Abstract
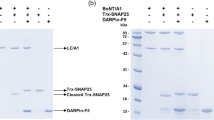
Clostridial botulinum neurotoxins (BoNTs) abort the process of neurotransmitter release at presynaptic motor nerve terminals, causing muscle paralysis. An enigmatic step in the intoxication process is the mechanism by which the neurotoxin heavy chain (HC) forms the conduit for the translocation of the light chain (LC) protease across the endosomal membrane into the cytosol, its site of action. Here we investigate the mechanism of LC translocation by using the combined detection of channel currents and substrate proteolysis, the two hallmark activities of BoNT. Our data are consistent with the translocation of the LC through the HC channel and show that the LC protease activity is retrieved in the trans compartment after translocation. We propose that the BoNT HC–LC complex embedded in the membrane is a transmembrane chaperone, a dynamic structural device that prevents aggregation and achieves translocation of the LC. In this regard, the complex is similar to the protein conducting/translocating channels of the endoplasmic reticulum, mitochondria and chloroplasts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schiavo, G. et al. Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature 359, 832–835 (1992).
Schiavo, G., Matteoli, M. & Montecucco, C. Neurotoxins affecting neuroexocytosis. Physiol. Rev. 80, 717–766 (2000).
Blasi, J. et al. Botulinum neurotoxin A selectively cleaves the synaptic protein SNAP-25. Nature 365, 160–163 (1993).
Sollner, T. et al. SNAP receptors implicated in vesicle targeting and fusion. Nature 362, 318–324 (1993).
Sutton, R.B., Fasshauer, D., Jahn, R. & Brunger, A.T. Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 Å resolution. Nature 395, 347–353 (1998).
Jahn, R. & Sudhof, T.C. Membrane fusion and exocytosis. Annu. Rev. Biochem. 68, 863–911 (1999).
Overly, C.C., Lee, K.D., Berthiaume, E. & Hollenbeck, P.J. Quantitative measurement of intraorganelle pH in the endosomal-lysosomal pathway in neurons by using ratiometric imaging with pyranine. Proc. Natl. Acad. Sci. USA 92, 3156–3160 (1995).
Sonawane, N.D., Thiagarajah, J.R. & Verkman, A.S. Chloride concentration in endosomes measured using a ratioable fluorescent Cl− indicator: evidence for chloride accumulation during acidification. J. Biol. Chem. 277, 5506–5513 (2002).
Lacy, D.B., Tepp, W., Cohen, A.C., DasGupta, B.R. & Stevens, R.C. Crystal structure of botulinum neurotoxin type A and implications for toxicity. Nat. Struct. Biol. 5, 898–902 (1998).
Swaminathan, S. & Eswaramoorthy, S. Structural analysis of the catalytic and binding sites of Clostridium botulinum neurotoxin B. Nat. Struct. Biol. 7, 693–699 (2000).
Donovan, J.J., Simon, M.I. & Montal, M. Insertion of diphtheria toxin into and across membranes: role of phosphoinositide asymmetry. Nature 298, 669–672 (1982).
Gambale, F. & Montal, M. Characterization of the channel properties of tetanus toxin in planar lipid bilayers. Biophys. J. 53, 771–783 (1988).
Oblatt-Montal, M., Yamazaki, M., Nelson, R. & Montal, M. Formation of ion channels in lipid bilayers by a peptide with the predicted transmembrane sequence of botulinum neurotoxin A. Protein Sci. 4, 1490–1497 (1995).
Hoch, D.H. et al. Channels formed by botulinum, tetanus, and diphtheria toxins in planar lipid bilayers: relevance to translocation of proteins across membranes. Proc. Natl. Acad. Sci. USA 82, 1692–1696 (1985).
Blaustein, R.O., Germann, W.J., Finkelstein, A. & DasGupta, B.R. The N-terminal half of the heavy chain of botulinum type A neurotoxin forms channels in planar phospholipid bilayers. FEBS Lett. 226, 115–120 (1987).
Sheridan, R.E. Gating and permeability of ion channels produced by botulinum toxin types A and E in PC12 cell membranes. Toxicon 36, 703–717 (1998).
Oh, K.J., Senzel, L., Collier, R.J. & Finkelstein, A. Translocation of the catalytic domain of diphtheria toxin across planar phospholipid bilayers by its own T domain. Proc. Natl. Acad. Sci. USA 96, 8467–8470 (1999).
