Abstract
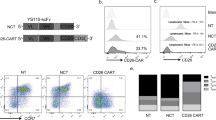
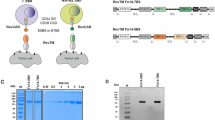
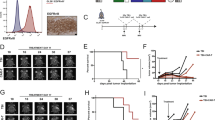
The epidermal growth factor receptor variant III (EGFRvIII) is exclusively expressed on the cell surface in ~50% of glioblastoma multiforme (GBM). This variant strongly and persistently activates the phosphatidylinositol 3-kinase-Akt signaling pathway in a ligand-independent manner resulting in enhanced tumorigenicity, cellular motility and resistance to chemoradiotherapy. Our group generated a recombinant single-chain variable fragment (scFv) antibody specific to the EGFRvIII, referred to as 3C10-scFv. In the current study, we constructed a lentiviral vector transducing the chimeric antigen receptor (CAR) that consisted of 3C10-scFv, CD3ζ, CD28 and 4-1BB (3C10-CAR). The 3C10-CAR-transduced peripheral blood mononuclear cells (PBMCs) and CD3+ T cells specifically lysed the glioma cells that express EGFRvIII. Moreover, we demonstrated that CAR CD3+ T cells migrated to the intracranial xenograft of GBM in the mice treated with 3C10-CAR PBMCs. An important and novel finding of our study was that a thalidomide derivative lenalidomide induced 3C10-CAR PBMC proliferation and enhanced the persistent antitumor effect of the cells in vivo. Lenalidomide also exhibited enhanced immunological synapses between the effector cells and the target cells as determined by CD11a and F-actin polymerization. Collectively, lentiviral-mediated transduction of CAR effectors targeting the EGFRvIII showed specific efficacy, and lenalidomide even intensified CAR cell therapy by enhanced formation of immunological synapses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005; 352: 987–996.
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 2009; 10: 459–466.
Rosenberg SA . Progress in human tumour immunology and immunotherapy. Nature 2001; 411: 380–384.
Johnson LA, Sampson JH . Immunotherapy approaches for malignant glioma from 2007 to 2009. Curr Neurol Neurosci Rep 2010; 10: 259–266.
Chung DS, Shin HJ, Hong YK . A new hope in immunotherapy for malignant gliomas: adoptive T cell transfer therapy. J Immunol Res 2014; 2014: 326545.
Wong AJ, Ruppert JM, Bigner SH, Grzeschik CH, Humphrey PA, Bigner DS et al. Structural alterations of the epidermal growth factor receptor gene in human gliomas. Proc Natl Acad Sci USA 1992; 89: 2965–2969.
Heimberger AB, Hlatky R, Suki D, Yang D, Weinberg J, Gilbert M et al. Prognostic effect of epidermal growth factor receptor and EGFRvIII in glioblastoma multiforme patients. Clin Cancer Res 2005; 11: 1462–1466.
Inda MM, Bonavia R, Mukasa A, Narita Y, Sah DW, Vandenberg S et al. Tumor heterogeneity is an active process maintained by a mutant EGFR-induced cytokine circuit in glioblastoma. Genes Dev 2010; 24: 1731–1745.
Wikstrand CJ, Hale LP, Batra SK, Hill ML, Humphrey PA, Kurpad SN et al. Monoclonal antibodies against EGFRvIII are tumor specific and react with breast and lung carcinomas and malignant gliomas. Cancer Res 1995; 55: 3140–3148.
Mellinghoff IK, Wang MY, Vivanco I, Haas-Kogan DA, Zhu S, Dia EQ et al. Molecular determinants of the response of glioblastomas to EGFR kinase inhibitors. N Engl J Med 2005; 353: 2012–2024.
Batra SK, Castelino-Prabhu S, Wikstrand CJ, Zhu X, Humphrey PA, Friedman HS et al. Epidermal growth factor ligand-independent, unregulated, cell-transforming potential of a naturally occurring human mutant EGFRvIII gene. Cell Growth Differ 1995; 6: 1251–1259.
Boockvar JA, Kapitonov D, Kapoor G, Schouten J, Counelis GJ, Bogler O et al. Constitutive EGFR signaling confers a motile phenotype to neural stem cells. Mol Cell Neurosci 2003; 24: 1116–1130.
Zoller S, Joos A, Dinter D, Back W, Horisberger K, Post S et al. Retrorectal tumors: excision by transanal endoscopic microsurgery. Rev Esp Enferm Dig 2007; 99: 547–550.
