Abstract
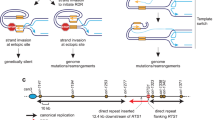
Coordination of homologous DNA recombination (HDR) with DNA replication maintains the fidelity of cell division. Exploiting Varshavsky's N-end rule to create a thermosensitive degron for conditional genetics in an avian cell line, we confirm that inactivation of the essential HDR enzyme Rad51 in a single cell cycle does not stop replicative DNA synthesis but, instead, causes G2 arrest. Rad51 complementation after the completion of replication overcomes this block, suggesting that HDR becomes necessary in G2. Indeed, DNA structures that bind activated replication protein A accumulate during the S phase, to be preferentially resolved during G2 by a Rad51-dependent mechanism. Breaks affecting a single chromatid predominate after the first cell cycle without Rad51, subsequently evolving into isochromatid lesions. We suggest a model for the vertebrate cell cycle in which HDR during the G2 phase is separated from DNA replication in S phase and chromosome segregation in M.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Paques, F. & Haber, J.E. Multiple pathways of recombination induced by double-strand breaks in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 63, 349–404 (1999).
West, S.C. Molecular views of recombination proteins and their control. Nat. Rev. Mol. Cell Biol. 4, 435–445 (2003).
Hoeijmakers, J.H. Genome maintenance mechanisms for preventing cancer. Nature 411, 366–374 (2001).
Wang, X. & Haber, J.E. Role of Saccharomyces single-stranded DNA-binding protein RPA in the strand invasion step of double-strand break repair. PLoS Biol. 2, e21 (2004).
Sugawara, N., Wang, X. & Haber, J.E. In vivo roles of Rad52, Rad54, and Rad55 proteins in Rad51-mediated recombination. Mol. Cell 12, 209–219 (2003).
Kanaar, R., Hoeijmakers, J.H. & van Gent, D.C. Molecular mechanisms of DNA double strand break repair. Trends Cell Biol. 8, 483–489 (1998).
Sung, P., Krejci, L., Van Komen, S. & Sehorn, M.G. Rad51 recombinase and recombination mediators. J. Biol. Chem. 278, 42729–42732 (2003).
Wyman, C., Ristic, D. & Kanaar, R. Homologous recombination-mediated double-strand break repair. DNA Repair (Amst.) 3, 827–833 (2004).
Symington, L.S. Role of RAD52 epistasis group genes in homologous recombination and double-strand break repair. Microbiol. Mol. Biol. Rev. 66, 630–670 (2002).
Sonoda, E. et al. Rad51-deficient vertebrate cells accumulate chromosomal breaks prior to cell death. EMBO J. 17, 598–608 (1998).
Takata, M. et al. The Rad51 paralog Rad51B promotes homologous recombinational repair. Mol. Cell. Biol. 20, 6476–6482 (2000).
Takata, M. et al. Chromosome instability and defective recombinational repair in knockout mutants of the five Rad51 paralogs. Mol. Cell. Biol. 21, 2858–2866 (2001).
Patel, K.J. et al. Involvement of Brca2 in DNA repair. Mol. Cell 1, 347–357 (1998).
Eppink, B., Wyman, C. & Kanaar, R. Multiple interlinked mechanisms to circumvent DNA replication roadblocks. Exp. Cell Res. 312, 2660–2665 (2006).
Dronkert, M.L. & Kanaar, R. Repair of DNA interstrand cross-links. Mutat. Res. 486, 217–247 (2001).
Patel, K.J. & Joenje, H. Fanconi anemia and DNA replication repair. DNA Repair (Amst.) 6, 885–890 (2007).
Lomonosov, M., Anand, S., Sangrithi, M., Davies, R. & Venkitaraman, A.R. Stabilization of stalled DNA replication forks by the BRCA2 breast cancer susceptibility protein. Genes Dev. 17, 3017–3022 (2003).
Henry-Mowatt, J. et al. XRCC3 and Rad51 modulate replication fork progression on damaged vertebrate chromosomes. Mol. Cell 11, 1109–1117 (2003).
Lusetti, S.L. & Cox, M.M. The bacterial RecA protein and the recombinational DNA repair of stalled replication forks. Annu. Rev. Biochem. 71, 71–100 (2002).
Khakhar, R.R., Cobb, J.A., Bjergbaek, L., Hickson, I.D. & Gasser, S.M. RecQ helicases: multiple roles in genome maintenance. Trends Cell Biol. 13, 493–501 (2003).
