Abstract
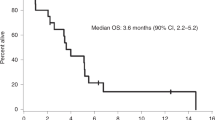
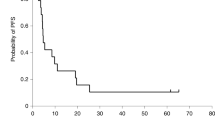
Brain and leptomeningeal metastases are common in breast cancer patients and our current treatments are ineffective. Reovirus type 3 is a replication competent, naturally occurring virus that usurps the activated Ras-signaling pathway (or an element thereof) of tumor cells and lyses them but leaves normal cells relatively unaffected. In this study we evaluated reovirus as an experimental therapeutic in models of central nervous system (CNS) metastasis from breast cancer. We found all breast cancer cell lines tested were susceptible to reovirus, with >50% of these cells lysed within 72 h of infection. In vivo neurotoxicity studies showed only mild local inflammation at the injection site and mild communicating hydrocephalus with neither diffuse encephalitis nor behavioral abnormalities at the therapeutically effective dose of reovirus (intracranial) (ie 107 plaque-forming units) or one dose level higher. In vivo, a single intratumoral administration of reovirus significantly reduced the size of tumors established from two human breast cancer cell lines and significantly prolonged survival. Intrathecal administration of reovirus also remarkably prolonged survival in an immunocompetent racine model of leptomeningeal metastases. These data suggest that the evaluation of reovirus as an experimental therapeutic for CNS metastases from breast cancer is warranted.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Posner JB, Chernik NL . Intracranial metastases from systemic cancer. Adv Neurol 1978; 19: 579–592.
Tsukata Y, Fouad A, Pickren JW, Lane WW . Central nervous system metastasis from breast carcinoma: autopsy study. Cancer 1983; 52: 2349–2354.
Lee YTN . Patterns of metastasis and natural courses of breast carcinoma. Cancer Metast Rev 1985; 4: 153–172.
Lee YTN . Breast carcinoma: pattern of recurrence and metastasis after mastestomy. Am J Clin Oncol 1984; 7: 334–344.
Boogerd W, VOs VW, Hart AAM, Baris G . Brain metastases in breast cancer: natural history, prognostic factors and outcome. J Neuro-Oncol 1993; 15: 165–174.
Kirn D, Martuza RL, Zwiebel J . Replication-selective virotherapy for cancer: biological principles, risk management and future directions. Nat Med 2001; 7: 781–787.
Makower D et al. Phase II clinical trial of intralesional administration of the oncolytic adenovirus ONYX-015 in patients with hepatobiliary tumors with correlative p53 studies. Clin Cancer Res 2003; 9: 693–702.
Habib N et al. Clinical trial of E1B-deleted adenovirus (dl1520) gene therapy for hepatocellular carcinoma. Cancer Gene Ther 2002; 9: 254–259.
Markert JM et al. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Therapy 2000; 7: 867–874.
Pecora AL et al. Phase I trial of intravenous administration of PV701, an oncolytic virus, in patients with advanced solid cancers. J Clin Oncol 2002; 20: 2251–2266.
Toda M, Rabkin SD, Martuza RL . Treatment of human breast cancer in a brain metastatic model by G207, a replication-competent multimutated herpes simplex virus 1. Hum Gene Ther 1998; 9: 2177–2185.
Randazzo BP et al. Treatment of experimental murine melanoma with a neuroattenuated herpes simplex virus 1 mutant. Virology 1995; 211: 94–101.
Tyler K, Fields BN . Reoviruses. In: Fields BN, Knipe DM, Howley PM, Chanock RM, Melnick JL, Monath TP (eds). Virology, 3nd edn Lippincott-Raven: Philadelphia, PA, 1996, pp 1597–1623.
Sherry B et al. Lymphocytes protect against and are not required for reovirus-induced myocarditis. J Virol 1993; 67: 6119–6124.
Raine CS, Fields BN . Reovirus type III encephalitis – a virologic and ultrastructural study. J Neropathol Exp Neurol 1973; 32: 19–33.
