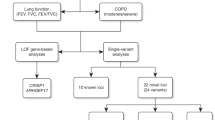
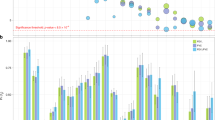
Abstract
Reduced lung function predicts mortality and is key to the diagnosis of chronic obstructive pulmonary disease (COPD). In a genome-wide association study in 400,102 individuals of European ancestry, we define 279 lung function signals, 139 of which are new. In combination, these variants strongly predict COPD in independent populations. Furthermore, the combined effect of these variants showed generalizability across smokers and never smokers, and across ancestral groups. We highlight biological pathways, known and potential drug targets for COPD and, in phenome-wide association studies, autoimmune-related and other pleiotropic effects of lung function–associated variants. This new genetic evidence has potential to improve future preventive and therapeutic strategies for COPD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
SpiroMeta GWAS summary statistics and UK Biobank GWAS summary statistics are available online via LD-Hub (http://ldsc.broadinstitute.org/ldhub/). Single-variant PheWAS results are available by request to the corresponding authors. The newly derived spirometry variables are available from UK Biobank (http://www.ukbiobank.ac.uk/).
Change history
19 April 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41588-024-01752-4
References
Young, R. P., Hopkins, R. & Eaton, T. E. Forced expiratory volume in one second: not just a lung function test but a marker of premature death from all causes. Eur. Respir. J. 30, 616–622 (2007).
GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390, 1151–1210 (2017).
GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390, 1211–1259 (2017).
Hobbs, B. D. et al. Genetic loci associated with chronic obstructive pulmonary disease overlap with loci for lung function and pulmonary fibrosis. Nat. Genet. 49, 426–432 (2017).
Salvi, S. S. & Barnes, P. J. Chronic obstructive pulmonary disease in non-smokers. Lancet 374, 733–743 (2009).
Nelson, M. R. et al. The support of human genetic evidence for approved drug indications. Nat. Genet. 47, 856–860 (2015).
Wilk, J. B. et al. A genome-wide association study of pulmonary function measures in the Framingham Heart Study. PLoS Genet. 5, e1000429 (2009).
Repapi, E. et al. Genome-wide association study identifies five loci associated with lung function. Nat. Genet. 42, 36–44 (2010).
Hancock, D. B. et al. Meta-analyses of genome-wide association studies identify multiple loci associated with pulmonary function. Nat. Genet. 42, 45–52 (2010).
Soler Artigas, M. et al. Genome-wide association and large-scale follow up identifies 16 new loci influencing lung function. Nat. Genet. 43, 1082–1090 (2011).
Cho, M. H. et al. A genome-wide association study of COPD identifies a susceptibility locus on chromosome 19q13. Hum. Mol. Genet. 21, 947–957 (2012).
Loth, D. W. et al. Genome-wide association analysis identifies six new loci associated with forced vital capacity. Nat. Genet. 46, 669–677 (2014).
Wain, L. V. et al. Novel insights into the genetics of smoking behaviour, lung function, and chronic obstructive pulmonary disease (UK BiLEVE): a genetic association study in UK Biobank. Lancet Respir. Med. 3, 769–781 (2015).
Lutz, S. M. et al. A genome-wide association study identifies risk loci for spirometric measures among smokers of European and African ancestry. BMC Genet. 16, 138 (2015).
Soler Artigas, M. et al. Sixteen new lung function signals identified through 1000 Genomes Project reference panel imputation. Nat. Commun. 6, 8658 (2015).
Hobbs, B. D. et al. Exome array analysis identifies a common variant in IL27 associated with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 194, 48–57 (2016).
Jackson, V. et al. Meta-analysis of exome array data identifies six novel genetic loci for lung function [version 3; referees: 2 approved]. Wellcome Open Res. 3, 4 (2018).
Wain, L. V. et al. Genome-wide association analyses for lung function and chronic obstructive pulmonary disease identify new loci and potential druggable targets. Nat. Genet. 49, 416–425 (2017).
Wyss, A. B. et al. Multiethnic meta-analysis identifies new loci for pulmonary function. Nat. Commun. 9, 2976 (2017).
Loh, P. R. et al. Efficient Bayesian mixed-model analysis increases association power in large cohorts. Nat. Genet. 47, 284–290 (2015).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291 (2015).
Yang, J. et al. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nat. Genet. 44, 369–375 (2012).
Pulit, S. L., de With, S. A. & de Bakker, P. I. Resetting the bar: Statistical significance in whole-genome sequencing-based association studies of global populations. Genet. Epidemiol. 41, 145–151 (2017).
