Key Points
-
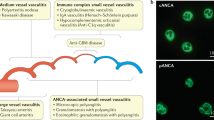
Aetiology, pathogenesis, pathology and clinical characteristics are the basis of the 2012 Chapel Hill Consensus Conference (CHCC) vasculitis nomenclature, and diagnostic and classification criteria for clinical practice are in development
-
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is now recognized by the CHCC nomenclature
-
The clinical approach to managing patients with AAV should include assessing: if the patient has proteinase 3-specific or myeloperoxidase-specific ANCAs; the extent and severity of disease; and disease activity
-
The dosage and duration of corticosteroid treatment as well as selection of patients with AAV for (and duration of) maintenance treatment are not well defined
-
Preventive treatment of infections and cardiovascular risk factors will improve outcome
-
Outcome measures should include not only disease activity, but also damage and quality of life
Abstract
The updated nomenclature for vasculitis defines this varied group of disorders by aetiology, specific features of pathogenesis and clinical symptoms; diagnostic and classification criteria for clinical practice are in development. Here, I review some important advances in the management of vasculitis within the category of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV), which encompasses microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA) and eosinophilic granulomatosis with polyangiitis (EGPA). The clinical approach to the management of the patient with AAV should include testing for ANCA specificity; proteinase 3 (PR3)-specific ANCAs are most often associated with GPA, whereas myeloperoxidase (MPO)-ANCAs are usually associated with MPA. Also important to the management of AAV is an assessment of the disease stage and severity, to enable tailored treatment based on an algorithm derived from controlled-trial data. Remaining questions pertain to the dosage and duration of corticosteroid treatment, the selection of patients for, and duration of, maintenance treatment after induction of remission, and the identification of safer and more effective therapies than are currently in use. Outcome measures should assess not only disease activity, but also damage and quality of life. Infections, cardiovascular events and malignancies also contribute to outcome, and their prevention should therefore be part of the clinical approach to managing patients with AAV.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hunder, G. G. et al. The American College of Rheumatology 1990 criteria for the classification of vasculitis. Introduction. Arthritis Rheum. 33, 1065–1067 (1990).
Fries, J. F. et al. The American College of Rheumatology 1990 criteria for the classification of vasculitis. Summary. Arthritis Rheum. 33, 1135–1136 (1990).
Jennette, J. C. et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum. 37, 187–192 (1994).
Watts, R. et al. Development and validation of a consensus methodology for the classification of the ANCA-associated vasculitides and polyarteritis nodosa for epidemiological studies. Ann. Rheum. Dis. 66, 222–227 (2007).
Liu, L. J., Chen, M., Yu, F., Zhao, M. H. & Wang, H. Y. Evaluation of a new algorithm in classification of systemic vasculitis. Rheumatology 47, 708–712 (2008).
Kamali, S. et al. Re-evaluation of 129 patients with systemic necrotizing vasculitis by using classification algorithm according to consensus methodology. Clin. Rheumatol. 31, 325–328 (2012).
Uribe, A. G. et al. Increased sensitivity of the European Medicines Agency algorithm for classification of childhood granulomatosis with polyangiitis. J. Rheumatol. 39, 1687–1697 (2012).
Abdulkader, R., Lane, S. E., Scott, D. G. & Watts, R. A. Classification of vasculitis: EMA classification using CHCC 2012 definitions. Ann. Rheum. Dis. 72, 1888 (2013).
Jennette, J. C. et al. 2012 Revised international Chapel Hill Consensus Conference nomenclature of vasculitides. Arthritis Rheum. 65, 1–11 (2013).
Lyons, P. A. et al. Genetically distinct subsets within ANCA-associated vasculitis. N. Engl. J. Med. 367, 214–223 (2012).
Chen, M., Kallenberg, C. G. & Zhao M. H. ANCA-negative pauci-immune crescentic glomerulonephritis. Nat. Rev. Nephrol. 5, 313–318 (2009).
