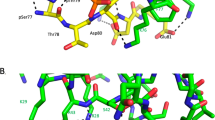
Abstract
Polo-like kinase-1 (Plk1) has a pivotal role in cell proliferation and is considered a potential target for anticancer therapy. The noncatalytic polo-box domain (PBD) of Plk1 forms a phosphoepitope binding module for protein-protein interaction. Here, we report the identification of minimal phosphopeptides that specifically interact with the PBD of human PLK1, but not those of the closely related PLK2 and PLK3. Comparative binding studies and analyses of crystal structures of the PLK1 PBD in complex with the minimal phosphopeptides revealed that the C-terminal SpT dipeptide functions as a high-affinity anchor, whereas the N-terminal residues are crucial for providing specificity and affinity to the interaction. Inhibition of the PLK1 PBD by phosphothreonine mimetic peptides was sufficient to induce mitotic arrest and apoptotic cell death. The mode of interaction between the minimal peptide and PBD may provide a template for designing therapeutic agents that target PLK1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
21 September 2010
In the version of this article initially published, two of the numbers shown in Table 1 had the wrong sign. The error has been corrected in the HTML and PDF versions of the article.
References
Barr, F.A., Sillje, H.H. & Nigg, E.A. Polo-like kinases and the orchestration of cell division. Nat. Rev. Mol. Cell Biol. 5, 429–440 (2004).
van de Weerdt, B.C. & Medema, R.H. Polo-like kinases: a team in control of the division. Cell Cycle 5, 853–864 (2006).
Lowery, D.M., Lim, D. & Yaffe, M.B. Structure and function of polo-like kinases. Oncogene 24, 248–259 (2005).
Eckerdt, F., Yuan, J. & Strebhardt, K. Polo-like kinases and oncogenesis. Oncogene 24, 267–276 (2005).
Strebhardt, K. & Ullrich, A. Targeting polo-like kinase 1 for cancer therapy. Nat. Rev. Cancer 6, 321–330 (2006).
Burns, T.F., Fei, P., Scata, K.A., Dicker, D.T. & El-Deiry, W.S. Silencing of the novel p53 target gene Snk/Plk2 leads to mitotic catastrophe in paclitaxel (taxol)-exposed cells. Mol. Cell. Biol. 23, 5556–5571 (2003).
Xie, S., Xie, B., Lee, M.Y. & Dai, W. Regulation of cell cycle checkpoints by polo-like kinases. Oncogene 24, 277–286 (2005).
Jang, Y.J., Lin, C.Y., Ma, S. & Erikson, R.L. Functional studies on the role of the C-terminal domain of mammalian polo-like kinase. Proc. Natl. Acad. Sci. USA 99, 1984–1989 (2002).
Lee, K.S., Grenfell, T.Z., Yarm, F.R. & Erikson, R.L. Mutation of the polo-box disrupts localization and mitotic functions of the mammalian polo kinase Plk. Proc. Natl. Acad. Sci. USA 95, 9301–9306 (1998).
Seong, Y.S. et al. A spindle checkpoint arrest and a cytokinesis failure by the dominant-negative polo-box domain of Plk1 in U-2 OS cells. J. Biol. Chem. 277, 32282–32293 (2002).
Elia, A.E., Cantley, L.C. & Yaffe, M.B. Proteomic screen finds pSer/pThr-binding domain localizing Plk1 to mitotic substrates. Science 299, 1228–1231 (2003).
Cheng, K.Y., Lowe, E.D., Sinclair, J., Nigg, E.A. & Johnson, L.N. The crystal structure of the human polo-like kinase-1 polo box domain and its phospho-peptide complex. EMBO J. 22, 5757–5768 (2003).
Elia, A.E. et al. The molecular basis for phospho-dependent substrate targeting and regulation of Plks by the polo-box domain. Cell 115, 83–95 (2003).
Kang, Y.H. et al. Self-regulation of Plk1 recruitment to the kinetochores is critical for chromosome congression and spindle checkpoint signaling. Mol. Cell 24, 409–422 (2006).
