Abstract
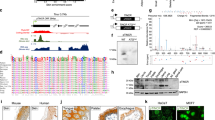
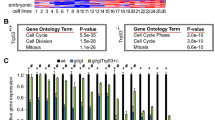
The 19q13 amplicon in pancreatic cancer cells contains a novel pancreatic differentiation 2 (PD2) gene (accession number AJ401156), which was identified by differential screening analysis. PD2 is the human homologue of the RNA polymerase II-associated factor 1 (hPaf1). In yeast, Paf1 is part of the transcription machinery, acting as a docking protein in between the complexes Rad6-Bre1, COMPASS-Dot1p, and the phosphorylated carboxyl terminal domain of the RNA polymerase II. As such, Paf1 is directly involved in transcription elongation via histone H2B ubiquitination and histone H3 methylation. The PD2 sequence is highly conserved from Drosophila to humans with up to 98% identity between rodent and human, suggesting the functional importance of PD2/hPaf1 to maintain cellular homeostasis. PD2 is a modular protein composed of RNA recognition motif, DEAD-boxes, an aspartic/serine (DS)-domain, a regulator of the chromosome condensation domain and myc-type helix–loop–helix domains. Our results further showed that PD2 is a nuclear 80 kDa protein, which interacts with RNA polymerase II. In addition, we have demonstrated that the overexpression of PD2 in the NIH 3T3 cells result in enhanced growth rates in vitro and tumor formation in vivo. Altogether, this paper presents strong evidence that the overexpression of PD2/hPaf1 is involved in cancer development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Abbreviations
- PD2:
-
pancreatic differentiation 2
- Paf1:
-
RNA polymerase II-associated factor
- HLH:
-
helix–loop–helix
- cAMP:
-
cyclic adenosine monophosphate
- kb:
-
kilobase
- bp:
-
base pair
- PCR:
-
polymerase chain reaction
- CGH:
-
comparative genomic hybridization
- RRM:
-
eukaryotic RNA recognition motif
- RCC1:
-
regulator of chromosome condensation domain
- RNAPII:
-
RNA polymerase II
- GTFs:
-
general transcription factors
References
Altomare DA, Tanno S, De RA, Klein-Szanto AJ, Tanno S, Skele KL et al. (2003). J Cell Biochem 88: 470–476.
Arboleda MJ, Lyons JF, Kabbinavar FF, Bray MR, Snow BE, Ayala R et al. (2003). Cancer Res 63: 196–206.
Batra SK, Metzgar RS, Hollingsworth MA . (1991a). Cell Growth Differ 2: 385–390.
Batra SK, Metzgar RS, Hollingsworth MA . (1991b). J Biol Chem 266: 6830–6833.
Bauer B, Baier G . (2002). Mol Immunol 38: 1087–1099.
Bellacosa A, de FD, Godwin AK, Bell DW, Cheng JQ, Altomare DA et al. (1995). Int J Cancer 64: 280–285.
Bellacosa A, Testa JR, Staal SP, Tsichlis PN . (1991). Science 254: 274–277.
Bellanne-Chantelot C, Lacroix B, Ougen P, Billault A, Beaufils S, Bertrand S et al. (1992). Cell 70: 1059–1068.
Berry R, Stevens TJ, Walter NA, Wilcox AS, Rubano T, Hopkins JA et al. (1995). Nat Genet 10: 415–423.
Betz JL, Chang M, Washburn TM, Porter SE, Mueller CL, Jaehning JA . (2002). Mol Genet Genomics 268: 272–285.
Bjorkqvist AM, Husgafvel-Pursiainen K, Anttila S, Karjalainen A, Tammilehto L, Mattson K et al. (1998). Genes Chromosomes Cancer 22: 79–82.
Blain SW, Massague J . (2002). Nat Med 8: 1076–1078.
Cheng JQ, Altomare DA, Klein MA, Lee WC, Kruh GD, Lissy NA et al. (1997). Oncogene 14: 2793–2801.
Cheng JQ, Ruggeri B, Klein WM, Sonoda G, Altomare DA, Watson DK et al. (1996). Proc Natl Acad Sci USA 93: 3636–3641.
Chirgwin JM, Przybyla AE, MacDonald RJ, Rutter WJ . (1979). Biochemistry 18: 5294–5299.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y et al. (1997). Cell 91: 231–241.
Gaudray P, Szepetowski P, Escot C, Birnbaum D, Theillet C . (1992). Mutat Res 276: 317–328.
Gingras AC, Raught B, Sonenberg N . (2001). Genes Dev 15: 807–826.
Grande MA, Van der Kraan I, de Jong L, Van Driel R . (1997). J Cell Sci 110: 1781–1791.
Griffin CA, Hruban RH, Long PP, Morsberger LA, Douna-Issa F, Yeo CJ . (1994). Genes Chromosomes Cancer 9: 93–100.