Chen, M. et al. Antineutrophil cytoplasmic autoantibody negative pauci-immune crescentic glomerulonephritis. J. Am.Soc. Nephrol. 18, 599–605 (2007).
Kain, R. et al. High prevalence of autoantibodies to hLAMP-2 in anti-neutrophil cytoplasmic antibody-associated vasculitis. J. Am. Soc. Nephrol. 23, 556–566 (2012).
Roth, A. J. et al. Anti-LAMP-2 antibodies are not prevalent in patients with antineutrophil cytoplasmic autoantibody glomerulonephritis. J. Am. Soc. Nephrol. 23, 545–555 (2012).
Roth, A. J. et al. Epitope specificity determines pathogenicity and detectability in ANCA-associated vasculitis. J. Clin. Invest. 123, 1773–1783 (2013).
Land, J., Rutgers, A. & Kallenberg, C. G. Anti-neutrophil cytoplasmic autoantibody pathogenicity revisited: pathogenic versus non-pathogenic anti-neutrophil cytoplasmic autoantibody. Nephrol. Dial. Transplant. 29, 739–745 (2014).
Sinico, R. A. et al. Prevalence and clinical significance of antineutrophil cytoplasmic antibodies in Churg-Strauss syndrome. Arthritis Rheum. 52, 2926–2935 (2005).
Sablé-Fourtassou, R. et al. Antineutrophil cytoplasmic antibodies and the Churg-Strauss syndrome. Ann. Intern. Med. 143, 632–638 (2005).
Kallenberg, C. G. Churg-Strauss syndrome: just one disease entity? Arthritis Rheum. 52, 2589–2593 (2005).
International study group for Behçet's disease. Criteria for diagnosis of Behçet's disease. Lancet 335, 1078–1080 (1990).
Craven, A. et al. ACR/EULAR-endorsed study to develop diagnostic and classification criteria for vasculitis (DCVAS). Clin. Exp. Immunol. 17, 619–621 (2013).
Schönermarck, U., Gross, W. L. & de Groot, K. Treatment of ANCA-associated vasculitis. Nat. Rev. Nephrol. 10, 25–36 (2013).
Garske, U. et al. Intra- and inter-rater reliability of endonasal activity estimation in granulomatosis with polyangiitis (Wegener's). Clin. Exp. Rheumatol. 30, (1 Suppl. 70), S22–S28 (2012).
Luqmani, R. A. et al. Birmingham Vasculitis Activity Score (BVAS) in systemic necrotizing vasculitis. QJM 87, 671–678 (1994).
Mukhtyar, C. et al. Modification and validation of the Birmingham Vasculitis Activity Score (version 3). Ann. Rheum. Dis. 68, 1827–1832 (2009).
Suppiah, R. et al. A cross-sectional study of the Birmingham Vasculitis Activity Score version 3 in systemic vasculitis. Rheumatology (Oxford) 50, 899–905 (2011).
Stone, J. H. et al. A disease-specific activity index for Wegener's granulomatosis: modification of the Birmingham Vasculitis Activity Score. International Network for the Study of the Systemic Vasculitides (INSSYS). Arthritis Rheum. 44, 912–920 (2001).
Mahr, A. D. et al. Assessment of the item selection and weighting in the Birmingham vasculitis activity score for Wegener's granulomatosis. Arthritis Rheum. 59, 884–891 (2008).
Flossmann, O. et al. Long-term patient survival in ANCA-associated vasculitis. Ann. Rheum. Dis. 70, 488–494 (2011).
Dolezalova, P. et al. Disease activity assessment in childhood vasculitis: development and preliminary validation of the Paediatric Vasculitis Activity Score (PVAS). Ann. Rheum. Dis. 72, 1628–1633 (2013).
Exley, A. R. et al. Development and initial validation of the Vasculitis Damage Index for the standardized clinical assessment of damage in the systemic vasculitides. Arthritis Rheum. 40, 371–380 (1997).
