M. Serial transplantation reveals the stem-cell-like regenerative potential of adult mouse hepatocytes. Am. J. Pathol. 151, 1273–1280 (1997). In this study, serial transplantations of hepatocytes confirmed that hepatocytes possess high proliferative potential.
Chen, X. & Calvisi, D. F. Hydrodynamic transfection for generation of novel mouse models for liver cancer research. Am. J. Pathol. 184, 912–923 (2014).
Fausto, N. & Campbell, J. S. Mouse models of hepatocellular carcinoma. Semin. Liver Dis. 30, 87–98 (2010).
Zender, L. et al. Cancer gene discovery in hepatocellular carcinoma. J. Hepatol. 52, 921–929 (2010).
Yimlamai, D. et al. Hippo pathway activity influences liver cell fate. Cell 157, 1324–1338 (2014).
Tschaharganeh, D. F. et al. Yes-associated protein up-regulates Jagged-1 and activates the Notch pathway in human hepatocellular carcinoma. Gastroenterology 144, 1530–1542.e12 (2013).
Fitamant, J. et al. YAP inhibition restores hepatocyte differentiation in advanced HCC, leading to tumor regression. Cell Rep. 10, 1692–1707 (2015).
Zhang, N. et al. The Merlin/NF2 tumor suppressor functions through the YAP oncoprotein to regulate tissue homeostasis in mammals. Dev. Cell 19, 27–38 (2010).
Dorrell, C. et al. Prospective isolation of a bipotential clonogenic liver progenitor cell in adult mice. Genes Dev. 25, 1193–1203 (2011).
Lemaigre, F. P. Hepatocytes as a source of cholangiocytes in injured liver. Hepatology 59, 726–728 (2014).
Malato, Y. et al. Fate tracing of mature hepatocytes in mouse liver homeostasis and regeneration. J. Clin. Invest. 121, 4850–4860 (2011).
Sekiya, S. & Suzuki, A. Hepatocytes, rather than cholangiocytes, can be the major source of primitive ductules in the chronically injured mouse liver. Am. J. Pathol. 184, 1468–1478 (2014).
Yanger, K. et al. Robust cellular reprogramming occurs spontaneously during liver regeneration. Genes Dev. 27, 719–724 (2013).
Sekiya, S. & Suzuki, A. Intrahepatic cholangiocarcinoma can arise from Notch-mediated conversion of hepatocytes. J. Clin. Invest. 122, 3914–3918 (2012).
Fan, B. et al. Cholangiocarcinomas can originate from hepatocytes in mice. J. Clin. Invest. 122, 2911–2915 (2012).
Zender, S. et al. A critical role for notch signaling in the formation of cholangiocellular carcinomas. Cancer Cell 23, 784–795 (2013). This study induced CCA by forced expression of the NOTCH1 intracellular domain in mouse livers, confirming the importance of the NOTCH pathway for iCCA development.
Tarlow, B. D. et al. Bipotential adult liver progenitors are derived from chronically injured mature hepatocytes. Cell Stem Cell 15, 605–618 (2014).
Guest, R. V. et al. Cell lineage tracing reveals a biliary origin of intrahepatic cholangiocarcinoma. Cancer Res. 74, 1005–1010 (2014).
Farber, E. Carcinoma of the liver in rats fed ethionine. AMA Arch. Pathol. 62, 445–453 (1956).
Opie, E. L. The pathogenesis of tumors of the liver produced by butter yellow. J. Exp. Med. 80, 231–246 (1944).
Evarts, R. P., Nagy, P., Marsden, E. & Thorgeirsson, S. S. A precursor-product relationship exists between oval cells and hepatocytes in rat liver. Carcinogenesis 8, 1737–1740 (1987).
Kitade, M. et al. Specific fate decisions in adult hepatic progenitor cells driven by MET and EGFR signaling. Genes Dev. 27, 1706–1717 (2013).
