Abstract
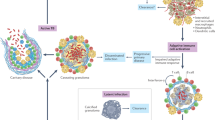
Virulent Mycobacterium tuberculosis inhibits apoptosis and triggers necrosis of host macrophages to evade innate immunity and delay the initiation of adaptive immunity. By contrast, attenuated M. tuberculosis induces macrophage apoptosis, an innate defence mechanism that reduces bacterial viability. In this Opinion article, we describe how virulent M. tuberculosis blocks production of the eicosanoid lipid mediator prostaglandin E2 (PGE2). PGE2 production by infected macrophages prevents mitochondrial damage and initiates plasma membrane repair, two processes that are crucial for preventing necrosis and inducing apoptosis. Thus, M. tuberculosis-mediated modulation of eicosanoid production determines the death modality of the infected macrophage, which in turn has a substantial impact on the outcome of infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Barry, C. E. 3rd et al. The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nature Rev. Microbiol. 7, 845–855 (2009).
Bhatt, K. & Salgame, P. Host innate immune response to Mycobacterium tuberculosis. J. Clin. Immunol. 27, 347–362 (2007).
Baena, A. & Porcelli, S. A. Evasion and subversion of antigen presentation by Mycobacterium tuberculosis. Tissue Antigens 74, 189–204 (2009).
Fink, S. L., Bergsbaken, T. & Cookson, B. T. Anthrax lethal toxin and Salmonella elicit the common cell death pathway of caspase-1-dependent pyroptosis via distinct mechanisms. Proc. Natl Acad. Sci. USA 105, 4312–4317 (2008).
Kroemer, G. et al. Classification of cell death: recommendations of the nomenclature committee on cell death 2009. Cell Death Differ. 16, 3–11 (2009).
Bergsbaken, T., Fink, S. L. & Cookson, B. T. Pyroptosis: host cell death and inflammation. Nature Rev. Microbiol. 7, 99–109 (2009).
Labbe, K. & Saleh, M. Cell death in the host response to infection. Cell Death Differ. 15, 1339–1349 (2008).
Haimovich, B. & Venkatesan, M. M. Shigella and Salmonella: death as a means of survival. Microbes Infect. 8, 568–577 (2006).
Bergsbaken, T. & Cookson, B. T. Macrophage activation redirects Yersinia-infected host cell death from apoptosis to caspase-1-dependent pyroptosis. PLoS Pathog. 3, e161 (2007).
Fink, S. L. & Cookson, B. T. Pyroptosis and host cell death responses during Salmonella infection. Cell. Microbiol. 9, 2562–2570 (2007).
Master, S. S. et al. Mycobacterium tuberculosis prevents inflammasome activation. Cell Host Microbe 3, 224–232 (2008).
Kurenuma, T. et al. The RD1 locus in the Mycobacterium tuberculosis genome contributes to activation of caspase-1 via induction of potassium ion efflux in infected macrophages. Infect. Immun. 77, 3992–4001 (2009).
Koo, I. C. et al. ESX-1-dependent cytolysis in lysosome secretion and inflammasome activation during mycobacterial infection. Cell. Microbiol. 10, 1866–1878 (2008).
Mayer-Barber, K. D. et al. Caspase-1 independent IL-1β production is critical for host resistance to Mycobacterium tuberculosis and does not require TLR signaling in vivo. J. Immunol. 184, 3326–3330 (2010).
Peters, N. C. et al. In vivo imaging reveals an essential role for neutrophils in leishmaniasis transmitted by sand flies. Science 321, 970–974 (2008).
Ritter, U., Frischknecht, F. & van Zandbergen, G. Are neutrophils important host cells for Leishmania parasites? Trends Parasitol. 25, 505–510 (2009).
Laskay, T., van Zandbergen, G. & Solbach, W. Neutrophil granulocytes – Trojan horses for Leishmania major and other intracellular microbes? Trends Microbiol. 11, 210–214 (2003).
Nogueira, C. V. et al. Rapid pathogen-induced apoptosis: a mechanism used by dendritic cells to limit intracellular replication of Legionella pneumophila. PLoS Pathog. 5, e1000478 (2009).