Hellmich, B. et al. EULAR recommendations for conducting clinical studies and/or clinical trials in systemic vasculitis: focus on anti-neutrophil cytoplasm antibody-associated vasculitis. Ann. Rheum. Dis. 66, 605–617 (2007).
Mukhtyar, C. et al. Outcomes from studies of antineutrophil cytoplasm antibody-associated vasculitis: a systemic review by the European League Against Rheumatism systemic vasculitis task force. Ann. Rheum. Dis. 67, 1004–1010 (2008).
Robson, J. et al. Damage in ANCA-associated vasculitides: long-term data from the European Vasculitis Study group (EUVAS) therapeutic trials. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-203927 (2013).
Tomasson, G. et al. Assessment of health-related quality of life as an outcome measure in granulomatosis with polyangiitis (Wegener's). Arthritis Care Res. 64, 273–279 (2012).
Basu, N. et al. The characterization and determinants of quality of life in ANCA associated vasculitis. Ann. Rheum. Dis. 73, 207–211 (2013).
Mukhtyar, C. et al. EULAR recommendations for the management of primary small and medium vessel vasculitis. Ann. Rheum. Dis. 68, 310–317 (2009).
Hoffman, G. S. Vasculitis treatment: is it time to change the standard of care for ANCA-associated vasculitis? Presse Med. 42, 643–650 (2013).
Furuta, S. & Jayne, D. R. . Antineutrophil cytoplasm antibody-associated vasculitis: recent developments. Kidney Int. 84, 244–249 (2013).
Luqmani, R. Maintenance of clinical remission in ANCA-associated vasculitis. Nat. Rev. Rheumatol. 9, 127–132 (2013).
Franssen, C., Gans, R., Kallenberg, C., Hageluken, C. & Hoorntje, S. Disease spectrum of patients with antineutrophil cytoplasmic autoantibodies of defined specificity: distinct differences between patients with anti-proteinase 3 and anti-myeloperoxidase antibodies. J. Intern. Med. 244, 209–216 (1998).
Franssen, C. F. et al. Antiproteinase 3- and antimyeloperoxidase-associated vasculitis. Kidney Int. 57, 2195–2206 (2000).
de Joode, A. A., Sanders, J. S. & Stegeman, C. A. Renal survival in proteinase 3 and myeloperoxidase ANCA-associated systemic vasculitis. Clin. J. Am. Soc. Nephrol. 8, 1709–1717 (2013).
Lionaki, S. et al. Classification of antineutrophil cytoplasmic autoantibody vasculitides: the role of antineutrophil cytoplasmic autoantibody specificity for myeloperoxidase or proteinase 3 in disease recognition and prognosis. Arthritis Rheum. 64, 3452–3462 (2012).
Walsh, M. et al. Risk factors for relapse of antineutrophil cytoplasmic antibody associated vasculitis. Arthritis Rheum. 64, 542–548 (2012).
Stone, J. H. et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N. Engl. J. Med. 363, 221–232 (2010).
Specks, U. et al. Efficacy of remission-induction regimens for ANCA-associated vasculitis. N. Engl. J. Med. 369, 417–427 (2013).
Mahr, A. et al. Revising the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: a cluster analysis. Ann. Rheum. Dis. 72, 1003–1010 (2013).
Pendergraft, W. F. 3rd & Niles, J. L. Trojan horses: drug culprits associated with antineutrophil cytoplasmic autoantibody (ANCA) vasculitis. Curr. Opin. Rheumatol. 26, 42–49 (2014).
Graf, J. Rheumatic manifestations of cocaine use. Curr. Opin. Rheumatol. 25, 50–55 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2013).
Jennette, J. C., Xiao, H., & Hu, P. Complement in ANCA-associated vasculitis. Semin. Nephrol. 33, 557–564 (2013).
Xiao, H. et al. C5a receptor (CD88) blockade protects against MPO-ANCA GN. J. Am. Soc. Nephrol. 25, 225–231 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2013).