Michel, B., Grompone, G., Flores, M.J. & Bidnenko, V. Multiple pathways process stalled replication forks. Proc. Natl. Acad. Sci. USA 101, 12783–12788 (2004).
Yu, V.P. et al. Gross chromosomal rearrangements and genetic exchange between nonhomologous chromosomes following BRCA2 inactivation. Genes Dev. 14, 1400–1406 (2000).
Dohmen, R.J., Wu, P. & Varshavsky, A. Heat-inducible degron: a method for constructing temperature-sensitive mutants. Science 263, 1273–1276 (1994).
Bachmair, A., Finley, D. & Varshavsky, A. In vivo half-life of a protein is a function of its amino-terminal residue. Science 234, 179–186 (1986).
Labib, K., Tercero, J.A. & Diffley, J.F. Uninterrupted MCM2–7 function required for DNA replication fork progression. Science 288, 1643–1647 (2000).
Fukagawa, T., Regnier, V. & Ikemura, T. Creation and characterization of temperature-sensitive CENP-C mutants in vertebrate cells. Nucleic Acids Res. 29, 3796–3803 (2001).
Sonoda, E. et al. Sister chromatid exchanges are mediated by homologous recombination in vertebrate cells. Mol. Cell. Biol. 19, 5166–5169 (1999).
Dodson, H. et al. Centrosome amplification induces by DNA damage occurs during a prolonged G2 phase and involves ATM. EMBO J. 23, 3864–3873 (2004).
Zou, L. & Elledge, S.J. Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science 300, 1542–1548 (2003).
Block, W.D., Yu, Y. & Lees-Miller, S.P. Phosphatidyl inositol 3-kinase-like serine/threonine protein kinases (PIKKs) are required for DNA damage-induced phosphorylation of the 32 kDa subunit of replication protein A at threonine 21. Nucleic Acids Res. 32, 997–1005 (2004).
Raderschall, E., Golub, E.I. & Haaf, T. Nuclear foci of mammalian recombination proteins are located at single-stranded DNA regions formed after DNA damage. Proc. Natl. Acad. Sci. USA 96, 1921–1926 (1999).
Savage, J.R. Classification and relationships of induced chromosomal structural changes. J. Med. Genet. 13, 103–122 (1976).
Haber, J.E. DNA recombination: the replication connection. Trends Biochem. Sci. 24, 271–275 (1999).
Aguilera, A. & Gomez-Gonzalez, B. Genome instability: a mechanistic view of its causes and consequences. Nat. Rev. Genet. 9, 204–217 (2008).
Michel, B. et al. Rescue of arrested replication forks by homologous recombination. Proc. Natl. Acad. Sci. USA 98, 8181–8188 (2001).
Kogoma, T. Recombination by replication. Cell 85, 625–627 (1996).
Kuzminov, A. Collapse and repair of replication forks in Escherichia coli. Mol. Microbiol. 16, 373–384 (1995).
Merrill, B.J. & Holm, C. The RAD52 recombinational repair pathway is essential in pol30 (PCNA) mutants that accumulate small single-stranded DNA fragments during DNA synthesis. Genetics 148, 611–624 (1998).
Tsuzuki, T. et al. Targeted disruption of the Rad51 gene leads to lethality in embryonic mice. Proc. Natl. Acad. Sci. USA 93, 6236–6240 (1996).
Shinohara, A., Ogawa, H. & Ogawa, T. Rad51 protein involved in repair and recombination in S. cerevisiae is a RecA-like protein. Cell 69, 457–470 (1992).
Muris, D.F. et al. Homologous recombination in the fission yeast Schizosaccharomyces pombe: different requirements for the rhp51+, rhp54+ and rad22+ genes. Curr. Genet. 31, 248–254 (1997).
Lopes, M., Foiani, M. & Sogo, J.M. Multiple mechanisms control chromosome integrity after replication fork uncoupling and restart at irreparable UV lesions. Mol. Cell 21, 15–27 (2006).
Mojas, N., Lopes, M. & Jiricny, J. Mismatch repair-dependent processing of methylation damage gives rise to persistent single-stranded gaps in newly replicated DNA. Genes Dev. 21, 3342–3355 (2007).
Stojic, L. et al. Mismatch repair-dependent G2 checkpoint induced by low doses of SN1 type methylating agents requires the ATR kinase. Genes Dev. 18, 1331–1344 (2004).
