Abstract
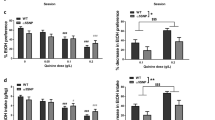
Smoking is the most important preventable cause of morbidity and mortality worldwide. Recent genome-wide association studies highlighted a human haplotype on chromosome 15 underlying the risk for tobacco dependence and lung cancer. Several polymorphisms in the CHRNA3-CHRNA5-CHRNB4 cluster coding for the nicotinic acetylcholine receptor (nAChR) α3, α5 and β4 subunits were implicated. In mouse models, we define a key role in the control of sensitivity to nicotine for the α5 subunit in dopaminergic (DAergic) neurons of the ventral tegmental area (VTA). We first investigated the reinforcing effects of nicotine in drug-naive α5−/− mice using an acute intravenous nicotine self-administration task and ex vivo and in vivo electrophysiological recordings of nicotine-elicited DA cell activation. We designed lentiviral re-expression vectors to achieve targeted re-expression of wild-type or mutant α5 in the VTA, in general, or in DA neurons exclusively. Our results establish a crucial role for α5*-nAChRs in DAergic neurons. These receptors are key regulators that determine the minimum nicotine dose necessary for DA cell activation and thus nicotine reinforcement. Finally, we demonstrate that a single-nucleotide polymorphism, the non-synonymous α5 variant rs16969968, frequent in many human populations, exhibits a partial loss of function of the protein in vivo. This leads to increased nicotine consumption in the self-administration paradigm. We thus define a critical link between a human predisposition marker, its expression in DA neurons and nicotine intake.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Changeux J-P . Nicotine addiction and nicotinic receptors: lessons from genetically modified mice. Nat Rev Neurosci 2010; 11: 389–401.
De Biasi M, Dani JA . Reward, addiction, withdrawal to nicotine. Annu Rev Neurosci 2011; 34: 105–130.
Salas R, Pieri F, De Biasi M . Decreased signs of nicotine withdrawal in mice null for the beta4 nicotinic acetylcholine receptor subunit. J Neurosci 2004; 24: 10035–10039.
Wang JC, Cruchaga C, Saccone NL, Bertelsen S, Liu P, Budde JP et al. Risk for nicotine dependence and lung cancer is conferred by mRNA expression levels and amino acid change in CHRNA5. Hum Mol Genet 2009; 18: 3125–3135.
Bierut LJ, Stitzel JA, Wang JC, Hinrichs AL, Grucza RA, Xuei X et al. Variants in nicotinic receptors and risk for nicotine dependence. Am J Psychiatry 2008; 165: 1163–1171.
Gotti C, Clementi F, Fornari A, Gaimarri A, Guiducci S, Manfredi I et al. Structural and functional diversity of native brain neuronal nicotinic receptors. Biochem Pharmacol 2009; 78: 703–711.
Klink R, de Kerchove d'Exaerde A, Zoli M, Changeux JP . Molecular and physiological diversity of nicotinic acetylcholine receptors in the midbrain dopaminergic nuclei. J Neurosci 2001; 21: 1452–1463.
Fowler CD, Lu Q, Johnson PM, Marks MJ, Kenny PJ . Habenular α5 nicotinic receptor subunit signalling controls nicotine intake. Nature 2011; 471: 597–601.
Chatterjee S, Santos N, Holgate J, Haass-Koffler CL, Hopf FW, Kharazia V et al. The α5 subunit regulates the expression and function of α4*-containing neuronal nicotinic acetylcholine receptors in the ventral-tegmental area. PLoS ONE 2013; 8: e68300.
Tuesta LM, Fowler CD, Kenny PJ . Recent advances in understanding nicotinic receptor signaling mechanisms that regulate drug self-administration behavior. Biochem Pharmacol 2011; 82: 984–995.
Salas R, Sturm R, Boulter J, De Biasi M . Nicotinic receptors in the habenulo-interpeduncular system are necessary for nicotine withdrawal in mice. J Neurosci 2009; 29: 3014–3018.
Di Ciano P, Everitt BJ . Contribution of the ventral tegmental area to cocaine-seeking maintained by a drug-paired conditioned stimulus in rats. Eur J Neurosci 2004; 19: 1661–1667.
