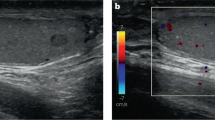
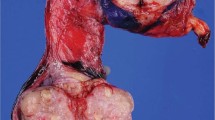
Abstract
Organ-sparing approaches are currently practiced in urology for many malignancies. Partial orchiectomy of germ cell tumors (GCT) provides potential benefits over radical surgery by reducing the need for androgen substitution, lessening psychological stress, and preserving fertility, with a durable cure rate. Furthermore, many testicular lesions detected clinically or by ultrasonography will be benign, in which case radical orchiectomy represents overtreatment. Partial orchiectomy for benign lesions allows preservation of endocrine and exocrine function, and reduced risk of local recurrence. However, selection criteria are not clear and one must always be suspicious that a GCT might exist. Carcinoma in situ that remains in the salvaged testicle is a challenge to treat. Radiation therapy is an option, although there is a high chance that patients will subsequently require hormonal replacement. Partial orchiectomy should be undertaken only in selected patients—men with bilateral testicular cancer or GCT in a solitary testis—if the size and location of the mass are amenable to surgery. Informed patient consent discussing radical orchiectomy as the gold standard is mandatory, and discussion of the risks associated with CIS and its treatment, as well as the need for androgen supplementation are paramount. Alternative strategies of organ preservation, such as radiotherapy, HIFU and chemotherapy, might be appropriate treatment options in the future. However, the safety and efficacy of these procedures needs to be demonstrated in comparison with partial orchiectomy in larger and prospective studies with longer follow-up.
Key Points
-
Partial orchiectomy of germ cell tumors (GCT) provides potential benefits over radical surgery by reducing the need for androgen substitution, lessening psychological stress, and preserving fertility, with a durable cure rate
-
Partial orchiectomy of benign testicular lesions reduces the proportion of patients overtreated with radical orchiectomy
-
Partial orchiectomy should be undertaken only in selected cases (bilateral testicular cancer or tumor in a solitary testis) at centers with sufficient experience of testicular cancer
-
Informed patient consent is mandatory, including discussion of radical orchiectomy as the gold standard, explanation of carcinoma in situ (CIS) and potential adjuvant treatment, and the possible need for androgen supplementation
-
Adjuvant radiation is not mandatory for patients with CIS but fertility issues and anxiety should be accounted for, along with a willingness to accept self-examination and imaging follow-up
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Heidenreich, A., Bonfig, R., Derschum, W., von Vietsch, H. & Wilbert, D. M. A conservative approach to bilateral testicular germ cell tumors. J. Urol. 153, 10–13 (1995).
Soh, E., Berman, L. H., Grant, J. W., Bullock, N. & Williams, M. V. Ultrasound-guided core-needle biopsy of the testis for focal indeterminate intratesticular lesions. Eur. Radiol. 18, 2990–2996 (2008).
Rogers, D. A. et al. Indications for hemiscrotectomy in the management of genitourinary tumors in children. J. Pediatr. Surg. 30, 1437–1439 (1995).
Heidenreich, A., Engelmann, U. H., Vietsch, H. V. & Derschum, W. Organ preserving surgery in testicular epidermoid cysts. J. Urol. 153, 1147–1150 (1995).
Haas, G. P., Shumaker, B. P. & Cerny, J. C. The high incidence of benign testicular tumors. J. Urol. 136, 1219–1220 (1986).
Kressel, K. et al. Benign testicular tumors: a case for testis preservation? Eur. Urol. 15, 200–204 (1988).
Carmignani, L. et al. High incidence of benign testicular neoplasms diagnosed by ultrasound. J. Urol. 170, 1783–1786 (2003).
Carmignani, L. et al. Prognostic parameters in adult impalpable ultrasonographic lesions of the testicle. J. Urol. 174, 1035–1038 (2005).
Richie, J. P. Detection and treatment of testicular cancer. CA Cancer J. Clin. 43, 151–175 (1993).
Patel, S. R., Richardson, R. L. & Kvols, L. Synchronous and metachronous bilateral testicular tumors. Mayo Clinic experience. Cancer 65, 1–4 (1990).
Pharris-Ciurej, N. D., Cook, L. S. & Weiss, N. S. Incidence of testicular cancer in the United States: has the epidemic begun to abate? Am. J. Epidemiol. 150, 45–46 (1999).