Gan, H. et al. Mycobacterium tuberculosis blocks crosslinking of annexin-1 and apoptotic envelope formation on infected macrophages to maintain virulence. Nature Immunol. 9, 1189–1197 (2008).
Duan, L., Gan, H., Arm, J. & Remold, H. G. Cytosolic phospholipase A2 participates with TNF-α in the induction of apoptosis of human macrophages infected with Mycobacterium tuberculosis H37Ra. J. Immunol. 166, 7469–7476 (2001).
Lee, J., Remold, H. G., Ieong, M. H. & Kornfeld, H. Macrophage apoptosis in response to high intracellular burden of Mycobacterium tuberculosis is mediated by a novel caspase-independent pathway. J. Immunol. 176, 4267–4274 (2006).
Oddo, M. et al. Fas ligand-induced apoptosis of infected human macrophages reduces the viability of intracellular Mycobacterium tuberculosis. J. Immunol. 160, 5448–5454 (1998).
Brookes, R. H. et al. CD8+ T cell-mediated suppression of intracellular Mycobacterium tuberculosis growth in activated human macrophages. Eur. J. Immunol. 33, 3293–3302 (2003).
Chen, M., Gan, H. & Remold, H. G. A mechanism of virulence: virulent Mycobacterium tuberculosis strain H37Rv, but not attenuated H37Ra, causes significant mitochondrial inner membrane disruption in macrophages leading to necrosis. J. Immunol. 176, 3707–3716 (2006).
Divangahi, M. et al. Mycobacterium tuberculosis evades macrophage defenses by inhibiting plasma membrane repair. Nature Immunol. 10, 899–906 (2009).
Chen, M. et al. Lipid mediators in innate immunity against tuberculosis: opposing roles of PGE2 and LXA4 in the induction of macrophage death. J. Exp. Med. 205, 2791–2801 (2008).
Keane, J., Remold, H. G. & Kornfeld, H. Virulent Mycobacterium tuberculosis strains evade apoptosis of infected alveolar macrophages. J. Immunol. 164, 2016–2020 (2000).
Hinchey, J. et al. Enhanced priming of adaptive immunity by a proapoptotic mutant of Mycobacterium tuberculosis. J. Clin. Invest. 117, 2279–2288 (2007).
Velmurugan, K. et al. Mycobacterium tuberculosis nuoG is a virulence gene that inhibits apoptosis of infected host cells. PLoS Pathog. 3, e110 (2007).
Wolf, L. A. & Laster, S. M. Characterization of arachidonic acid-induced apoptosis. Cell Biochem. Biophys. 30, 353–368 (1999).
Chang, D. J., Ringold, G. M. & Heller, R. A. Cell killing and induction of manganous superoxide dismutase by tumor necrosis factor-α is mediated by lipoxygenase metabolites of arachidonic acid. Biochem. Biophys. Res. Commun. 188, 538–546 (1992).
Peterson, D. A. et al. Polyunsaturated fatty acids stimulate superoxide formation in tumor cells: a mechanism for specific cytotoxicity and a model for tumor necrosis factor? Biochem. Biophys. Res. Commun. 155, 1033–1037 (1988).
Jayadev, S., Linardic, C. M. & Hannun, Y. A. Identification of arachidonic acid as a mediator of sphingomyelin hydrolysis in response to tumor necrosis factor α. J. Biol. Chem. 269, 5757–5763 (1994).
Finstad, H. S. et al. Cell proliferation, apoptosis and accumulation of lipid droplets in U937-1 cells incubated with eicosapentaenoic acid. Biochem. J. 336, 451–459 (1998).
Rocca, B. & FitzGerald, G. A. Cyclooxygenases and prostaglandins: shaping up the immune response. Int. Immunopharmacol. 2, 603–630 (2002).
Murakami, M. et al. Regulation of prostaglandin E2 biosynthesis by inducible membrane-associated prostaglandin E2 synthase that acts in concert with cyclooxygenase-2. J. Biol. Chem. 275, 32783–32792 (2000).
