Abstract
Fundamental studies on graphene (Gr) and its real device applications have been affected by unavoidable defects and impurities which are usually present in synthesized Gr. Therefore, post treatment methods on Gr have been an important subject of research followed by the community. Here, we demonstrate a post-treatment of cm-sized CVD-grown graphene in a Radio Frequency-generated low-pressure plasma of methane and hydrogen to remove oxygen functional groups and heal the structural defects. The optimum plasma treatment parameters, such as pressure, plasma power, and the ratio of the gases, are optimized using in-situ optical emission spectroscopy. This way we present an optimal healing condition monitored with in situ OES. A twofold increase in the conductivity of plasma-treated Gr samples was obtained. Plasma treatment conditions give insights into the possible underlying mechanisms, and the method presents an effective way to obtain improved Gr quality.
Similar content being viewed by others
Introduction
Preparing high-quality CVD-grown graphene (Gr) layers to be implemented in electronic devices has been a great challenge, in both growth and transferring steps. Some examples of undesired effects in Gr after the growth and transferring steps are usually defects, corrugation, cracks, metal and polymer residuals, oxygen functional groups, etc1,2,3,4. Such unlikely damages have limited fabrication technology and large-scale production ability of Gr for widespread and scalable applications like, for example, electronics, energy conversion and storage, chemical and biological sensing, catalysis, or biomedicine hitherto5,6,7,8,9,10,11,12.
As no replacement is yet present for the Gr layer's scalable production otherwise than the current CVD technology and follows up transferring process, different approaches have been suggested to improve the transferred Gr layer's quality based on some alternative treatments.
A critical aspect of current transfer methods that should be resolved is polymer residuals. Various methods, including current-induced cleaning13 and annealing at high temperatures in ultrahigh vacuum or Ar/H2 or N2/H2 environment14,15 have been used to remove polymer residues. In addition to the difficulty of the process and the need for high temperatures, these methods create defects and disorders in Gr16. Hence, new non-destructive methods are demanded to remove the polymer residues and eliminate the defects formed within the transfer process.
Plasma treatment is an efficient method for Gr post-processing and defect engineering17,18, and also provides a promising technique for Gr surface cleaning and healing for a high surface area with a fast processing time19,20,21,22. However, the exploration of plasma processing is still an open field to be developed for further functionality. Different gases like Hydrogen, Argon, and Nitrogen have been used to eliminate polymer residues from the surface of Gr20,21. Although Hydrogen plasma can clean polymer contaminations, defects in the Gr are produced through chemical and physical processes19,23,24. Using a mixture of Hydrogen and Methane gases within various steps has shown low defect density and better removal of polymers. Four different gas combinations of hydrogen and methane were studied in previous studies to obtain a proper method16. A microwave plasma source has been used to generate hydrogen-methane (30/20 sccm) mixed plasma, which had the pressure of 5.8 Torr and the power of 300 W in the whole treatment processes (30 s). The results of that study showed that the simultaneous use of hydrogen and methane as plasma gas would have better results by controlling the surface reactions during the plasma treatment and achieving a nearly perfect and residual free Gr. There is also another study which used the same mixture of methane and hydrogen plasma at 700–900 °C at much lower pressure (0.2–0.4 Torr) and longer treatment duration (30 min)25. However, based on the aforementioned studies that used plasma treatment, no in-situ analyses were made to investigate the exact mechanism of Gr healing process. Thus, to accomplish the mechanism of plasma treatment, in this work, we use in-situ optical emission spectroscopy (OES) to reveal the exact composition of plasma, and monitor the plasma parameter and effective plasma components in the graphene healing and polymer residual removal process. OES is an ideal method to measure the intensity of light emitted from the plasma to detect plasma components in hostile plasma reaction environments. Its versatility is evident as it can be applied for any research that involves the formation of plasma. Spectroscopy is a non-invasive and non-destructive method that does not affect plasma and material processing26,27,28. We introduce a method that obliterates polymer residues, reduces the number of Gr layers, and eliminates defects and oxygen functional groups on the surface of Gr without damaging it by using a mixture of methane and hydrogen plasma without using high temperature treatment. Also, the treated sample is shown to have enhanced electrical transport properties. Results can be considered for obtaining better electronic devices through plasma treatment and illuminate the deriving mechanisms behind plasma treatment of Gr.