Jemal, A. et al. Cancer statistics, 2009. CA Cancer J. Clin. 59, 225–249 (2009).
Krege, S. et al. European consensus conference on diagnosis and treatment of germ cell cancer: a report of the second meeting of the European Germ Cell Cancer Consensus group (EGCCCG): part I. Eur. Urol. 53, 478–496 (2008).
International Germ Cell Consensus Classification: a prognostic factor-based staging system for metastatic germ cell cancers. International Germ Cell Cancer Collaborative Group. J. Clin. Oncol. 15, 594–603 (1997).
Zuniga, A., Kakiashvili, D. & Jewett, M. A. Surveillance in stage I nonseminomatous germ cell tumours of the testis. BJU Int. 104, 1351–1356 (2009).
Large, M. C., Sheinfeld, J. & Eggener, S. E. Retroperitoneal lymph node dissection: reassessment of modified templates. BJU Int. 104, 1369–1375 (2009).
Lawrentschuk, N. & Webb, D. R. Inserting testicular prostheses: a new surgical technique for difficult cases. BJU Int. 95, 1111–1114 (2005).
Bang, A. K. et al. Testosterone production is better preserved after 16 than 20 Gray irradiation treatment against testicular carcinoma in situ cells. Int. J. Radiat. Oncol. Biol. Phys. 75, 672–676 (2009).
Toren, P. J. et al. Small incidentally discovered testicular masses in infertile men—is active surveillance the new standard of care? J. Urol. 183, 1373–1377 (2010).
Carkaci, S., Ozkan, E., Lane, D. & Yang, W. T. Scrotal sonography revisited. J. Clin. Ultrasound 38, 21–37 (2010).
Kirkham, A. P. et al. Targeted testicular excision biopsy: when and how should we try to avoid radical orchidectomy? Clin. Radiol. 64, 1158–1165 (2009).
Dogra, V. S., Gottlieb, R. H., Rubens, D. J. & Liao, L. Benign intratesticular cystic lesions: US features. Radiographics 21 (Spec. No), S273–S281 (2001).
Cho, J. H., Chang, J. C., Park, B. H., Lee, J. G. & Son, C. H. Sonographic and MR imaging findings of testicular epidermoid cysts. AJR Am. J. Roentgenol. 178, 743–748 (2002).
DeCastro, B. J., Peterson, A. C. & Costabile, R. A. A 5-year followup study of asymptomatic men with testicular microlithiasis. J. Urol. 179, 1420–1423 (2008).
Parenti, G. C., Zago, S., Lusa, M., Campioni, P. & Mannella, P. Association between testicular microlithiasis and primary malignancy of the testis: our experience and review of the literature. Radiol. Med. 112, 588–596 (2007).
Parenti, G. C. et al. Imaging of the scrotum: role of MRI. Radiol. Med. 114, 414–424 (2009).
Shannon, B. A., Cohen, R. J., de Bruto, H. & Davies, R. J. The value of preoperative needle core biopsy for diagnosing benign lesions among small, incidentally detected renal masses. J. Urol. 180, 1257–1261 (2008).
Capelouto, C. C., Clark, P. E., Ransil, B. J. & Loughlin, K. R. A review of scrotal violation in testicular cancer: is adjuvant local therapy necessary? J. Urol. 153, 981–985 (1995).
Giguere, J. K. et al. The clinical significance of unconventional orchiectomy approaches in testicular cancer: a report from the Testicular Cancer Intergroup Study. J. Urol. 139, 1225–1228 (1988).
Heidenreich, A. Contralateral testicular biopsy in testis cancer: current concepts and controversies. BJU Int. 104, 1346–1350 (2009).
Steiner, H. et al. Frozen section analysis-guided organ-sparing approach in testicular tumors: technique, feasibility, and long-term results. Urology 62, 508–513 (2003).
Elert, A. et al. Accuracy of frozen section examination of testicular tumors of uncertain origin. Eur. Urol. 41, 290–293 (2002).
Tokuc, R., Sakr, W., Pontes, J. E. & Haas, G. P. Accuracy of frozen section examination of testicular tumors. Urology 40, 512–516 (1992).
Hopps, C. V. & Goldstein, M. Ultrasound guided needle localization and microsurgical exploration for incidental nonpalpable testicular tumors. J. Urol. 168, 1084–1087 (2002).