Palmer, L. J. et al. Familial aggregation and heritability of adult lung function: results from the Busselton Health Study. Eur. Respir. J. 17, 696–702 (2001).
Wilk, J. B. et al. Evidence for major genes influencing pulmonary function in the NHLBI family heart study. Genet. Epidemiol. 19, 81–94 (2000).
Benyamin, B. et al. GWAS of butyrylcholinesterase activity identifies four novel loci, independent effects within BCHE and secondary associations with metabolic risk factors. Hum. Mol. Genet. 20, 4504–4514 (2011).
Hammarsjo, A., Wang, Z., Vaz, R. & Taylan, F. Novel KIAA0753 mutations extend the phenotype of skeletal ciliopathies. Sci Rep. 7, 15585 (2017).
Stephen, J. et al. Mutations in KIAA0753 cause Joubert syndrome associated with growth hormone deficiency. Hum. Genet. 136, 399–408 (2017).
Loukil, A., Tormanen, K. & Sütterlin, C. The daughter centriole controls ciliogenesis by regulating Neurl-4 localization at the centrosome. J. Cell Biol. 216, 1287–1300 (2017).
He, R. et al. LRRC45 is a centrosome linker component required for centrosome cohesion. Cell Rep. 4, 1100–1107 (2013).
Conkar, D. et al. The centriolar satellite protein CCDC66 interacts with CEP290 and functions in cilium formation and trafficking. J Cell Sci. 130, 1450–1462 (2017).
Uhlén, M. et al. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Hao, K. et al. Lung eQTLs to help reveal the molecular underpinnings of asthma. PLoS Genet. 8, e1003029 (2012).
Lamontagne, M. et al. Refining susceptibility loci of chronic obstructive pulmonary disease with lung eqtls. PLoS One 8, e70220 (2013).
Obeidat, M. et al. GSTCD and INTS12 regulation and expression in the human lung. PLoS One 8, e74630 (2013).
Westra, H. J. et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat. Genet. 45, 1238–1243 (2013).
GTEx Consortium. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Kundaje, A. et al. Integrative analysis of 111 reference human epigenomes. Nature 518, 317–330 (2015).
Sun, B. B. et al. Genomic atlas of the human plasma proteome. Nature 558, 73–79 (2018).
Finucane, H. K. et al. Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat. Genet. 47, 1228–1235 (2015).
Iotchkova, V. et al. Discovery and refinement of genetic loci associated with cardiometabolic risk using dense imputation maps. Nat. Genet. 48, 1303–1312 (2016).
Zhou, J. & Troyanskaya, O. G. Predicting effects of noncoding variants with deep learning-based sequence model. Nat. Meth. 12, 931–934 (2015).
Cotto, K. C. et al. DGIdb 3.0: a redesign and expansion of the drug–gene interaction database. Nucleic Acids Res. 46, D1068–D1073 (2017).
Slack, R. et al. P112 discovery of a novel, high affinity, small molecule αvβ6 inhibitor for the treatment of idiopathic pulmonary fibrosis. QJM 109, S60–S60 (2016).
Raab-Westphal, S., Marshall, J. F. & Goodman, S. L. Integrins as therapeutic targets: successes and cancers. Cancers (Basel) 9, (e110 (2017).
Merrill, J. T. et al. Efficacy and safety of atacicept in patients with systemic lupus erythematosus: results of a twenty-four-week, multicenter, randomized, double-blind, placebo-controlled, parallel-arm, phase IIb study. Arthritis Rheumatol. 70, 266–276 (2018).
Aschard, H. et al. Evidence for large-scale gene-by-smoking interaction effects on pulmonary function. Int. J. Epidemiol. 46, 894–904 (2017).
Lokke, A., Lange, P., Scharling, H., Fabricius, P. & Vestbo, J. Developing COPD: a 25 year follow up study of the general population. Thorax 61, 935–939 (2006).
Pulley, J. M. et al. Accelerating precision drug development and drug repurposing by leveraging human genetics. Assay. Drug. Dev. Technol. 15, 113–119 (2017).
Yengo, L. et al. Meta-analysis of genome-wide association studies for height and body mass index in ~700,000 individuals of European ancestry. Hum. Mol. Genet. 27, 3641–3649 (2018).
Wain, L. V. et al. Whole exome re-sequencing implicates CCDC38 and cilia structure and function in resistance to smoking related airflow obstruction. PLoS Genet. 10, e1004314 (2014).
Black, P. N. et al. Changes in elastic fibres in the small airways and alveoli in COPD. Eur. Respir. J. 31, 998–1004 (2008).