Sugimoto, Y. & Narumiya, S. Prostaglandin E receptors. J. Biol. Chem. 282, 11613–11617 (2007).
D'Avila, H. et al. Mycobacterium bovis bacillus Calmette–Guérin induces TLR2-mediated formation of lipid bodies: intracellular domains for eicosanoid synthesis in vivo. J. Immunol. 176, 3087–3097 (2006).
Almeida, P. E. et al. Mycobacterium bovis bacillus Calmette–Guérin infection induces TLR2-dependent peroxisome proliferator-activated receptor γ expression and activation: functions in inflammation, lipid metabolism, and pathogenesis. J. Immunol. 183, 1337–1345 (2009).
Levy, B. D., Clish, C. B., Schmidt, B., Gronert, K. & Serhan, C. N. Lipid mediator class switching during acute inflammation: signals in resolution. Nature Immunol. 2, 612–619 (2001).
Serhan, C. N., Chiang, N. & Van Dyke, T. E. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nature Rev. Immunol. 8, 349–361 (2008).
Tobin, D. M. et al. The lta4h locus modulates susceptibility to mycobacterial infection in zebrafish and humans. Cell 140, 717–730 (2010).
Zamzami, N. et al. Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J. Exp. Med. 181, 1661–1672 (1995).
Green, D. R. & Reed, J. C. Mitochondria and apoptosis. Science 281, 1309–1312 (1998).
Green, D. R. & Kroemer, G. The pathophysiology of mitochondrial cell death. Science 305, 626–629 (2004).
Gan, H. et al. Enhancement of antimycobacterial activity of macrophages by stabilization of inner mitochondrial membrane potential. J. Infect. Dis. 191, 1292–1300 (2005).
Roy, D. et al. A process for controlling intracellular bacterial infections induced by membrane injury. Science 304, 1515–1518 (2004).
Togo, T., Alderton, J. M., Bi, G. Q. & Steinhardt, R. A. The mechanism of facilitated cell membrane resealing. J. Cell Sci. 112, 719–731 (1999).
Granger, B. L. et al. Characterization and cloning of lgp110, a lysosomal membrane glycoprotein from mouse and rat cells. J. Biol. Chem. 265, 12036–12043 (1990).
Novikoff, P. M., Tulsiani, D. R., Touster, O., Yam, A. & Novikoff, A. B. Immunocytochemical localization of α-D-mannosidase II in the Golgi apparatus of rat liver. Proc. Natl Acad. Sci. USA 80, 4364–4368 (1983).
Martinez, I. et al. Synaptotagmin VII regulates Ca2+-dependent exocytosis of lysosomes in fibroblasts. J. Cell Biol. 148, 1141–1149 (2000).
Burgoyne, R. D., O'Callaghan, D. W., Hasdemir, B., Haynes, L. P. & Tepikin, A. V. Neuronal Ca2+-sensor proteins: multitalented regulators of neuronal function. Trends Neurosci. 27, 203–209 (2004).
Togo, T., Alderton, J. M. & Steinhardt, R. A. Long-term potentiation of exocytosis and cell membrane repair in fibroblasts. Mol. Biol. Cell 14, 93–106 (2003).
Regan, J. W. EP2 and EP4 prostanoid receptor signaling. Life Sci. 74, 143–153 (2003).
Bafica, A. et al. Host control of mycobacterium tuberculosis is regulated by 5-lipoxygenase-dependent lipoxin production. J. Clin. Invest. 115, 1601–1606 (2005).
Divangahi, M., Desjardins, D., Nunes-Alves, C., Remold, H. G. & Behar, S. M. Eicosanoid pathways regulate adaptive immunity to Mycobacterium tuberculosis. Nature Immunol. 11 Jul 2010 (doi:10.1038/ni.1904).
van der Wel, N. N. et al. M. tuberculosis and M. leprae translocate from the phagolysosome to the cytosol in myeloid cells. Cell 129, 1287–1298 (2007).