Powell, T. M. & Tarter, T. H. Management of nonpalpable incidental testicular masses. J. Urol. 176, 96–98 (2006).
Berney, D. M. Staging and classification of testicular tumours: pitfalls from macroscopy to diagnosis. J. Clin. Pathol. 61, 20–24 (2008).
Chung, P. W., Jewett, M. A. & Warde, P. R. Testicular radiation for primary seminoma in a solitary testis. Can. J. Urol. 13, 2975–2977 (2006).
Kratzik, C., Schatzl, G., Lackner, J. & Marberger, M. Transcutaneous high-intensity focused ultrasonography can cure testicular cancer in solitary testis. Urology 67, 1269–1273 (2006).
Rolle, L. et al. Microsurgical “testis-sparing” surgery for nonpalpable hypoechoic testicular lesions. Urology 68, 381–385 (2006).
Billmire, D. F. Germ cell tumors. Surg. Clin. North Am. 86, 489–503 (2006).
Corrie, D., Mueller, E. J. & Thompson, I. M. Management of ultrasonically detected nonpalpable testis masses. Urology 38, 429–431 (1991).
Horstman, W. G., Haluszka, M. M. & Burkhard, T. K. Management of testicular masses incidentally discovered by ultrasound. J. Urol. 151, 1263–1265 (1994).
Connolly, S. S. et al. Carefully selected intratesticular lesions can be safely managed with serial ultrasonography. BJU Int. 98, 1005–1007 (2006).
Müller, T. et al. Management of incidental impalpable intratesticular masses of < or = 5 mm in diameter. BJU Int. 98, 1001–1004 (2006).
Leroy, X., Rigot, J. M., Aubert, S., Ballereau, C. & Gosselin, B. Value of frozen section examination for the management of nonpalpable incidental testicular tumors. Eur. Urol. 44, 458–460 (2003).
Hindley, R. G., Chandra, A., Saunders, A. & O'Brien, T. S. Impalpable testis cancer. BJU Int. 92, 572–574 (2003).
Comiter, C. V. et al. Nonpalpable intratesticular masses detected sonographically. J. Urol. 154, 1367–1369 (1995).
Ishida, M., Hasegawa, M., Kanao, K., Oyama, M. & Nakajima, Y. Non-palpable testicular embryonal carcinoma diagnosed by ultrasound: a case report. Jpn J. Clin. Oncol. 39, 124–126 (2009).
Passman, C. et al. Testicular lesions other than germ cell tumours: feasibility of testis-sparing surgery. BJU Int. 103, 488–491 (2009).
Giannarini, G. et al. Long-term followup after elective testis sparing surgery for Leydig cell tumors: a single center experience. J. Urol. 178, 872–876 (2007).
Loeser, A. et al. Testis-sparing surgery versus radical orchiectomy in patients with Leydig cell tumors. Urology 74, 370–372 (2009).
Jou, P. & Maclennan, G. T. Leydig cell tumor of the testis. J. Urol. 181, 2299–2300 (2009).
Philip, P. P. Feminizing tumours of the testis. Postgrad. Med. J. 36, 190–196 (1960).
Dieckmann, K .P. & Skakkebaek, N. E. Carcinoma in situ of the testis: review of biological and clinical features. Int. J. Cancer 83, 815–822 (1999).
Heidenreich, A. et al. Organ. sparing surgery for malignant germ cell tumor of the testis. J. Urol. 166, 2161–2165 (2001).
Hoei-Hansen, C. E., Rajpert-De Meyts, E., Daugaard, G. & Skakkebaek, N. E. Carcinoma in situ testis, the progenitor of testicular germ cell tumours: a clinical review. Ann. Oncol. 16, 863–868 (2005).
Krege, S. et al. European consensus conference on diagnosis and treatment of germ cell cancer: a report of the second meeting of the European Germ Cell Cancer Consensus Group (EGCCCG): part II. Eur. Urol. 53, 497–513 (2008).
Dieckmann, K. P., Kulejewski, M., Pichlmeier, U. & Loy, V. Diagnosis of contralateral testicular intraepithelial neoplasia (TIN) in patients with testicular germ cell cancer: systematic two-site biopsies are more sensitive than a single random biopsy. Eur. Urol. 51, 175–183 (2007).