Methods and materials
Gr was synthesized using CVD, on highly pure Cu foil (20 µm thickness) substrate as a catalyst, and by using acetylene gas (C2H2) as the carbon source. Usage of C2H2 for Gr growth and CH4 for Gr healing experiments is due to several reasons, e.g., Acetylene's p-bond allows it to adhere firmly to transition metals. It facilitates the dissociation of the hydrocarbon for the formation of graphene sheets29. A high pyrolysis rate in Acetylene also enables it to significantly decrease the defects in graphene, which is due to the divacancy defects healing mechanism30. In addition, the use of Acetylene has been reported to reduce growth temperature31. The synthesized Gr was transferred on Si/SiO2 substrate by wet transfer method using PMMA polymer and FeCl3 (1 M) etchant. The Gr healing and polymer residual removing process was carried out via a mixture of methane and hydrogen plasma (30 sccm of CH4 and 45 sccm of H2) using a low-pressure RF capacitively coupled plasma reactor (RF-CCP). The gas pressure and RF plasma power were 80 mTorr and 80 W, respectively. To characterize the plasma generated in the chamber, OES was performed for different conditions such as gas ratio (CH4/H2), different powers (in the range of 10–200 watts), and pressures between 40 and 200 mTorr. The spectrum of plasma generated in a low-pressure chamber is transferred to the spectrometer (AvaSpec 3648, Avantes) by an optical fiber placed in front of a quartz window embedded inside the chamber's body to collect light from areas of plasma where the sample was placed. The sample's topographical data was acquired by atomic force microscopy (AFM) in non-contact mode (EasyScan 2, Nanosurf) under ambient conditions. Raman spectroscopy was carried out with a Teksan Raman microscope spectrometer (Takram P50C0R10, \(\lambda\) = 532 nm). The conductivity test of Gr was performed using a Keithley 2450 source meter in a two-probe mode.
Results and discussion
In the plasma medium, the decomposition of CH4 occurs mainly by the electron impact and dehydrogenation process and forming active methane species (CHx). To determine the repairing performances during the CH4 plasma treatment, the density functional theory (DFT) calculations have been carried out in previous studies25. One of the most likely methane radicals to form in plasma is CH2, which initially forms a bond with two carbon atoms in the vacancy region and can repair the defect25. Hydrogen atoms released from this process can also form molecular hydrogen considering the barrier energy of the reaction between the vacancy and other possible species in the chamber. It can be concluded that the CH and CH2 radicals are dominant in the repairing process due to the lower barrier energy. The amount of hydrogen generated in this process is not enough to perform the expected hydrogen plasma roles, so we have to add pure H2 to the chamber. Hydrogen plays a vital role in controlling the structure, domains, and layers and removing oxidizing agents25. Therefore, methane with a ratio of hydrogen to the carbon of 4 to 1 is more suitable in this process16,25,32. Acetylene plasma products include species with an even number of carbon atoms, while the process of filling vacancies is done by active single carbon species, especially CH and CH232,33. Production of dust particles is another drawback of using Acetylene34. The balance between etching and Gr growth can be adjusted through various parameters such as gas ratio, pressure, and plasma power16,25.
Figure 1a indicates the OES of plasma from a mixture of hydrogen/methane gas under optimal conditions (power 80 W, pressure 80 mTorr, methane/hydrogen 30/45), including Hy and CHx radiation. In the hydrogen spectrum, Hα line (H (n = 3 → n = 2)), Hβ line (H (n = 4 → n = 2)) and Hγ line (H (n = 5 → n = 2)) are related to the Ballmer series of hydrogen in 656, 486 and 434 wavelengths, respectively. The CH radical emission peak is also seen at 430 nm. The presence of hydrogen affects the conversion of CHx species to more active species. It prevents the recombination of methane molecules, significantly affecting the deposition process and eliminating graphene lattice defects. All possible excitation processes are listed below35:
It should be noted that H alpha and H gamma emission lines follow almost the same procedure as the H beta emission line35.