Weerdenburg, E. M., Peters, P. J. & van der Wel, N. N. How do mycobacteria activate CD8+ T cells? Trends Microbiol. 18, 1–10 (2009).
Volkman, H. E. et al. Tuberculous granuloma induction via interaction of a bacterial secreted protein with host epithelium. Science 327, 466–469 (2010).
Chackerian, A. A., Alt, J. M., Perera, T. V., Dascher, C. C. & Behar, S. M. Dissemination of Mycobacterium tuberculosis is influenced by host factors and precedes the initiation of T-cell immunity. Infect. Immun. 70, 4501–4509 (2002).
Aronoff, D. M. et al. E-prostanoid 3 receptor deletion improves pulmonary host defense and protects mice from death in severe Streptococcus pneumoniae infection. J. Immunol. 183, 2642–2649 (2009).
Medeiros, A. I., Serezani, C. H., Lee, S. P. & Peters-Golden, M. Efferocytosis impairs pulmonary macrophage and lung antibacterial function via PGE2/EP2 signaling. J. Exp. Med. 206, 61–68 (2009).
Acknowledgements
H.R.G. and S.M.B. are supported by the US National Institutes of Health (grant R01 AI073774).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Entrez Genome Project
Mycobacterium bovis bacille Calmette–Guérin
Salmonella enterica subsp. enterica serovar Typhimurium
FURTHER INFORMATION
Glossary
- Apoptosis
-
A type of programmed cell death defined by chromatin condensation (pyknosis) and fragmentation, blebbing of the plasma membrane and formation of apoptotic bodies. The plasma membrane of an apoptotic cell remains intact and contains proteins that are cross-linked by transglutaminases such as annexin 1.
- Efferocytosis
-
The uptake of apoptotic cells or apoptotic bodies by phagocytic cells.
- Eicosanoid
-
A lipid mediator that is derived from arachidonic acid. Eicosanoids include prostaglandins, lipoxins, leukotrienes, prostacyclins, thromboxanes and hydroxyeicosatetraenoic acid compounds.
- Mitochondrial membrane potential
-
The electrochemical gradient across the mitochondrial membranes, given the symbol ΔΨm. Complexes I, III and IV of the electron transport system in the inner mitochondrial membrane pump protons against their concentration gradient from the mitochondrial matrix into the inter-membrane space, making the matrix more negative.
- Mitochondrial permeability transition
-
An increase in the permeability of the mitochondrial membranes to molecules of less than 1,500 daltons.
- Necrosis
-
A form of cell death that is characterized by swelling of cytoplasmic organelles, including the mitochondria, and a loss of plasma membrane integrity.
- Plasma membrane microdisruption
-
A pore formed by damage of the plasma membrane, as determined by measuring the diffusion of fluorescent dextran, an inert impermeant molecule.
- Prostanoid
-
A lipid metabolite of arachidonic acid that is a product of the cyclooxygenase cascade and of specific prostanoid synthases.
- sn-2 position
-
The second (that is, middle) carbon atom in the glycerol backbone of phospholipids, providing a link for fatty acids.
Rights and permissions
About this article
Cite this article
Behar, S., Divangahi, M. & Remold, H. Evasion of innate immunity by Mycobacterium tuberculosis: is death an exit strategy?. Nat Rev Microbiol 8, 668–674 (2010). https://doi.org/10.1038/nrmicro2387
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2387
This article is cited by
-
γδ T cells: origin and fate, subsets, diseases and immunotherapy
Signal Transduction and Targeted Therapy (2023)
-
The Mycobacterium tuberculosis methyltransferase Rv2067c manipulates host epigenetic programming to promote its own survival
Nature Communications (2023)
-
Analysis of long non-coding RNA expression profile of bovine monocyte-macrophage infected by Mycobacterium avium subsp. paratuberculosis
BMC Genomics (2022)
-
Untargeted metabolomics analysis reveals Mycobacterium tuberculosis strain H37Rv specifically induces tryptophan metabolism in human macrophages
BMC Microbiology (2022)
-
Regulated necrosis, a proinflammatory cell death, potentially counteracts pathogenic infections
Cell Death & Disease (2022)