Hampsey M, Reinberg D . (2003). Cell 113: 429–432.
Heselmeyer K, Macville M, Schrock E, Blegen H, Hellstrom AC, Shah K et al. (1997). Genes Chromosomes Cancer 19: 233–240.
Jones PF, Jakubowicz T, Hemmings BA . (1991). Cell Regul 2: 1001–1009.
Kallioniemi A, Kallioniemi OP, Piper J, Tanner M, Stokke T, Chen L et al. (1994). Proc Natl Acad Sci USA 91: 2156–2160.
Kennedy SG, Kandel ES, Cross TK, Hay N . (1999). Mol Cell Biol 19: 5800–5810.
Khatib ZA, Matsushime H, Valentine M, Shapiro DN, Sherr CJ, Look AT . (1993). Cancer Res 53: 5535–5541.
Kim YW, Kern HF, Mullins TD, Koriwchak MJ, Metzgar RS . (1989). Pancreas 4: 353–362.
Lan MS, Batra SK, Qi WN, Metzgar RS, Hollingsworth MA . (1990). J Biol Chem 265: 15294–15299.
Lee KA, Green MR . (1990). Methods Enzymol 181: 20–30.
Lengauer C, Kinzler KW, Vogelstein B . (1998). Nature 396: 643–649.
Lieber M, Mazzetta J, Nelson-Rees W, Kaplan M, Todaro G . (1975). Int J Cancer 15: 741–747.
Meyer T, Begitt A, Lodige I, van Rossum M, Vinkemeier U . (2002). EMBO J 21: 344–354.
Mueller CL, Porter SE, Hoffman MG, Jaehning JA . (2004). Mol Cell 14: 447–456.
Petersen I, Langreck H, Wolf G, Schwendel A, Psille R, Vogt P et al. (1997). Br J Cancer 75: 79–86.
Ried T, Petersen I, Holtgreve-Grez H, Speicher MR, Schrock E, du MS et al. (1994). Cancer Res 54: 1801–1806.
Ruggeri BA, Huang L, Wood M, Cheng JQ, Testa JR . (1998). Mol Carcinog 21: 81–86.
Shin I, Yakes FM, Rojo F, Shin NY, Bakin AV, Baselga J et al. (2002). Nat Med 8: 1145–1152.
Singh AP, Moniaux N, chauhan SC, Meza JL, Batra SK . (2004). Cancer Res 64: 622–630.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL . (1987). Science 235: 177–182.
Squazzo SL, Costa PJ, Lindstrom DL, Kumer KE, Simic R, Jennings JL et al. (2002). EMBO J 21: 1764–1774.
Werner CA, Dohner H, Joos S, Trumper LH, Baudis M, Barth TF et al. (1997). Am J Pathol 151: 335–342.
Wilcox AS, Khan AS, Hopkins JA, Sikela JM . (1991). Nucleic Acids Res 19: 1837–1843.
Woodard GE, Lin L, Zhang JH, Agarwal SK, Marx SJ, Simonds WF . (2005). Oncogene 24: 1272–1276.
Acknowledgements
The work reported in this manuscript was initiated in the late 1980s by Drs Batra and Hollingsworth under the direction and sponsorship of Professor Richard S Metzgar (Duke University Medical Center, NC, USA), whom authors gratefully acknowledge for his thoughtful and scholarly insights, exemplary leadership, generous support and lasting friendship. We would like to thank Mr Jason Jokerst for making mouse monoclonal antibodies, Mr Erik Moore for technical assistance, the Molecular Biology Core Facility, UNMC, for oligonucleotide synthesis and DNA sequencing, Monoclonal Antibody Core Facility for making antibodies and Ms Kristi LW Berger for editorial assistance. Dr Nemos acknowledges the Travel Award Fellowship from the American/French Philippe Foundation. This work was supported, in part, by grants from the NIH (CA78590 and CA72712).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moniaux, N., Nemos, C., Schmied, B. et al. The human homologue of the RNA polymerase II-associated factor 1 (hPaf1), localized on the 19q13 amplicon, is associated with tumorigenesis. Oncogene 25, 3247–3257 (2006). https://doi.org/10.1038/sj.onc.1209353
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209353
Keywords
This article is cited by
-
Transcription recycling assays identify PAF1 as a driver for RNA Pol II recycling
Nature Communications (2021)
-
Novel role of PAF1 in attenuating radiosensitivity in cervical cancer by inhibiting IER5 transcription
Radiation Oncology (2020)
-
A peptide encoded by circular form of LINC-PINT suppresses oncogenic transcriptional elongation in glioblastoma
Nature Communications (2018)
-
The PAF complex regulation of Prmt5 facilitates the progression and maintenance of MLL fusion leukemia
Oncogene (2018)
-
Paf1 and Ctr9 subcomplex formation is essential for Paf1 complex assembly and functional regulation
Nature Communications (2018)