Figure 1b shows a schematic of plasma processing. The active hydrogen species produced in plasma are potent etching agents that clear the polymer residuals and oxygen functional groups by physical and chemical reactions. As a result, the entire surface of graphene is wholly exposed to plasma; therefore, the defects that exist under the polymer residue or the others induced to the Gr lattice due to the removal of the impurities can be eliminated by the CH4 active species during the mechanism mentioned before.
To achieve the optimal graphene processing conditions, a large number of experiments were performed with different conditions of pressure, gas ratio, and plasma power. A comparison between these conditions and the amount of active species formed by spectroscopy can be found in Supplementary Information. When the amount of hydrogen was too high, we saw the graphene layers degradation and even its complete disappearance from the substrate. On the other side, we saw amorphous carbon deposition on the substrate and even the chamber's inner walls when the methane content was higher than the optimal value.
According to experiments, the optimal conditions for achieving high-quality graphene are established when the ratio of Ha to CH is numerically around 3.5. This ratio alone is not a determining parameter; at high powers, the ions in the plasma will be more energetic, exposure of energetic ions to graphene causes defects or even total damage of the layer. On the other hand, the creation of active species occurs less at low powers. Even active Hydrogen species do not have enough numbers and energy to etch the polymer. In addition, there is a lack of CH species to eliminate defects and vacancies in low power plasma. For the characterization of the induced changes, we use Raman spectroscopy. The Raman spectrum of Gr has two distinct bands, which are labeled as G (1580 cm−1) and 2D (2700 cm−1). In the presence of disorder in Gr lattice, we can also see D-band (1350 cm−1) in the spectrum at about half of the frequency of the 2D-band. The G-band is related to electron–phonon coupling interaction, and its position can change with the state of disorder in materials36. For our sample, before and after the plasma treatment, G-peak's position and intensity did not change.
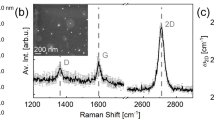
Figure 2a shows the AFM images of graphene before and after plasma treatment with optimized conditions. After plasma processing, graphene has a smoother surface with less roughness, which indicates the removal of polymer residuals on the surface of Gr by plasma etching. The D peak in Fig. 2b is related to the defects in the Gr. Environmental effects and also transferring of Gr through wet method can be the reason for these defects. As we can see in Fig. 2, the D peak intensity decreases after the plasma processing, which means that the sheets have fewer defects than before plasma processing. The quality is higher for the plasma-treated sample. By increasing the number of layers, the width of the 2D peak (2690 cm−1) gets wider37. The 2D peak in bulk graphite has two components, 2D1 and 2D2, and by decreasing the number of layers, it becomes sharp and forms single peak38. This is also evident in Fig. 2b for the sample after the plasma processing. Also, the I2D/IG intensity ratio is a function of Gr layer thickness. For a single layer of Gr, this ratio is about 4.1 and for the bulk is about 0.31, so it means by increasing the number of the layers, this ratio gets smaller39. After plasma processing, the I2D/IG intensity ratio increases, which means that some layers are lifted from the sample, and the Gr layer becomes thinner40. These changes are depicted in Fig. 2. Therefore, both the defect healing and polymer removal are resulted by using our plasma treatment. Further characterizations like high resolution transmission electron microscopy (HRTEM) are suggested to better reveal the role of plasma treatment on the healing of the defects in Gr. But according to the nature of the plasma gas and the chemical reactions presented in Fig. 1 we conclude that the changes in the Raman response and transport properties are also affected by the healing effects in the Gr layer.
(a) AFM of Gr layer before (left image) and after (left image) plasma treatment. (b) Raman spectra of a transferred CVD Gr layers on SiO2 substrate before and after the plasma processing. (c) Gr surface roughness, and I2D/IG value for the Gr before and after plasma processing (the line is plotted as a guide to the eye).