Martinez, F. J. et al. A new approach for identifying patients with undiagnosed chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care. Med. 195, 748–756 (2017).
Strug, L. J. et al. Cystic fibrosis gene modifier SLC26A9 modulates airway response to CFTR-directed therapeutics. Hum. Mol. Genet. 25, 4590–4600 (2016).
The 1000 Genomes Project Consortium. A map of human genome variation from population-scale sequencing. Nature 467, 1061 (2010).
Bycroft, C. et al. Genome-wide genetic data on ~500,000 UK Biobank participants. Preprint at https://www.biorxiv.org/content/early/2017/07/20/166298 (2017).
McCarthy, S. et al. A reference panel of 64,976 haplotypes for genotype imputation. Nat. Genet. 48, 1279–1283 (2016).
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011).
Wakefield, J. Reporting and interpretation in genome-wide association studies. Int. J. Epidemiol. 37, 641–653 (2008).
van de Bunt, M., Cortes, A., Brown, M. A., Morris, A. P. & McCarthy, M. I. Evaluating the performance of fine-mapping strategies at common variant GWAS loci. PLoS Genet. 11, e1005535 (2015).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
Shihab, H. A. et al. Predicting the functional, molecular, and phenotypic consequences of amino acid substitutions using hidden Markov models. Hum. Mutat. 34, 57–65 (2013).
Jansen, R. et al. Conditional eQTL analysis reveals allelic heterogeneity of gene expression. Hum. Mol. Genet. 26, 1444–1451 (2017).
Battle, A., Brown, C. D., Engelhardt, B. E. & Montgomery, S. B. Genetic effects on gene expression across human tissues. Nature 550, 204–213 (2017).
Kamburov, A., Stelzl, U., Lehrach, H. & Herwig, R. The ConsensusPathDB interaction database: 2013 update. Nucleic Acids Res. 41, D793–D800 (2013).
MacArthur, J. et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 45, D896–D901 (2017).
Leslie, R., O’Donnell, C. J. & Johnson, A. D. GRASP: analysis of genotype-phenotype results from 1390 genome-wide association studies and corresponding open access database. Bioinformatics 30, i185–i194 (2014).
Acknowledgements
This research has been conducted using the UK Biobank Resource under applications 648, 4892 and 26041. L. Wain holds a GSK/British Lung Foundation Chair in Respiratory Research. M. Tobin is supported by a Wellcome Trust Investigator Award (WT202849/Z/16/Z). M. Tobin and L. Wain have been supported by the Medical Research Council (MRC) (MR/N011317/1). The research was partially supported by the National Institute for Health Research (NIHR) Leicester Biomedical Research Centre; the views expressed are those of the author(s) and not necessarily those of the National Health Service (NHS), the NIHR or the Department of Health. I.H. was partially supported by the NIHR Nottingham Biomedical Research Centre; the views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health. This research used the ALICE and SPECTRE High Performance Computing Facilities at the University of Leicester. Additional acknowledgments and funding details for other co-authors and contributing studies (including the SpiroMeta consortium) are in the Supplementary Note.
Author information
Authors and Affiliations
Consortia
Contributions
All authors critically reviewed the manuscript before submission. K.S., U.S.S.G., S.K., S.M.K., T.L., P.S.B., T.H.B., E.R.B., Y.B., Z.C., J.D.C., J.D., D.L.D., C.G., A.G., K.H., J.D.H., J.E.H., P.J., C.L., L.Li, N.L., J.C.M., H.R., I.Sayers, D.D.S., R.T-S., J.C.W., P.G.W., L.M.Y., O.T.R., M.K., O.P., U.G., I.R., I.J.D., N.M.P., H.S., A.L.J., J.F.W., E.Z., M.J., N.W., A.S.B., R.A.S., D.A.M., M.H.C., D.P.S., I.P.H., M.D.T. and L.V.W. contributed to the conception and design of the study. N.S., A.L.G., A.M.E., V.E.J., B.D.H., C.A.M., C.Batini, K.A.F., K.S., P.S., Xingnan Li, R.B., N.F.R., M.O., J.Zhao, M.W., S.W., K.A.K., J.P.C., B.B.S., J.Zhou, J.H., M.I., S.E.H., J.M., S.E., I.Surakka, V.V., T.L., R.J.A., F.D., J.D.H., P.K.J., Xuan Li, A.Mahajan, J.C.M., D.C.N., M.M.P., D.P., D.Q., R.R., H.R., D.S., P.R.H.J.T., M.V., L.M.Y., O.G.T., N.M.P., N.W., E.K.S., C.H., A.P.M., A.S.B., R.A.S., M.H.C., D.P.S., M.D.T. and L.V.W. undertook data analysis. N.S., A.L.G., A.M.E., V.E.J., C.A.M., C.Batini, K.A.F., K.S., P.S., Xingnan