Hoffman, G. S. et al. Wegener Granulomatosis: an analysis of 158 patients. Ann. Intern. Med. 116, 488–498 (1992).
Jayne, D. et al. A randomized trial of maintenance therapy for vasculitis associated with antineutrophil cytoplasmic antibodies. N. Engl. J. Med. 349, 36–44 (2003).
Pagnoux, C. et al. Azathioprine or methotrexate maintenance for ANCA-associated vasculitis. N. Eng. J. Med. 359, 2790–2803 (2008).
Harper, L. et al. Pulse versus daily oral cyclophosphamide for induction of remission in ANCA-associated vasculitis: long-term follow-up. Ann. Rheum. Dis. 71, 955–960 (2012).
Monach, P. A. Biomarkers in vasculitis. Curr. Opin. Rheumatol. 26, 24–30 (2014).
McKinney, E. F. et al. A CD8+ T cell transcription signature predicts prognosis in autoimmune disease. Nat. Med. 16, 568–591 (2010).
Stegeman, C. A. et al. Association of chronic nasal carriage of Staphylococcus aureus and higher relapse rates in Wegener granulomatosis. Ann. Intern. Med. 120, 12–17 (1994).
Boomsma, M. M. et al. Prediction of relapses in Wegener's granulomatosis by measurement of antineutrophil cytoplasmic antibody levels: a prospective study. Arthritis Rheum. 43, 2025–2033 (2000).
Sanders, J. S., Huitma, M. G., Kallenberg, C. G. & Stegeman, C. A. Prediction of relapses in PR3-ANCA-associated vasculitis by assessing responses of ANCA titers to treatment. Rheumatology (Oxford) 45, 724–729 (2006).
Tomasson, G., Grayson, P. C., Mahr, A. D., Lavalley, M. & Merkel, P. A. Value of ANCA measurements during remission to predict a relapse of ANCA-associated vasculitis—a meta-analysis. Rheumatology (Oxford) 51, 100–109 (2012).
Cartin-Ceba, R. et al. Rituximab for remission induction and maintenance in refractory granulomatosis with polyangiitis (Wegener's): ten-year experience at a single center. Arthritis Rheum. 64, 3770–3778 (2012).
Smith, R. M. et al. Rituximab for remission maintenance in relapsing antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum. 64, 3760–3769 (2012).
Guillevin, L. et al. Rituximab versus azathioprine for maintenance in ANCA-associated vasculitis. A prospective study in 117 patients. Presse Med. 42, 679 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2013).
Khan, A., Lawson, C. A., Quinn, M. A., Isdale, A. H. & Green, M. J. Successful treatment of ANCA-negative Wegener's granulomatosis with rituximab. Int. J. Rheumatol. http://dx.doi.org/10.1155/2010/846063 (2010).
Mahr, A. et al. Eosinophilic granulomatosis with polyangiitis (Churg Strauss): evolutions in classifications, etiopathogenesis, assessment and management. Curr. Opin. Rheumatol. 26, 16–23 (2014).
Moosig, F., Gross, W. L., Herrmann, K., Bremer, J. P. & Hellmich, B. Targeting interleukin-5 in refractory and relapsing Churg-Strauss syndrome. Ann. Intern. Med. 155, 341–343 (2011).
Hermann, K., Gross, W. L. & Moosig, F. Extended follow-up after stopping mepolizumab in relapsing/refractory Churg-Strauss syndrome. Clin. Exp. Rheumatol. 30, (1 Suppl. 70), S62–S65 (2012).
Wall, N. & Harper, L. Complications of long-term therapy for ANCA-associated system vasculitis. Nat. Rev. Nephrol. 8, 523–533 (2012).
Harper, L. Morbidity in patients with ANCA-associated vasculitis. Presse Med. 42, 612–616 (2013).
Charlier, C. et al. Risk factors for major infections in Wegener granulomatosis: analysis of 113 patients. Ann. Rheum. Dis. 68, 658–663 (2009).