Finally, to see the effect of the polymer removal, defects healing, and thickness decrease on the transport properties, I–V measurements were performed. In Fig. 3, the I–V curve of graphene before and after plasma processing is shown. Gr with hydroxyl and carboxyl functional groups has a semiconductive property and can result in Schottky contact. In our sample before plasma treatment, the I–V curve briefly deviates from a completely linear behavior, which might be due to the presence of a small amount of hydroxyl and carboxyl functional group41,42,43. This nonlinearity in the I–V curve disappeared after plasma treatment, which might be due to the removal of the hydroxyl and carboxyl functional groups by plasma treatment. The inset of Fig. 3 shows the resistivity of the samples calculated from the slope of the I–V curve at the origin and also the probe distances. The error bar is obtained from several measurements performed for each sample. As can be seen, the resistivity of the Gr is decreased by plasma treatment. The electrical properties of graphene can be due to the scattering of charged impurities41. In-plane conductivity of Gr is significantly dependent on Gr structure and purity such that the presence of structural disorders, defects, and bonding functional groups such as oxygen groups reduces the electrical conductivity of Gr44. Vacancies that are scattering centers45 and defects reduce the mobility of charge carriers that cause increasing the Gr resistivity46,47. Also, conductivity is inversely proportional to the number of layers, so that single-layer graphene has the highest conductivity48. It is known that the sample conductivity increases after the healing process and is related to the recovery of sp2 C=C bonds (graphitization) in graphitic structures49. In our sample, both the thickness change and defects healing have resulted in the observed increase of conductivity, and we cannot separate their contributions.
Conclusions
In summary, we obtained the optimal plasma conditions (pressure, power, and gas ratio) of a mixture of methane and hydrogen plasma by OES to balance the etching and deposition rate and reduce the defects and disorder of Gr, and also remove the polymer residuals properly. These were evidenced by comparing the Raman and AFM measurements before and after plasma treatment. Also, we showed that by plasma treatment, the electrical conductivity increased by ~ 100% and can result from defects healing and thickness change. Results are important towards improving Gr quality for using in electronic devices (Supplementary Information).
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Chen, Y., Gong, X. L. & Gai, J. G. Progress and challenges in transfer of large-area graphene films. Adv. Sci. 3, 1–15 (2016).
Kang, J., Shin, D., Bae, S. & Hong, B. H. Graphene transfer: Key for applications. Nanoscale 4, 5527–5537 (2012).
Ma, L. P., Ren, W. & Cheng, H. M. Transfer methods of graphene from metal substrates: A review. Small Methods 3, 1–13 (2019).
Li, X. et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 324, 1312–1314 (2009).
Rozada, R., Paredes, J. I., Villar-Rodil, S., Martínez-Alonso, A. & Tascón, J. M. D. Towards full repair of defects in reduced graphene oxide films by two-step graphitization. Nano Res. 6, 216–233 (2013).
Huang, X. et al. Graphene-based materials: Synthesis, characterization, properties, and applications. Small 7, 1876–1902 (2011).
Shao, Y. et al. Nitrogen-doped graphene and its electrochemical applications. J. Mater. Chem. 20, 7491–7496 (2010).
Gunlycke, D., Areshkin, D. A., Li, J., Mintmire, J. W. & White, C. T. Graphene nanostrip digital memory device. Nano Lett. 7, 3608–3611 (2007).
Avouris, P. & Dimitrakopoulos, C. Graphene: Synthesis and applications. Mater. Today 15, 86–97 (2012).
Zhang, Y., Nayak, T. R., Hong, H. & Cai, W. Graphene: A versatile nanoplatform for biomedical applications. Nanoscale 4, 3833–3842 (2012).
Yang, Y., Asiri, A. M., Tang, Z., Du, D. & Lin, Y. Graphene based materials for biomedical applications. Mater. Today 16, 365–373 (2013).
Georgakilas, V. et al. Noncovalent functionalization of graphene and graphene oxide for energy materials, biosensing, catalytic, and biomedical applications. Chem. Rev. 116, 5464–5519 (2016).
Moser, J., Barreiro, A. & Bachtold, A. Current-induced cleaning of graphene. Appl. Phys. Lett. 91, 1–4 (2007).