Smart, O.S., Breed, J., Smith, G.R. & Sansom, M.S. A novel method for structure-based prediction of ion channel conductance properties. Biophys. J. 72, 1109–1126 (1997).
Mandel, R., Ryser, H.J., Ghani, F., Wu, M. & Peak, D. Inhibition of a reductive function of the plasma membrane by bacitracin and antibodies against protein disulfide-isomerase. Proc. Natl. Acad. Sci. USA 90, 4112–4116 (1993).
Siegert, R., Leroux, M.R., Scheufler, C., Hartl, F.U. & Moarefi, I. Structure of the molecular chaperone prefoldin: unique interaction of multiple coiled coil tentacles with unfolded proteins. Cell 103, 621–632 (2000).
Tsai, B., Rodighiero, C., Lencer, W.I. & Rapoport, T.A. Protein disulfide isomerase acts as a redox-dependent chaperone to unfold cholera toxin. Cell 104, 937–948 (2001).
Stebbins, C.E. & Galan, J.E. Maintenance of an unfolded polypeptide by a cognate chaperone in bacterial type III secretion. Nature 414, 77–81 (2001).
Shindyalov, I.N. & Bourne, P.E. Protein structure alignment by incremental combinatorial extension (CE) of the optimal path. Protein Eng. 11, 739–747 (1998).
Ren, J. et al. Interaction of diphtheria toxin T domain with molten globule-like proteins and its implications for translocation. Science 284, 955–957 (1999).
Beckmann, R. et al. Architecture of the protein-conducting channel associated with the translating 80S ribosome. Cell 107, 361–372 (2001).
Breyton, C., Haase, W., Rapoport, T.A., Kuhlbrandt, W. & Collinson, I. Three-dimensional structure of the bacterial protein-translocation complex SecYEG. Nature 418, 662–665 (2002).
Truscott, K.N. et al. A presequence- and voltage-sensitive channel of the mitochondrial preprotein translocase formed by Tim23. Nat. Struct. Biol. 8, 1074–1082 (2001).
Hinnah, S.C., Wagner, R., Sveshnikova, N., Harrer, R. & Soll, J. The chloroplast protein import channel toc75: pore properties and interaction with transit peptides. Biophys. J. 83, 899–911 (2002).
Heins, L. et al. The preprotein conducting channel at the inner envelope membrane of plastids. EMBO J. 21, 2616–2625 (2002).
Montal, M. Formation of bimolecular membranes from lipid monolayers. Methods Enzymol. 32, 545–554 (1974).
Li, L. & Singh, B.R. High-level expression, purification, and characterization of recombinant type A botulinum neurotoxin light chain. Protein Expr. Purif. 17, 339–344 (1999).
Blanes-Mira, C. et al. Thermal stabilization of the catalytic domain of botulinum neurotoxin E by phosphorylation of a single tyrosine residue. Biochemistry 40, 2234–2242 (2001).
Acevedo, B. et al. Development and validation of a quantitative ELISA for the measurement of PSA concentration. Clin. Chim. Acta 317, 55–63 (2002).
Canaves, J.M. & Montal, M. Assembly of a ternary complex by the predicted minimal coiled-coil-forming domains of syntaxin, SNAP-25, and synaptobrevin. A circular dichroism study. J. Biol. Chem. 273, 34214–34221 (1998).
Li, L. & Singh, B.R. Spectroscopic analysis of pH-induced changes in the molecular features of type A botulinum neurotoxin light chain. Biochemistry 39, 6466–6474 (2000).
Acknowledgements
We thank M. Goodnough for BoNT A and its individual chains; B.R. Singh and R.C. Stevens for the recombinant clones of BoNT A HC, LC and SNAP-25b; and N. Gude and J. Santos for perceptive comments. The project was supported by the U.S. Army Medical Research and Materiel Command.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Koriazova, L., Montal, M. Translocation of botulinum neurotoxin light chain protease through the heavy chain channel. Nat Struct Mol Biol 10, 13–18 (2003). https://doi.org/10.1038/nsb879
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb879
This article is cited by
-
High Yield Preparation of Functionally Active Catalytic-Translocation Domain Module of Botulinum Neurotoxin Type A That Exhibits Uniquely Different Enzyme Kinetics
The Protein Journal (2017)
-
Pre-Clinical Study of a Novel Recombinant Botulinum Neurotoxin Derivative Engineered for Improved Safety
Scientific Reports (2016)