Berthelsen, J. G. & Skakkebaek, N. E. Gonadal function in men with testis cancer. Fertil. Steril. 39, 68–75 (1983).
Dieckmann, K. P., Linke, J., Pichlmeier, U., Kulejewski, M. & Loy, V. Spermatogenesis in the contralateral testis of patients with testicular germ cell cancer: histological evaluation of testicular biopsies and a comparison with healthy males. BJU Int. 99, 1079–1085 (2007).
Hoei-Hansen, C. E., Holm, M., Rajpert- De Meyts, E. & Skakkebaek, N. E. Histological evidence of testicular dysgenesis in contralateral biopsies from 218 patients with testicular germ cell cancer. J. Pathol. 200, 370–374 (2003).
Weissbach, L. Organ preserving surgery of malignant germ cell tumors. J. Urol. 153, 90–93 (1995).
Albers, P. Management of stage I testis cancer. Eur. Urol. 51, 34–43 (2007).
Petersen, P. M. et al. Impaired testicular function in patients with carcinoma-in-situ of the testis. J. Clin. Oncol. 17, 173–179 (1999).
Dieckmann, K. P., Besserer, A. & Loy, V. Low-dose radiation therapy for testicular intraepithelial neoplasia. J. Cancer Res. Clin. Oncol. 119, 355–359 (1993).
Dieckmann, K. P. & Loy, V. The value of the biopsy of the contralateral testis in patients with testicular germ cell cancer: the recent German experience. APMIS 106, 13–20 (1998).
Heidenreich, A., Vorreuther, R., Neubauer, S., Zumbe, J. & Engelmann, U. H. Paternity in patients with bilateral testicular germ cell tumors. Eur. Urol. 31, 2468 (1997).
Petersen, P. M., Giwercman, A., Skakkebaek, N. E. & Rorth, M. Gonadal function in men with testicular cancer. Semin. Oncol. 25, 224–233 (1998).
Giwercman, A. et al. Localized irradiation of testes with carcinoma in situ: effects on Leydig cell function and eradication of malignant germ cells in 20 patients. J. Clin. Endocrinol. Metab. 73, 596–603 (1991).
Petersen, P. M., Daugaard, G., Rorth, M. & Skakkebaek, N. E. Endocrine function in patients treated for carcinoma in situ in the testis with irradiation. APMIS 111, 93–98 (2003).
Petersen, P. M. et al. Effect of graded testicular doses of radiotherapy in patients treated for carcinoma-in-situ in the testis. J. Clin. Oncol. 20, 1537–1543 (2002).
Grigsby, P. W. & Perez, C. A. The effects of external beam radiotherapy on endocrine function in patients with carcinoma of the prostate. J. Urol. 135, 726–727 (1986).
Nader, S., Schultz, P. N., Cundiff, J. H., Hussey, D. H. & Samaan, N. A. Endocrine profiles of patients with testicular tumors treated with radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 9, 1723–1726 (1983).
Shalet, S. M., Tsatsoulis, A., Whitehead, E. & Read, G. Vulnerability of the human Leydig cell to radiation damage is dependent upon age. J. Endocrinol. 120, 161–165 (1989).
Ehrlich, Y., Konichezky, M., Yossepowitch, O. & Baniel, J. Multifocality in testicular germ cell tumors. J. Urol. 181, 1114–1119 (2009).
Stoehr, B. et al. Routine scrotal ultrasonography during the follow-up of patients with testicular cancer leads to earlier detection of asynchronous tumours and a high rate of organ preservation. BJU Int. 105, 1118–1120 (2010).
Montie, J. E. Carcinoma in situ of the testis and bilateral carcinoma. Urol. Clin. North Am. 20, 127–132 (1993).
Che, M. et al. Bilateral testicular germ cell tumors: twenty-year experience at M. D. Anderson Cancer Center. Cancer 95, 1228–1233 (2002).
Colls, B. M., Harvey, V. J., Skelton, L., Thompson, P. I. & Frampton, C. M. Bilateral germ cell testicular tumors in New Zealand: experience in Auckland and Christchurch 1978–1994. J. Clin. Oncol. 14, 2061–2065 (1996).
Osterlind, A. et al. Risk of bilateral testicular germ cell cancer in Denmark: 1960–1984. J. Natl Cancer Inst. 83, 1391–1395 (1991).