Paul RW, Choi AHC, Lee PWK . The alpha-anomeric form of sialic acid is the minimal receptor determinant recognized by reovirus. Virology 1989; 172: 382–385.
Choi AHC, Paul RW, Lee PWK . Reovirus binds to multiple plasma membrane proteins on mouse L fibroblasts. Virology 1990; 178: 316–320.
Barton ES et al. Junction adhesion molecule is a receptor for reovirus. Cell 2001; 104: 441–451.
Coffey MC et al. Reovirus therapy of tumors with activated ras pathway. Science 1998; 282: 1332–1334.
Strong JE, Tang D, Lee PW . Evidence that the epidermal growth factor receptor on host cells confers reovirus infection efficiency. Virology 1993; 197: 405–411.
Strong JE, Lee PW . The v-erbB oncogene confers enhanced cellular susceptibility to reovirus infection. J Virol 1996; 70: 612–616.
Strong J et al. The molecular basis of viral oncolysis: usurpation of the Ras signaling pathway by reovirus. EMBO J 1998; 17: 3351–3362.
Norman KL et al. Reovirus oncolysis of human breast cancer. Hum Gene Ther 2002; 13: 641–652.
Wilcox ME et al. Reovirus as an oncolytic agent against experimental human malignant gliomas. J Natl Cancer Inst 2001; 93: 903–912.
Yang WQ et al. Reovirus prolongs survival and reduces the frequency of spinal and leptomeningeal metastases from medulloblastoma. Cancer Res 2003; 63: 3162–3172.
Hirasawa K et al. Oncolytic reovirus against ovarian and colon cancer. Cancer Res 2002; 62: 1696–1701.
Hirasawa K et al. Systemic reovirus therapy of metastatic cancer in immune-competent mice. Cancer Res 2003; 63: 348–353.
Alain T et al. Reovirus therapy of lymphoid malignancies. Blood 2002; 100: 4146–4153.
Thirukkumaran CM et al. Reovirus oncolysis as a novel purging strategy for autologous stem cell transplantation. Blood 2003; 102: 377–387.
Etoh T et al. Oncolytic viral therapy for human pancreatic cancer cells by reovirus. Clin Cancer Res 2003; 9: 1218–2123.
Slamon DJ et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 1989; 244: 707–712.
Ross JS, Fletcher JA . The HER-2/neu oncogene in breast cancer: prognostic factor, predictive factor, and target for therapy. Oncologist 1998; 3: 237–252.
Ben-levy R, Paterson HF, Marshall CJ, Yarden YA . A single autophosphorylation site confers oncogenicity to the Neu/ErbB-2 receptor and enables coupling to the MAP kinase pathway. EMBO J 1994; 13: 3302–3311.
Koenders PG et al. Epidermal growth factor receptor-negative tumors are predominantly confined to the subgroup of estradiol receptor-positive human primary breast cancers. Cancer Res 1991; 51: 4544–4548.
Verbeek BS et al. c-Src protein expression is increased in human breast cancer. An immunohistochemical and biochemical analysis. J Pathol 1996; 180: 383–388.
Walter EZ et al. Impaired spatial cognition and synaptic potentiation in a murine model of human immunodeficiency virus type 1 encephalitis. Exp Neurol 1992; 116: 122–132.
Thifault S et al. Comparisons between C57BL/6J and A/J mice in motor activity and coordination, hole-poking, and spatial learning. Brain Res Bull 2001; 58: 213–218.
Sutherland RJ, Richard HD . Place navigation by rats in a swimming pool. Canad J Psychol 1984; 38: 322–347.
Morris RGM, Garrud P, Rawlins JNP, O'Keefe J . Place navigation impaired in rats with heppocampal lesions. Nature 1982; 297: 681–683.
Sagar SM, Price KJ . An experimental model of leptomeningeal metastases employing rat mammary carcinoma cells. J Neurooncol 1995; 23: 15–21.