Sogo, J.M., Lopes, M. & Foiani, M. Fork reversal and ssDNA accumulation at stalled replication forks owing to checkpoint defects. Science 297, 599–602 (2002).
Lopes, M. et al. The DNA replication checkpoint response stabilizes stalled replication forks. Nature 412, 557–561 (2001).
Meister, P. et al. Temporal separation of replication and recombination requires the intra-S checkpoint. J. Cell Biol. 168, 537–544 (2005).
Liberi, G. et al. Rad51-dependent DNA structures accumulate at damaged replication forks in sgs1 mutants defective in the yeast ortholog of BLM RecQ helicase. Genes Dev. 19, 339–350 (2005).
Branzei, D. et al. Ubc9- and mms21-mediated sumoylation counteracts recombinogenic events at damaged replication forks. Cell 127, 509–522 (2006).
Pfander, B., Moldovan, G.L., Sacher, M., Hoege, C. & Jentsch, S. SUMO-modified PCNA recruits Srs2 to prevent recombination during S phase. Nature 436, 428–433 (2005).
Sanchez-Diaz, A., Kanemaki, M., Marchesi, V. & Labib, K. Rapid depletion of budding yeast proteins by fusion to a heat-inducible degron. Sci. STKE 223, pl8 (2004).
Yu, D.S. et al. Dynamic control of Rad51 recombinase by self-association and interaction with BRCA2. Mol. Cell 12, 1029–1041 (2003).
Niedzwiedz, W. et al. The Fanconi anaemia gene FANCC promotes homologous recombination and error-prone DNA repair. Mol. Cell 15, 607–620 (2004).
Gallant, P. & Nigg, E.A. Cyclin B2 undergoes cell cycle-dependent nuclear translocation and, when expressed as a non-destructible mutant, causes mitotic arrest in HeLa cells. J. Cell Biol. 117, 213–224 (1992).
Sangrithi, M.N. et al. Initiation of DNA replication requires the RECQL4 protein mutated in Rothmund-Thomson syndrome. Cell 121, 887–898 (2005).
Sonoda, E. et al. Scc1/Rad21/Mcd1 is required for sister chromatid cohesion and kinetochore function in vertebrate cells. Dev. Cell 1, 759–770 (2001).
Acknowledgements
We are grateful to E. Nigg (Max Planck Institute, Martinsreid, Germany) for providing anti–cyclin B, K. Labib (PICR, Manchester) for providing pKL187 and S. Takeda (Kyoto University, Japan) for providing the Rad51−/−(tetRAD51) DT40 strain. We thank N. Ayoub for help in establishing the degron-EGFP system in DT40, E. Rajendra for technical help with the DT40 experiments and critical review of the manuscript, and members of our laboratory for helpful discussions. X.S. is supported by a studentship from A*STAR, Singapore, and J.A.B. by a Wellcome Trust grant to A.R.V. Work in A.R.V.'s laboratory is supported by the UK Medical Research Council.
Author information
Authors and Affiliations
Contributions
X.S. and J.A.B. together performed the experiments, with J.A.B. as the primary contributor to Figures 4c and 5c and Supplementary Figure 3, and X.S. as the primary contributor to the remaining figures. J.A.B. was instrumental in optimizing the experimental conditions for the use of N-end degrons in DT40 cells and provided guidance to X.S. X.S., J.A.B. and A.R.V. interpreted the data. X.S. and J.A.B. prepared the figures. A.R.V. supervised the work and wrote the paper with the help of X.S. and J.A.B.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 (PDF 968 kb)
Rights and permissions
About this article
Cite this article
Su, X., Bernal, J. & Venkitaraman, A. Cell-cycle coordination between DNA replication and recombination revealed by a vertebrate N-end rule degron-Rad51. Nat Struct Mol Biol 15, 1049–1058 (2008). https://doi.org/10.1038/nsmb.1490
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1490
This article is cited by
-
Acute hydroxyurea-induced replication blockade results in replisome components disengagement from nascent DNA without causing fork collapse
Cellular and Molecular Life Sciences (2020)
-
AND-1 fork protection function prevents fork resection and is essential for proliferation
Nature Communications (2018)
-
Loss of BRCA1 or BRCA2 markedly increases the rate of base substitution mutagenesis and has distinct effects on genomic deletions
Oncogene (2017)
-
Phenotypes on demand via switchable target protein degradation in multicellular organisms
Nature Communications (2016)
-
DNA replication and homologous recombination factors: acting together to maintain genome stability
Chromosoma (2013)