Pontieri FE, Tanda G, Orzi F, Di Chiara G . Effects of nicotine on the nucleus accumbens and similarity to those of addictive drugs. Nature 1996; 382: 255–257.
Tolu S, Eddine R, Marti F, David V, Graupner M, Pons S et al. Co-activation of VTA DA and GABA neurons mediates nicotine reinforcement. Mol Psychiatry 2012; 18: 382–393.
Salas R, Orr-Urtreger A, Broide RS, Beaudet A, Paylor R, De Biasi M . The nicotinic acetylcholine receptor subunit alpha 5 mediates short-term effects of nicotine in vivo. Mol Pharmacol 2003; 63: 1059–1066.
Martellotta MC, Kuzmin A, Zvartau E, Cossu G, Gessa GL, Fratta W . Isradipine inhibits nicotine intravenous self-administration in drug-naive mice. Pharmacol Biochem Behav 1995; 52: 271–274.
Pons S, Fattore L, Cossu G, Tolu S, Porcu E, McIntosh JM et al. Crucial role of alpha4 and alpha6 nicotinic acetylcholine receptor subunits from ventral tegmental area in systemic nicotine self-administration. J Neurosci 2008; 28: 12318–12327.
Marti F, Arib O, Morel C, Dufresne V, Maskos U, Corringer P-J et al. Smoke extracts and nicotine, but not tobacco extracts, potentiate firing and burst activity of ventral tegmental area dopaminergic neurons in mice. Neuropsychopharmacology 2011; 36: 2244–2257.
Exley R, Maubourguet N, David V, Eddine R, Evrard A, Pons S et al. Distinct contributions of nicotinic acetylcholine receptor subunit alpha4 and subunit alpha6 to the reinforcing effects of nicotine. Proc Natl Acad Sci USA 2011; 108: 7577–7582.
Naldini L, Blömer U, Gallay P, Ory D, Mulligan R, Gage FH et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272: 263–267.
Maskos U, Molles BE, Pons S, Besson M, Guiard BP, Guilloux J-P et al. Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature 2005; 436: 103–107.
Tolu S, Avale ME, Nakatani H, Pons S, Parnaudeau S, Tronche F et al. A versatile system for the neuronal subtype specific expression of lentiviral vectors. FASEB J 2010; 24: 723–730.
Grace AA, Bunney BS . The control of firing pattern in nigral dopamine neurons: burst firing. J Neurosci 1984; 4: 2877–2890.
Grace AA, Bunney BS . The control of firing pattern in nigral dopamine neurons: single spike firing. J Neurosci 1984; 4: 2866–2876.
Benowitz NL . Pharmacology of nicotine: addiction, smoking-induced disease, and therapeutics. Annu Rev Pharmacol Toxicol 2008; 49: 57–71.
Albuquerque EX, Pereira EFR, Alkondon M, Rogers SW . Mammalian nicotinic acetylcholine receptors: from structure to function. Physiol Rev 2009; 89: 73–120.
Kuryatov A, Berrettini W, Lindstrom J . Acetylcholine receptor (AChR)Alpha5 subunit variant associated with risk for nicotine dependence and lung cancer reduces (alpha4beta2)2alpha5 AChR function. Mol Pharmacol 2010; 79: 119–125.
Jackson KJ, Martin BR, Changeux JP, Damaj MI . Differential role of nicotinic acetylcholine receptor subunits in physical and affective nicotine withdrawal signs. J Pharmacol Exp Ther 2008; 325: 302–312.
Jackson KJ, Marks MJ, Vann RE, Chen X, Gamage TF, Warner JA et al. The role of alpha5 nicotinic acetylcholine receptors in the pharmacological and behavioral effects of nicotine in mice. J Pharmacol Exp Ther 2010; 334: 137–146.
Mameli-Engvall M, Evrard A, Pons S, Maskos U, Svensson TH, Changeux J-P et al. Hierarchical control of dopamine neuron-firing patterns by nicotinic receptors. Neuron 2006; 50: 911–921.
Tsuneki H, Klink R, Léna C, Korn H, Changeux JP . Calcium mobilization elicited by two types of nicotinic acetylcholine receptors in mouse substantia nigra pars compacta. Eur J Neurosci 2000; 12: 2475–2485.