Benhamouche, S. et al. Nf2/Merlin controls progenitor homeostasis and tumorigenesis in the liver. Genes Dev. 24, 1718–1730 (2010).
Tschaharganeh, D. F. et al. p53-dependent Nestin regulation links tumor suppression to cellular plasticity in liver cancer. Cell 158, 579–592 (2014).
Saha, S. K. et al. Mutant IDH inhibits HNF-4α to block hepatocyte differentiation and promote biliary cancer. Nature 513, 110–114 (2014).
Hsia, C. C., Evarts, R. P., Nakatsukasa, H., Marsden, E. R. & Thorgeirsson, S. S. Occurrence of oval-type cells in hepatitis B virus-associated human hepatocarcinogenesis. Hepatology 16, 1327–1333 (1992).
Roskams, T. & Desmet, V. Ductular reaction and its diagnostic significance. Semin. Diagn. Pathol. 15, 259–269 (1998).
Akita, H. et al. MYC activates stem-like cell potential in hepatocarcinoma by a p53-dependent mechanism. Cancer Res. 74, 5903–5913 (2014).
Kakarala, M. & Wicha, M. S. Implications of the cancer stem-cell hypothesis for breast cancer prevention and therapy. J. Clin. Oncol. 26, 2813–2820 (2008).
Yong, K. J. et al. Oncofetal gene SALL4 in aggressive hepatocellular carcinoma. N. Engl. J. Med. 368, 2266–2276 (2013).
Hernandez-Gea, V., Toffanin, S., Friedman, S. L. & Llovet, J. M. Role of the microenvironment in the pathogenesis and treatment of hepatocellular carcinoma. Gastroenterology 144, 512–527 (2013).
He, G. et al. Identification of liver cancer progenitors whose malignant progression depends on autocrine IL-6 signaling. Cell 155, 384–396 (2013). This study demonstrates that cancer progenitor cells can only induce tumours when introduced into a liver with chronic damage, underlining the importance of the tumour microenvironment for HCC development.
Naugler, W. E. et al. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science 317, 121–124 (2007).
Park, E. J. et al. Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell 140, 197–208 (2010).
Guo, Y., Xu, F., Lu, T., Duan, Z. & Zhang, Z. Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat. Rev. 38, 904–910 (2012).
Makarova-Rusher, O. V., Medina-Echeverz, J., Duffy, A. G. & Greten, T. F. The yin and yang of evasion and immune activation in HCC. J. Hepatol. 62, 1420–1429 (2015).
Wan, S. et al. Tumor-associated macrophages produce interleukin 6 and signal via STAT3 to promote expansion of human hepatocellular carcinoma stem cells. Gastroenterology 147, 1393–1404 (2014).
Hoshida, Y. et al. Gene expression in fixed tissues and outcome in hepatocellular carcinoma. N. Engl. J. Med. 359, 1995–2004 (2008).
Maeda, S. et al. IkB kinaseb/nuclear factor-kB activation controls the development of liver metastasis by way of interleukin-6 expression. Hepatology 50, 1851–1860 (2009).
Elsharkawy, A. M. & Mann, D. A. Nuclear factor-kB and the hepatic inflammation–fibrosis–cancer axis. Hepatology 46, 590–597 (2007).
Luedde, T. & Schwabe, R. F. NF-kB in the liver — linking injury, fibrosis and hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 8, 108–118 (2011).
Marquardt, J. U. et al. Curcumin effectively inhibits oncogenic NF-κB signaling and restrains stemness features in liver cancer. J. Hepatol. 63, 661–669 (2015).
Karin, M. Nuclear factor-kB in cancer development and progression. Nature 441, 431–436 (2006).
Jakubowski, A. et al. TWEAK induces liver progenitor cell proliferation. J. Clin. Invest. 115, 2330–2340 (2005).