Minoshima, Y. et al. The constitutive centromere component CENP-50 is required for recovery from spindle damage. Mol. Cell. Biol. 25, 10315–10328 (2005).
Foltz, D.R. et al. The human CENP-A centromeric nucleosome-associated complex. Nat. Cell Biol. 8, 458–469 (2006).
Okada, M. et al. The CENP-H-I complex is required for the efficient incorporation of newly synthesized CENP-A into centromeres. Nat. Cell Biol. 8, 446–457 (2006).
Hanisch, A., Wehner, A., Nigg, E.A. & Sillje, H.H. Different Plk1 functions show distinct dependencies on Polo-Box domain-mediated targeting. Mol. Biol. Cell 17, 448–459 (2006).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Elia, A.E. et al. The molecular basis for phospho-dependent substrate targeting and regulation of Plks by the polo-box domain. Cell 115, 83–95 (2003).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Brünger, A.T. et al. Crystallography & NMR system: A new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
McRee, D.E. XtalView/Xfit–A versatile program for manipulating atomic coordinates and electron density. J. Struct. Biol. 125, 156–165 (1999).
Acknowledgements
We thank F.J. Gonzalez, C. Vinson and S. Garfield for critical reading of the manuscript, and R. Erikson (Harvard University) and W. Dai (New York University School of Medicine) for reagents and helpful suggestions. This research was supported in part by the Intramural Research Program of the National Cancer Institute (E.A., J.B.M., M.C.N., T.R.B., A.W. and K.S.L.), National Institutes of Health grant R01 GM60594 (M.B.Y.), Korea Basic Science Institute project N28079 (J.K.B.), a Korean Ministry of Education grant (D.H.L.) and a Japanese government grant (A.O.). This project was funded in whole or in part with federal funds from the National Cancer Institute, National Institutes of Health, under contract N01-CO-12400 and HHSN261200800001E. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products or organizations imply endorsement by the US government.
Author information
Authors and Affiliations
Contributions
K.S.L., T.M., D.L., J.-E.P., S.R.S., F.L., Y.H.K., A.W., M.B.Y. and T.R.B. designed the experiments; S.-M.Y., T.M., D.L., J.K.B., J.-E.P., S.R.S., F.L., Y.H.K., C.L., N.-K.S. and S.L. conducted the experiments; K.S.L., T.M., D.L., S.R.S., A.W., M.B.Y., T.R.B., J.B.M., D.-H.L., M.C.N., E.A., A.O., D.-Y.Y. and Y.L. analyzed the data; and K.S.L., T.M., D.L., S.R.S., A.W., M.B.Y., F.L. and T.R.B. wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Table 1 and Supplementary Methods (PDF 10352 kb)
Rights and permissions
About this article
Cite this article
Yun, SM., Moulaei, T., Lim, D. et al. Structural and functional analyses of minimal phosphopeptides targeting the polo-box domain of polo-like kinase 1. Nat Struct Mol Biol 16, 876–882 (2009). https://doi.org/10.1038/nsmb.1628
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1628
This article is cited by
-
The Eyes Absent family members EYA4 and EYA1 promote PLK1 activation and successful mitosis through tyrosine dephosphorylation
Nature Communications (2024)
-
Synthesis and evaluation of small molecule-based derivatives as inhibitors of polo-box domain of polo-like kinase-1
Journal of Analytical Science and Technology (2023)
-
Crystal Structures of Plk1 Polo-Box Domain Bound to the Human Papillomavirus Minor Capsid Protein L2-Derived Peptide
Journal of Microbiology (2023)
-
A cryptic hydrophobic pocket in the polo-box domain of the polo-like kinase PLK1 regulates substrate recognition and mitotic chromosome segregation
Scientific Reports (2019)
-
A NCAPG2-Derived Phosphopeptide Selectively Binds to the Polo-Box Domain of PLK1 and Inhibits Cancer Cell Proliferation
International Journal of Peptide Research and Therapeutics (2019)