McGregor, J. G. et al. Glucocorticosteroids and relapse and infection rates in anti-neutrophil cytoplasmic antibody disease. Clin. J. Am. Soc. Nephrol. 7, 240–247 (2012).
Goupil, R. et al. Lymphopenia and treatment-related infectious complications in ANCA-associated vasculitis. Clin. J. Am. Soc. Nephrol. 8, 416–423 (2013).
Stegeman, C. A., Tervaert, J. W., de Jong, P. E. & Kallenberg, C. G. Trimethoprim-sulfamethoxazole (co-trimoxazole) for the prevention of relapses of Wegener's granulomatosis. N. Engl. J. Med. 335, 16–20 (1996).
Besada, E. & Nossent, J. C. Should Pneumocystis jiroveci prophylaxis be recommended with rituximab treatment in ANCA-associated vasculitis? Clin. Rheumatol. 32, 1677–1681 (2013).
Pugnet, G. et al. Progressive multifocal encephalopathy after cyclophosphamide in granulomatosis with polyangiitis (Wegener) patients: case report and review of the literature. Clin. Exp. Rheumatol. 31, (1 Suppl. 75), S62–S64 (2013).
Molloy, E. S. & Calabrese, L. H. Progressive multifocal leukoencephalopathy associated with immune suppressive therapy in rheumatic diseases: involving role of biologic therapies. Arthritis Rheum. 64, 3043–3051 (2012).
Besada, E., Koldingsnes, W. & Nossent, J. C. Long-term efficacy and safety of pre-emptive maintenance therapy with rituximab in granulomatosis with polyangiitis: results from a single center. Rheumatology (Oxford) 52, 2041–2047 (2013).
van Assen, S. et al. EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum.Dis. 70, 414–422 (2011).
Tichelaar, Y. I., Kluin-Nelemans, H. J., & Meijer, K. Infections and inflammatory diseases as risk factors for venous thrombosis. A systemic review. Thromb. Haemost. 107, 827–837 (2012).
Merkel, P. A. et al. Brief communication: high incidence of venous thrombotic events among patients with Wegener granulomatosis: the Wegener's Clinical Occurrence of Thrombosis (WeCLOT) study. Ann. Intern. Med. 142, 620–626 (2005).
Stassen, P. M., Derks, R. P, Kallenberg, C. G. & Stegeman, C. A. Venous thromboembolism in ANCA-associated vasculitis—incidence and risk factors. Rheumatology (Oxford) 47, 530–534 (2008).
Allenbach, Y. et al. High frequency of venous thromboembolic events in Churg-Strauss syndrome, Wegener's granulomatosis and microscopic polyangiitis but not polyarteritis nodosa: a systematic retrospective study on 1130 patients. Ann. Rheum. Dis. 68, 564–567 (2009).
Suppiah, R. et al. A model to predict cardiovascular events in patients with newly diagnosed Wegener's granulomatosis and microscopic polyangiitis. Arthritis Care Res. 63, 588–596 (2011).
Faurschou, M. et al. Increased morbidity from ischemic heart disease in patients with Wegener's granulomatosis. Arthritis Rheum. 60, 1187–1192 (2009).
Morgan, M. D. et al. Increased incidence of cardiovascular events in patients with antineutrophil cytoplasmic antibody-associated vasculitides: a matched-pair cohort study. Arthritis Rheum. 60, 3493–3500 (2009).
Cohen Tervaert, J. W. Cardiovascular disease due to accelerated atherosclerosis in systemic vasculitides. Best Pract. Res. Clin. Rheumatol. 27, 33–44 (2013).
Parker, B. & Bruce, I. SLE and metabolic syndrome. Lupus 22, 1259–1266 (2013).
Pankhurst, T., Savage, C. O., Gordon, C. & Harper, L. Malignancy is increased in ANCA-associated vasculitis. Rheumatology (Oxford) 43, 1532–1535 (2004).