Cao, H. et al. Large-scale graphitic thin films synthesized on Ni and transferred to insulators: Structural and electronic properties. J. Appl. Phys. 107, 044310 (2010).
Ishigami, M., Chen, J. H., Cullen, W. G., Fuhrer, M. S. & Williams, E. D. Atomic structure of graphene on SiO2. Nano Lett. 7, 1643–1648 (2007).
Yun, H. et al. Removal of photoresist residues and healing of defects on graphene using H2 and CH4 plasma. Appl. Surf. Sci. 463, 802–808 (2019).
Vinchon, P., Glad, X., Robert Bigras, G., Martel, R. & Stafford, L. Preferential self-healing at grain boundaries in plasma-treated graphene. Nat. Mater. https://doi.org/10.1038/s41563-020-0738-0 (2020).
Vinchon, P. et al. A combination of plasma diagnostics and Raman spectroscopy to examine plasma-graphene interactions in low-pressure argon radiofrequency plasmas. J. Appl. Phys. 126, 233302 (2019).
Ferrah, D. et al. XPS investigations of graphene surface cleaning using H2- and Cl2-based inductively coupled plasma. Surf. Interface Anal. 48, 451–455 (2016).
Cunge, G. et al. Dry efficient cleaning of poly-methyl-methacrylate residues from graphene with high-density H2 and H2–N2 plasmas. J. Appl. Phys. 118, 123302 (2015).
Lim, Y. D. et al. Si-compatible cleaning process for graphene using low-density inductively coupled plasma. ACS Nano 6, 4410–4417 (2012).
Cheng, M. et al. Restoration of graphene from graphene oxide by defect repair. Carbon 50, 2581–2587 (2012).
Geng, D. et al. Direct top-down fabrication of large-area graphene arrays by an in situ etching method. Adv. Mater. 27, 4195–4199 (2015).
Fang, L., Yuan, W., Wang, B. & Xiong, Y. applied surface science growth of graphene on Cu foils by microwave plasma chemical vapor deposition: The effect of in-situ hydrogen plasma post-treatment. Appl. Surf. Sci. 383, 28–32 (2016).
Zhu, D. et al. Healing of reduced graphene oxide with methane + hydrogen plasma. Carbon 120, 274–280 (2017).
Donné, A. J. H. Introduction to plasma diagnostics. Fusion Sci. Technol. 53, 379–386 (2008).
Coburn, J. W. & Chen, M. Optical emission spectroscopy of reactive plasmas: A method for correlating emission intensities to reactive particle density. J. Appl. Phys. 51, 3134–3136 (1980).
Fassel, V. A. Quantitative elemental analyses by plasma emission spectroscopy. Science 202, 183 (1978).
Seah, C., Chai, S. & Mohamed, A. R. Mechanisms of graphene growth by chemical vapour deposition on transition metals. Carbon N. Y. 70, 1–21 (2013).
Qi, M. et al. Hydrogen Kinetics on Scalable Graphene Growth by Atmospheric Pressure Chemical Vapor Deposition with Acetylene (2013).
Nandamuri, G., Roumimov, S. & Solanki, R. Chemical vapor deposition of graphene films. Nanotechnology 21(14), 145604. https://doi.org/10.1088/0957-4484/21/14/145604 (2010).
Benedikt, J. Plasma-chemical reactions: Low pressure acetylene plasmas. J. Phys. D. Appl. Phys. 43, 043001 (2010).
Deschenaux, C., Affolter, A., Magni, D., Hollenstein, C. & Fayet, P. Investigations of CH4, C2H2 and C2H4 dusty RF plasmas by means of FTIR absorption spectroscopy and mass spectrometry. J. Phys. D. Appl. Phys. 32, 1876–1886 (1999).
Berndt, J., Kovačević, E., Stefanović, I. & Boufendi, L. Controlled dust formation in pulsed rf plasmas. J. Appl. Phys. 106, 063309 (2009).
Pastol, A. & Catherine, Y. Optical emission spectroscopy for diagnostic and monitoring of CH4 plasmas used for a-C: H deposition. J. Phys. D. Appl. Phys. 23, 799–805 (1990).