Fossa, S. D., Opjordsmoen, S. & Haug, E. Androgen replacement and quality of life in patients treated for bilateral testicular cancer. Eur. J. Cancer 35, 1220–1225 (1999).
Holzbeierlein, J. M., Sogani, P. C. & Sheinfeld, J. Histology and clinical outcomes in patients with bilateral testicular germ cell tumors: the Memorial Sloan Kettering Cancer Center experience 1950 to 2001. J. Urol. 169, 2122–2125 (2003).
Tekin, A., Aygun, Y. C., Aki, F. T. & Ozen, H. Bilateral germ cell cancer of the testis: a report of 11 patients with a long-term follow-up. BJU Int. 85, 864–868 (2000).
Heidenreich, A., Höltl, W., Albrecht, W., Pont, J. & Engelmann, U. H. Testis-preserving surgery in bilateral testicular germ cell tumours. Br. J. Urol. 79, 253–257 (1997).
Houlgatte, A. et al. Paternity in a patient with seminoma and carcinoma in situ in a solitary testis treated by partial orchidectomy. BJU Int. 84, 374–375 (1999).
Kazem, I. & Danella, J. F. Organ preservation for the treatment of contralateral testicular seminoma. Radiother. Oncol. 53, 45–47 (1999).
Sawyer, E. J., Oliver, R. T., Tobias, J. S. & Badenoch, D. F. A lesson in the management of testicular cancer in a patient with a solitary testis. Postgrad. Med. J. 75, 481–483 (1999).
Bos, S. D. & Ypma, A. F. Synchronous bilateral seminoma testis treated with unilateral orchiectomy and contralateral irradiation: a therapeutic option. Scand. J. Urol. Nephrol. 27, 559–561 (1993).
Izard, M. A. Leydig cell function and radiation: a review of the literature. Radiother. Oncol. 34, 1–8 (1995).
Tomita, E. et al. Successful testis preservation for bilateral testicular tumors with a new chemotherapy-based protocol: initial results of three cases. Int. J. Urol. 14, 879–882 (2007).
Wang, X. & Sun, J. High-intensity focused ultrasound in patients with late-stage pancreatic carcinoma. Chin. Med. J. (Engl.) 115, 1332–1335 (2002).
Blana, A., Walter, B., Rogenhofer, S. & Wieland, W. F. High-intensity focused ultrasound for the treatment of localized prostate cancer: 5-year experience. Urology 63, 297–300 (2004).
Jun-Qun, Z., Guo-Min., W., Bo, Y., Gong-Xian, W. & Shen-Xu, H. Short-term results of 89 cases of rectal carcinoma treated with high-intensity focused ultrasound and low-dose radiotherapy. Ultrasound Med. Biol. 30, 57–60 (2004).
Li, C. X. et al. Analysis of clinical effect of high-intensity focused ultrasound on liver cancer. World J. Gastroenterol. 10, 2201–2204 (2004).
Uchida, T. et al. Five years experience of transrectal high-intensity focused ultrasound using the Sonablate device in the treatment of localized prostate cancer. Int. J. Urol. 13, 228–233 (2006).
Wu, F. et al. A randomised clinical trial of high-intensity focused ultrasound ablation for the treatment of patients with localised breast cancer. Br. J. Cancer 89, 2227–2233 (2003).
Wu, F. et al. Extracorporeal high intensity focused ultrasound ablation in the treatment of patients with large hepatocellular carcinoma. Ann. Surg. Oncol. 11, 1061–1069 (2004).
Acknowledgements
We acknowledge the contribution of Janice Yau, a Biomedical Communications student at the University of Toronto, Toronto, Canada for creating the original artwork for Figure 2.
Charles P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Zuniga, A., Lawrentschuk, N. & Jewett, M. Organ-sparing approaches for testicular masses. Nat Rev Urol 7, 454–464 (2010). https://doi.org/10.1038/nrurol.2010.100
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2010.100
This article is cited by
-
Diagnosis and management of indeterminate testicular lesions
Nature Reviews Urology (2024)
-
Organ-sparing surgery is the treatment of choice in benign testicular tumors
World Journal of Urology (2014)
-
Carcinoma in situ des Hodens
Der Pathologe (2012)
-
Carcinoma in situ des Hodens
Der Pathologe (2011)