Ramirez-Latorre J, Yu CR, Qu X, Perin F, Karlin A, Role L . Functional contributions of alpha5 subunit to neuronal acetylcholine receptor channels. Nature 1996; 380: 347–351.
Picciotto MR, Zoli M, Rimondini R, Léna C, Marubio LM, Pich EM et al. Acetylcholine receptors containing the beta2 subunit are involved in the reinforcing properties of nicotine. Nature 1998; 391: 173–177.
Fowler CD, Tuesta L, Kenny PJ . Role of α5* nicotinic acetylcholine receptors in the effects of acute and chronic nicotine treatment on brain reward function in mice. Psychopharmacology 2013; 229: 503–513.
Frahm S, Slimak MA, Ferrarese L, Santos-Torres J, Antolin-Fontes B, Auer S et al. Aversion to Nicotine Is Regulated by the Balanced Activity of beta4 and alpha5 Nicotinic Receptor Subunits in the Medial Habenula. Neuron 2011; 70: 522–535.
Scholze P, Koth G, Orr-Urtreger A, Huck S . Subunit composition of α5-containing nicotinic receptors in the rodent habenula. J Neurochem 2012; 121: 551–560.
Exley R, McIntosh JM, Marks MJ, Maskos U, Cragg SJ . Striatal α5 nicotinic receptor subunit regulates dopamine transmission in dorsal striatum. J Neurosci 2012; 32: 2352–2356.
Kravitz AV, Tye LD, Kreitzer AC . Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nat Neurosci 2012; 15: 816–818.
Kitai S, Shepard P, Callaway J, Scroggs R . Afferent modulation of dopamine neuron firing patterns. Curr Opin Neurobiol 1999; 9: 1–8.
Mansvelder HD, McGehee DS . Long-term potentiation of excitatory inputs to brain reward areas by nicotine. Neuron 2000; 27: 349–357.
Acknowledgements
We would like to thank Stefania Tolu for helpful comments on the manuscript. This work was supported by the Institut Pasteur, Centre National de la Recherche Scientifique CNRS UMR 3571, UMR 7102 and ATIP programme, the Agence Nationale pour la Recherche (ANR Neuroscience, Neurologie et Psychiatrie 2009, and ANR BLANC 2012), la Fondation pour la Recherche Médicale (FRM, équipe 2013 PF), fondation pour la the Neuropole de Recherche Francilien (NeRF) of Ile de France, the Bettencourt Schueller Foundation, National Cancer Institute INCa BIO-SILC programme, Ecole des Neurosciences de Paris (ENP), FP7 ERANET Neuron NICO-GENE network, LabEx GENMED funded by ANR, and NIH grants DA029157 and U19CA148127. This work was supported by the Department of Biomedical Sciences, Division of Neuroscience and Clinical Pharmacology, University of Cagliari, Italy. The laboratories of Philippe Faure, Uwe Maskos and Bertrand Lambolez are part of the École des Neurosciences de Paris Ile-de-France RTRA network. PF and UM are members of the Laboratory of Excellence, LabEx Bio-Psy.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Morel, C., Fattore, L., Pons, S. et al. Nicotine consumption is regulated by a human polymorphism in dopamine neurons. Mol Psychiatry 19, 930–936 (2014). https://doi.org/10.1038/mp.2013.158
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2013.158
Keywords
This article is cited by
-
Midbrain projection to the basolateral amygdala encodes anxiety-like but not depression-like behaviors
Nature Communications (2022)
-
An innate contribution of human nicotinic receptor polymorphisms to COPD-like lesions
Nature Communications (2021)
-
Concatemers to re-investigate the role of α5 in α4β2 nicotinic receptors
Cellular and Molecular Life Sciences (2021)
-
The Intergenerational Transmission of Developmental Nicotine Exposure-Induced Neurodevelopmental Disorder-Like Phenotypes is Modulated by the Chrna5 D397N Polymorphism in Adolescent Mice
Behavior Genetics (2021)
-
Profound alteration in reward processing due to a human polymorphism in CHRNA5: a role in alcohol dependence and feeding behavior
Neuropsychopharmacology (2019)