Al-Nedawi K, Meehan B, Micallef J, Lhotak V, May L, Guha A et al. Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat Cell Biol 2008; 10: 619–624.
Montgomery RB, Guzman J, O’Rourke DM, Stahl WL . Expression of oncogenic epidermal growth factor receptor family kinases induces paclitaxel resistance and alters beta-tubulin isotype expression. J Biol Chem 2000; 275: 17358–17363.
Lammering G, Hewit TH, Valerie K, Contessa JN, Amorino GP, Dent P et al. EGFRvIII-mediated radioresistance through a strong cytoprotective response. Oncogene 2003; 22: 5545–5553.
Lammering G, Hewit TH, Holmes M, Valerie K, Hawkins W, Lin PS et al. Inhibition of the type III epidermal growth factor receptor variant mutant receptor by dominant-negative EGFR-CD533 enhances malignant glioma cell radiosensitivity. Clin Cancer Res 2004; 10: 6732–6743.
Okamoto S, Yoshikawa K, Obata Y, Shibuya M, Aoki S, Yoshida J et al. Monoclonal antibody against the fusion junction of a deletion-mutant epidermal growth factor receptor. Br J Cancer 1996; 73: 1366–1372.
Nakayashiki N, Yoshikawa K, Nakamura K, Hanai N, Okamoto K, Okamoto S et al. Production of a single-chain variable fragment antibody recognizing type III mutant epidermal growth factor receptor. Jpn J Cancer Res 2000; 91: 1035–1043.
Ohno M, Natsume A, Ichiro Iwami K, Iwamizu H, Noritake K, Ito D et al. Retrovirally engineered T-cell-based immunotherapy targeting type III variant epidermal growth factor receptor, a glioma-associated antigen. Cancer Sci 2010; 101: 2518–2524.
Ohno M, Ohkuri T, Kosaka A, Tanahashi K, June CH, Natsume A et al. Expression of miR-17-92 enhances anti-tumor activity of T-cells transduced with the anti-EGFRvIII chimeric antigen receptor in mice bearing human GBM xenografts. J Immunother Cancer 2013; 1: 21.
Eshhar Z, Waks T, Gross G, Schindler DG . Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proc Natl Acad Sci USA 1993; 90: 720–724.
Morgan RA, Johnson LA, Davis JL, Zheng Z, Woolard KD, Reap EA et al. Recognition of glioma stem cells by genetically modified T cells targeting EGFRvIII and development of adoptive cell therapy for glioma. Hum Gene Ther 2012; 23: 1043–1053.
Cartellieri M, Bachmann M, Feldmann A, Bippes C, Stamova S, Wehner R et al. Chimeric antigen receptor-engineered T cells for immunotherapy of cancer. J Biomed Biotechnol 2010; 2010: 956304.
Facoetti A, Nano R, Zelini P, Morbini P, Benericetti E, Ceroni M et al. Human leukocyte antigen and antigen processing machinery component defects in astrocytic tumors. Clin Cancer Res 2005; 11: 8304–8311.
Curran KJ, Pegram HJ, Brentjens RJ . Chimeric antigen receptors for T cell immunotherapy: current understanding and future directions. J Gene Med 2012; 14: 405–415.
Rolle CE, Sengupta S, Lesniak MS . Mechanisms of immune evasion by gliomas. Adv Exp Med Biol 2012; 746: 53–76.
Sakamaki I, Kwak LW, Cha SC, Yi Q, Lerman B, Chen J et al. Lenalidomide enhances the protective effect of a therapeutic vaccine and reverses immune suppression in mice bearing established lymphomas. Leukemia 2014; 28: 329–337.
Ramsay AG, Johnson AJ, Lee AM, Gorgun G, Le Dieu R, Blum W et al. Chronic lymphocytic leukemia T cells show impaired immunological synapse formation that can be reversed with an immunomodulating drug. J Clin Invest 2008; 118: 2427–2437.
Huppa JB, Davis MM . T-cell-antigen recognition and the immunological synapse. Nat Rev Immunol 2003; 3: 973–983.
Lu L, Payvandi F, Wu L, Zhang LH, Hariri RJ, Man HW et al. The anti-cancer drug lenalidomide inhibits angiogenesis and metastasis via multiple inhibitory effects on endothelial cell function in normoxic and hypoxic conditions. Microvasc Res 2009; 77: 78–86.
Desai M, Newberry K, Ou Z, Wang M, Zhang L . Lenalidomide in relapsed or refractory mantle cell lymphoma: overview and perspective. Ther Adv Hematol 2014; 5: 91–101.