Yang M et al. Whole-body optical imaging of green flourescent protein-expressing tumors and metastases. Proc Natl Acad Sci USA 2000; 97: 1206–1211.
Jacobs C, Rubsamen H . Expression of pp60c-src protein kinase in adult and fetal human tissue: High activities in some sarcomas and mammary carcinomas. Cancer Res 1983; 43: 1696–1702.
Shackney SE et al. Intracellular coexpression of epidermal growth factor receptor, Her-2/neu, and p21ras in human breast cancers: evidence for the existence of distinctive patterns of genetic evolution that are common to tumors from different patients. Clin Cancer Res 1998; 4: 913–928.
Carter P et al. Humanization of an antip185HER2 antibody for human cancer therapy. Proc Natl Acad Sci USA 1992; 89: 4285–4289.
Clynes RA, Towers TL, Presta LG, Ravetch JV . Inhibitory Fc receptors modulate in vivo cytoxicity against tumor targets. Nat Med 2000; 6: 443–446.
Ikeda K et al. Oncolytic virus therapy of multiple tumors in the brain requires suppression of innate and elicited antiviral response. Nat Med 1999; 5: 881–887.
Clarke P et al. Caspase 8-dependent sensitization of cancer cells to TRAIL-induced apoptosis following reovirus-infection. Oncogene 2001; 20: 6910–6919.
Connolly JL et al. Reovirus-induced apoptosis requires activation of transcription factor NF-kappaB. J Virol 2000; 74: 2981–2989.
Clarke P et al. Reovirus-induced apoptosis is mediated by TRAIL. J Virol 2000; 74: 8135–8139.
Clarke P et al. Reovirus infection activates JNK and the JNK-dependent transcription factor c-Jun. J Virol 2001; 75: 11275–11283.
Richardson-Burns SM, Kominsky DJ, Tyler KL . Reovirus-induced neuronal apoptosis is mediated by caspase 3 and is associated with the activation of death receptors. J Neurovirol 2002; 8: 365–380.
Kominsky DJ, Bickel RJ, Tyler KL . Reovirus-induced apoptosis requires both death receptor- and mitochondrial-mediated caspase-dependent pathways of cell death. Cell Death Differ 2002; 9: 926–933.
Morris D et al. A phase I clinical trial evaluating intralesional Reolysin (reovirus) in histologically confirmed malignancies. Proc Am Soc Clin Oncol 2002; 21:92:24a.
Acknowledgements
This work was funded by the National Cancer Institute of Canada (NCIC) with funds raised from the Canadian Cancer Society, the Canadian Institutes of Health Research (CIHR), the Alberta Cancer Board and the Kid's Cancer Care Foundation (KCCF). WQY is a KCCF fellow and DS is an Alberta Heritage for Medical Research fellow. KN is supported by an AHFMR studentship. We thank the families of Dr Micheal Longinotto, Gordon Stewart, Grant Tims and Clark Smith for their support.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yang, W., Senger, D., Lun, X. et al. Reovirus as an experimental therapeutic for brain and leptomeningeal metastases from breast cancer. Gene Ther 11, 1579–1589 (2004). https://doi.org/10.1038/sj.gt.3302319
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302319
Keywords
This article is cited by
-
Oncolytic HSV1 targets different growth phases of breast cancer leptomeningeal metastases
Cancer Gene Therapy (2023)
-
Phase 1 Clinical Trial of Intratumoral Reovirus Infusion for the Treatment of Recurrent Malignant Gliomas in Adults
Molecular Therapy (2014)
-
Brain metastases as preventive and therapeutic targets
Nature Reviews Cancer (2011)
-
Intrathecal gene therapy for treatment of leptomeningeal carcinomatosis
Journal of Neuro-Oncology (2011)
-
Intravenous administration of Reolysin®, a live replication competent RNA virus is safe in patients with advanced solid tumors
Investigational New Drugs (2010)