Faurschou, M. et al. Cancer preceding Wegener's granulomatosis: a case–control study. Rheumatology (Oxford) 48, 421–424 (2009).
Talar-Williams, C. et al. Cyclophosphamide-induced cystitits and bladder cancer in patients with Wegener's granulomatosis. Ann. Intern. Med. 124, 477–484 (1996).
Westman, K. W., Bygren, P. G., Olsson, H., Ranstam, J. & Wieslander, J. Relapse rate, renal survival, and cancer morbidity in patients with Wegener's granulomatosis or microscopic polyangiitis with renal involvement. J. Am. Soc. Nephrol. 9, 842–852 (1998).
Le Guenno, G. et al. Incidence and predictors of urotoxic adverse events in cyclophosphamide-treated patients with systemic necrotizing vasculitides. Arthritis Rheum. 63, 1435–1445 (2011).
Heijl, C. et al. Incidence of malignancy in patients treated for antineutrophil cytoplasmic antibody-associated vasculitis: follow-up data from European vasculitis study group clinical trials. Ann. Rheum. Dis. 70, 1415–1421 (2011).
Wegener's Granulomatosis Etanercept Trial (WGET) Research Group. Etanercept plus standard therapy for Wegener's granulomatosis. N. Engl. J. Med. 352, 351–361 (2005).
Stone, J. H. et al. Solid malignancies among patients in the Wegener's granulomatosis etanercept trial. Arthritis Rheum. 54, 1608–1618 (2006).
Silva, F. et al. Solid malignancies among etanercept-treated patients with granulomatosis with polyangiitis (Wegener's): long-term follow up of a multicenter longitudinal cohort. Arthritis Rheum. 63, 2495–2503 (2011).
Mahr, A. Heijl, C., Le Guenno, G. & Faurschou, M. ANCA-associated vasculitis and malignancy: current evidence for cause and consequence relationships. Best Pract. Res. Clin. Rheumatol. 27, 45–56 (2013).
Kallenberg, C. G. Is B-cell depletion first choice in antineutrophil cytoplasmic antibody-associated vasculitis? Curr. Opin. Rheumatol. 26, 292–298 (2014).
Jones, R. et al. A randomized trial of mycophenolate mofetil versus cyclophosphamide for remission induction of ANCA-associated vasculitis: “MYCYC”. Presse Med. 42, 678–679 (2013).
Walsh, M. et al. Long-term follow-up of patients with severe ANCA-associated vasculitis comparing plasma exchange to intravenous methylprednisolone treatment is unclear. Kidney Int. 84, 397–402 (2013).
Hiemstra, T. F. et al. Mycophenolate mofetil vs azathioprine for remission maintenance in antineutrophil cytoplasmic antibody-associated vasculitis: a randomized controlled trial. JAMA 304, 2381–2388 (2010).
Acknowledgements
This work was supported by the European Union Seventh Framework Programme (FP7/2007-2013) grant number 261382.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Kallenberg, C. Key advances in the clinical approach to ANCA-associated vasculitis. Nat Rev Rheumatol 10, 484–493 (2014). https://doi.org/10.1038/nrrheum.2014.104
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.104
This article is cited by
-
Primary angiitis of the CNS and ANCA-associated vasculitis: from pathology to treatment
Rheumatology International (2023)
-
The clinical features and treatment of eosinophilic granulomatosis with polyangiitis (EGPA) in Turkey: one or two distinct diseases?
Internal and Emergency Medicine (2022)
-
Blood CD3-(CD56 or 16)+ natural killer cell distributions are heterogeneous in healthy adults and suppressed by azathioprine in patients with ANCA-associated vasculitides
BMC Immunology (2021)
-
Classification of ANCA-associated vasculitis: differences based on ANCA specificity and clinicopathologic phenotype
Rheumatology International (2021)
-
Clinical implication of plasma exchange on life-threatening antineutrophil cytoplasmic antibody-associated vasculitis
BMC Pulmonary Medicine (2020)