Li, N.F.R., M.O., M.W., K.A.K., B.B.S., S.K., M.I., R.J.A., C.Brandsma, J.D., F.D., R.E., C.G., A.G., A.L.H., J.D.H., G.H., P.K.J., C.L., Xuan Li, K.L., L.Lind, J.L., J.C.M., A.Murray, R.P., M.M.P., M.L.P., D.J.P., D.P., D.Q., R.R., H.R., I.Sayers, B.H.S., M.S., L.M.Y., O.G.T., N.M.P., H.S., J.F.W., B.S., M.J., N.W., C.H., A.P.M., A.S.B., R.A.S., R.G.W., M.H.C., D.P.S., I.P.H., M.D.T. and L.V.W. contributed to data acquisition and/or interpretation. N.S., A.L.G., A.M.E., I.P.H., M.D.T. and L.V.W. drafted the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The following authors report potential conflicts of interest: K.S. is an employee of GlaxoSmithKline (GSK) and may own company stock. Z.C. reports grants from GSK and Merck. J.D. reports personal fees and nonfinancial support from Merck Sharp & Dohme (MSD) and Novartis, and grants from British Heart Foundation, European Research Council, MSD, NIHR, NHS Blood and Transplant, Novartis, Pfizer, UK MRC, Wellcome Trust and AstraZeneca. J.D.H. is an employee of GlaxoSmithKline and may own company stock. N.L. is an employee and shareholder of GSK. J.C.M. was a Merck employee during this study, and is now a Celgene employee. D.C.N. has been a Merck & Co. employee during this study and is now an employee at Biogen Inc. H.R. has been a Merck & Co. employee during this study and is now an employee at Biogen Inc. I.S. has received support from GSK and Boehringer Ingelheim. R.T.-S. is an employee and shareholder of GlaxoSmithKline. M.v.d.B. reports grants paid to the University from Astra Zeneca, TEVA, GSK and Chiesi outside the submitted work. J.C.W. is an employee of GSK and may own company stock. L.M.Y.-A. is an employee of GSK and may own company stock. For H.S., Helmholtz Center Munich is funded by the German Federal Ministry of Education and Research (BMBF) and by the State of Bavaria, Competence Network Asthma and COPD (ASCONET), network COSYCONET (subproject 2, BMBF FKZ 01GI0882), funded by the BMBF. In the past three years, E.K.S. received honoraria from Novartis for Continuing Medical Education Seminars and grant and travel support from GlaxoSmithKline. A.S.B. reports grants from Merck, Pfizer, Novartis, Biogen and AstraZeneca and personal fees from Novartis. R.A.S. is an employee and shareholder in GSK. R.G.W. reports that the China Kadoorie Biobank study has received grant support from GSK. M.H.C. has received grant support from GSK. I.P.H. has funded research collaborations with GSK, Boehringer Ingelheim and Orion. M.D.T. receives funding from GSK for a collaborative research project outside of the submitted work. L.V.W. receives funding from GSK for a collaborative research project outside of the submitted work.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Note and Supplementary Figures 1–10 and Supplementary Tables 1–3, 14–16, 19–22, 24 and 27
Supplementary Tables
Supplementary Tables 4–13, 17, 18, 23, 25, 26, 28 and 29
Supplementary Data 1
Region plots for 139 novel signals of association with lung function
Supplementary Data 2
Region plots for 140 previous signals of association with lung function
Rights and permissions
About this article
Cite this article
Shrine, N., Guyatt, A.L., Erzurumluoglu, A.M. et al. New genetic signals for lung function highlight pathways and chronic obstructive pulmonary disease associations across multiple ancestries. Nat Genet 51, 481–493 (2019). https://doi.org/10.1038/s41588-018-0321-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-018-0321-7
This article is cited by
-
Proteomic associations with forced expiratory volume: a Mendelian randomisation study
Respiratory Research (2024)
-
Association and mediation between educational attainment and respiratory diseases: a Mendelian randomization study
Respiratory Research (2024)
-
Lung function and the risk of frailty in the European population: a mendelian randomization study
European Journal of Medical Research (2024)
-
Human genetic associations of the airway microbiome in chronic obstructive pulmonary disease
Respiratory Research (2024)
-
Genetics of chronic respiratory disease
Nature Reviews Genetics (2024)