Muller GW, Chen R, Huang SY, Corral LG, Wong LM, Patterson RT et al. Amino-substituted thalidomide analogs: potent inhibitors of TNF-alpha production. Bioorg Med Chem Lett 1999; 9: 1625–1630.
Corral LG, Haslett PA, Muller GW, Chen R, Wong LM, Ocampo CJ et al. Differential cytokine modulation and T cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-alpha. J Immunol 1999; 163: 380–386.
LeBlanc R, Hideshima T, Catley LP, Shringarpure R, Burger R, Mitsiades N et al. Immunomodulatory drug costimulates T cells via the B7-CD28 pathway. Blood 2004; 103: 1787–1790.
Luptakova K, Rosenblatt J, Glotzbecker B, Mills H, Stroopinsky D, Kufe T et al. Lenalidomide enhances anti-myeloma cellular immunity. Cancer Immunol Immunother 2013; 62: 39–49.
Galustian C, Meyer B, Labarthe MC, Dredge K, Klaschka D, Henry J et al. The anti-cancer agents lenalidomide and pomalidomide inhibit the proliferation and function of T regulatory cells. Cancer Immunol Immunother 2009; 58: 1033–1045.
Idler I, Giannopoulos K, Zenz T, Bhattacharya N, Nothing M, Dohner H et al. Lenalidomide treatment of chronic lymphocytic leukaemia patients reduces regulatory T cells and induces Th17 T helper cells. Br J Haematol 2010; 148: 948–950.
Dustin ML, Depoil D . New insights into the T cell synapse from single molecule techniques. Nat Rev Immunol 2011; 11: 672–684.
Grakoui A, Bromley SK, Sumen C, Davis MM, Shaw AS, Allen PM et al. The immunological synapse: a molecular machine controlling T cell activation. Science 1999; 285: 221–227.
Ramsay AG, Clear AJ, Kelly G, Fatah R, Matthews J, Macdougall F et al. Follicular lymphoma cells induce T-cell immunologic synapse dysfunction that can be repaired with lenalidomide: implications for the tumor microenvironment and immunotherapy. Blood 2009; 114: 4713–4720.
Gomez TS, Hamann MJ, McCarney S, Savoy DN, Lubking CM, Heldebrant MP et al. Dynamin 2 regulates T cell activation by controlling actin polymerization at the immunological synapse. Nat Immunol 2005; 6: 261–270.
Yokosuka T, Sakata-Sogawa K, Kobayashi W, Hiroshima M, Hashimoto-Tane A, Tokunaga M et al. Newly generated T cell receptor microclusters initiate and sustain T cell activation by recruitment of Zap70 and SLP-76. Nat Immunol 2005; 6: 1253–1262.
Giglio P, Dhamne M, Hess KR, Gilbert MR, Groves MD, Levin VA et al. Phase 2 trial of irinotecan and thalidomide in adults with recurrent anaplastic glioma. Cancer 2012; 118: 3599–3606.
Alexander BM, Wang M, Yung WK, Fine HA, Donahue BA, Tremont IW et al. A phase II study of conventional radiation therapy and thalidomide for supratentorial, newly-diagnosed glioblastoma (RTOG 9806). J Neurooncol 2013; 111: 33–39.
Acknowledgements
This work was supported by a Grant-in Aid for Scientific Research on Innovative Areas from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) (AN no. 23107010) and a Grant-in-Aid for Young Scientists B (MO no. 25861270). We would like to thank Mr Masamitsu Harada (Cellgene) for his constructive suggestions and Mr Yutaka Hattori (Keyence) for his technical assistance in immunofluorescence imaging.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Kuramitsu, S., Ohno, M., Ohka, F. et al. Lenalidomide enhances the function of chimeric antigen receptor T cells against the epidermal growth factor receptor variant III by enhancing immune synapses. Cancer Gene Ther 22, 487–495 (2015). https://doi.org/10.1038/cgt.2015.47
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2015.47
This article is cited by
-
Outcomes of first therapy after CD19-CAR-T treatment failure in large B-cell lymphoma
Leukemia (2023)
-
CAR-T cell combination therapy: the next revolution in cancer treatment
Cancer Cell International (2022)
-
Lenalidomide enhances the efficacy of anti-BCMA CAR-T treatment in relapsed/refractory multiple myeloma: a case report and revies of the literature
Cancer Immunology, Immunotherapy (2022)
-
CAR T-cell immunotherapy: a powerful weapon for fighting hematological B-cell malignancies
Frontiers of Medicine (2021)
-
Driving cars to the clinic for solid tumors
Gene Therapy (2018)