Bird, T. G. et al. Bone marrow injection stimulates hepatic ductular reactions in the absence of injury via macrophage-mediated TWEAK signaling. Proc. Natl Acad. Sci. USA 110, 6542–6547 (2013).
Boulter, L. et al. Macrophage-derived Wnt opposes Notch signaling to specify hepatic progenitor cell fate in chronic liver disease. Nat. Med. 18, 572–579 (2012).
Feng, G. S. Conflicting roles of molecules in hepatocarcinogenesis: paradigm or paradox. Cancer Cell 21, 150–154 (2012). This review critically discusses the context and cell type dependency of key oncogenic pathways for liver cancer development and progression.
Li, S. C. et al. Cancer genomic research at the crossroads: realizing the changing genetic landscape as intratumoral spatial and temporal heterogeneity becomes a confounding factor. Cancer Cell. Int. 14, 115 (2014).
Lanaya, H. et al. EGFR has a tumour-promoting role in liver macrophages during hepatocellular carcinoma formation. Nat. Cell Biol. 16, 972–981 (2014).
Timchenko, N. A. Cell-type specific functions of EGFR are involved in development of hepatocellular carcinoma. Hepatology 62, 314–316 (2015).
Hefetz-Sela, S. et al. Acquisition of an immunosuppressive protumorigenic macrophage phenotype depending on c-Jun phosphorylation. Proc. Natl Acad. Sci. USA 111, 17582–17587 (2014).
Ji, J. et al. Hepatic stellate cell and monocyte interaction contributes to poor prognosis in hepatocellular carcinoma. Hepatology 62, 481–495 (2015).
Trautwein, C., Friedman, S. L., Schuppan, D. & Pinzani, M. Hepatic fibrosis: concept to treatment. J. Hepatol. 62, S15–S24 (2015).
Coulouarn, C. & Clement, B. Stellate cells and the development of liver cancer: therapeutic potential of targeting the stroma. J. Hepatol. 60, 1306–1309 (2014).
Coulouarn, C. et al. Hepatocyte–stellate cell cross-talk in the liver engenders a permissive inflammatory microenvironment that drives progression in hepatocellular carcinoma. Cancer Res. 72, 2533–2542 (2012).
Kang, N., Gores, G. J. & Shah, V. H. Hepatic stellate cells: partners in crime for liver metastases? Hepatology 54, 707–713 (2011).
Gentilini, A. et al. Role of the stromal-derived factor-1 (SDF-1)–CXCR4 axis in the interaction between hepatic stellate cells and cholangiocarcinoma. J. Hepatol. 57, 813–820 (2012).
Greten, T. F., Wang, X. W. & Korangy, F. Current concepts of immune based treatments for patients with HCC: from basic science to novel treatment approaches. Gut 64, 842–848 (2015).
Bruix, J., Gores, G. J. & Mazzaferro, V. Hepatocellular carcinoma: clinical frontiers and perspectives. Gut 63, 844–855 (2014).
Quetglas, I. M., Moeini, A., Pinyol, R. & Llovet, J. M. Integration of genomic information in the clinical management of HCC. Best Pract. Res. Clin. Gastroenterol. 28, 831–842 (2014).
Sprinzl, M. F. & Galle, P. R. Immune control in hepatocellular carcinoma development and progression: role of stromal cells. Semin. Liver Dis. 34, 376–388 (2014).
Sprinzl, M. F. et al. Sorafenib inhibits macrophage-induced growth of hepatoma cells by interference with insulin-like growth factor-1 secretion. J. Hepatol. 62, 863–870 (2015).
Sangro, B. et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J. Hepatol. 59, 81–88 (2013). This is the first clinical trial to evaluate the safety and efficacy of checkpoint blockade in HCC. The results underline the potential of immunotherapy as a novel therapeutic approach for liver cancer.
Snyder, A. et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N. Engl. J. Med. 371, 2189–2199 (2014).
Heo, J. et al. Randomized dose-finding clinical trial of oncolytic immunotherapeutic vaccinia JX-594 in liver cancer. Nat. Med. 19, 329–336 (2013).
Le, D. T. et al. Safety and survival with GVAX pancreas prime and Listeria Monocytogenes-expressing mesothelin (CRS-207) boost vaccines for metastatic pancreatic cancer. J. Clin. Oncol. 33, 1325–1333 (2015).
Wang, J., Reiss Binder, K., Khatri, R., Jaffee, E. & Laheru, D. Immune therapy in GI malignancies: a review. J. Clin. Oncol. 33, 1745–1753 (2015).
Bruix, J., Han, K. H., Gores, G., Llovet, J. M. & Mazzaferro, V. Liver cancer: approaching a personalized care. J. Hepatol. 62, S144–S156 (2015).
Forner, A., Llovet, J. M. & Bruix, J. Hepatocellular carcinoma. Lancet 379, 1245–1255 (2012).
Rizvi, S. & Gores, G. J. Pathogenesis, diagnosis, and management of cholangiocarcinoma. Gastroenterology 145, 1215–1229 (2013).
Qian, M. B., Utzinger, J., Keiser, J. & Zhou, X. N. Clonorchiasis. Lancet http://dx.doi.org/10.1016/S0140-6736(15)60313-0 (2015).
Acknowledgements
This project was supported by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute, Bethesda, Maryland, USA (to S.S.T.). J.U.M. is supported by grants from the German Research Foundation (MA 4443/2-1), German Cancer Aid (DKH 110989) and the Volkswagen Foundation (Lichtenberg program). J.B.A. is supported by grants from the Danish Cancer Society (Knæk cancer program), the Novo Nordisk Foundation (Hallas-Møller fellowship), the Danish Medical Research Council (Sapere Aude program) and the A.P. Møller Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Liver cirrhosis
-
A slow and progressive replacement of healthy liver tissue by fibrotic scar tissue, with subsequent loss of organ function. Cirrhosis occurs as the common end point of the majority of chronic liver diseases.
- Precision medicine
-
A form of treatment that focuses on the individual factors of a disease and uses next-generation technologies to improve therapy.
- Multicentric HCCs
-
(Multicentric hepatocellular carcinomas). More than one independent tumour with different clonal origins detected in the liver.
- Genome substitution patterns
-
Patterns of nucleotide substitution across the whole tumour genome. Characteristic patterns can be detected in different tumours and/or aetiological backgrounds.
- Cellular reprogramming
-
Conversion of somatic cells to a more stem-like state or to a different developmental lineage.
- Hydrodynamic gene delivery
-
A highly efficient method for delivery of membrane-impermeable genetic information by physical force. A solution is rapidly injected at high volume to enable the delivery of genetic information to the target cell.
Rights and permissions
About this article
Cite this article
Marquardt, J., Andersen, J. & Thorgeirsson, S. Functional and genetic deconstruction of the cellular origin in liver cancer. Nat Rev Cancer 15, 653–667 (2015). https://doi.org/10.1038/nrc4017
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc4017
This article is cited by
-
Revealing the role of necroptosis microenvironment: FCGBP + tumor-associated macrophages drive primary liver cancer differentiation towards cHCC-CCA or iCCA
Apoptosis (2024)
-
Rhamnetin ameliorates non-alcoholic steatosis and hepatocellular carcinoma in vitro
Molecular and Cellular Biochemistry (2023)
-
A comprehensive analysis of the potential role of necroptosis in hepatocellular carcinoma using single-cell RNA Seq and bulk RNA Seq
Journal of Cancer Research and Clinical Oncology (2023)
-
Non-invasive imaging in the diagnosis of combined hepatocellular carcinoma and cholangiocarcinoma
Abdominal Radiology (2023)
-
Criteria for preclinical models of cholangiocarcinoma: scientific and medical relevance
Nature Reviews Gastroenterology & Hepatology (2023)