Beams, R., Gustavo Cançado, L. & Novotny, L. Raman characterization of defects and dopants in graphene. J. Phys. Condens. Matter 27, 083002 (2015).
Merlen, A., Buijnsters, J. G. & Pardanaud, C. A guide to and review of the use of multiwavelength Raman spectroscopy for characterizing defective aromatic carbon solids: From graphene to amorphous carbons. Coatings 7, 153 (2017).
Ferrari, A. C. et al. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 97, 1–4 (2006).
Das, A., Chakraborty, B. & Sood, A. K. Raman spectroscopy of graphene on different substrates and influence of defects. Bull. Mater. Sci. 31, 579–584 (2008).
Fan, H. N. et al. Exploration of the sodium ion ordered transfer mechanism in a MoSe2@Graphene composite for superior rate and lifespan performance. J. Mater. Chem. A 7, 13736–13742 (2019).
Pirkle, A. et al. The effect of chemical residues on the physical and electrical properties of chemical vapor deposited graphene transferred to SiO2. Appl. Phys. Lett. 99, 2009–2012 (2011).
Na, M. & Rhee, S. W. Electronic characterization of Al/PMMA[poly(methyl methacrylate)]/p-Si and Al/CEP(cyanoethyl pullulan)/p-Si structures. Org. Electron. 7, 205–212 (2006).
Farmer, D. B. et al. Chemical doping and electron-hole conduction asymmetry in graphene devices. Nano Lett. 9, 388–392 (2009).
Zeng, W. et al. Defect-engineered reduced graphene oxide sheets with high electric conductivity and controlled thermal conductivity for soft and flexible wearable thermoelectric generators. Nano Energy 54, 163–174 (2018).
Chen, J. H., Cullen, W. G., Jang, C., Fuhrer, M. S. & Williams, E. D. Defect scattering in graphene. Phys. Rev. Lett. 102, 1–4 (2009).
Vicarelli, L., Heerema, S. J., Dekker, C. & Zandbergen, H. W. Controlling defects in graphene for optimizing the electrical properties of graphene nanodevices. ACS Nano 9, 3428–3435 (2015).
Lherbier, A. et al. Transport properties of graphene containing structural defects. Phys. Rev. B Condens. Matter Mater. Phys. 86, 1–18 (2012).
Cao, M. S., Wang, X. X., Cao, W. Q. & Yuan, J. Ultrathin graphene: Electrical properties and highly efficient electromagnetic interference shielding. J. Mater. Chem. C 3, 6589–6599 (2015).
Su, C. Y. et al. Highly efficient restoration of graphitic structure in graphene oxide using alcohol vapors. ACS Nano 4, 5285–5292 (2010).
Acknowledgements
S.M. Mohseni acknowledges Iran National Science Foundation: INSF for a grant No. 99002503.
Author information
Authors and Affiliations
Contributions
M.S.: performed the experiments, analyzed the data, writing—original draft, review and editing, conceptualization, resources, methodology, investigation, validation. P.B.: performed the experiments, analyzed the data, writing—review and editing, visualization, resources, investigation, validation. L.J.: analyzed the data, writing—review and editing, resources, methodology, conceptualization, investigation, validation. A.M.: writing—review and editing, validation. M.M., B.S.: supervision, resources, writing—review & editing, project administration, funding acquisition, validation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salehi, M., Bastani, P., Jamilpanah, L. et al. Low defect and high electrical conductivity of graphene through plasma graphene healing treatment monitored with in situ optical emission spectroscopy. Sci Rep 11, 20334 (2021). https://doi.org/10.1038/s41598-021-99421-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-99421-7
This article is cited by
-
Optimization of the Radiofrequency Low-Pressure Cold Plasma Conditions for Decontamination of Saffrons
Food and Bioprocess Technology (2024)
-
Temperature dependence of electrical conductivity and variable hopping range mechanism on graphene oxide films
Scientific Reports (2023)
-
An innovative model for conductivity of graphene-based system by networked nano-sheets, interphase and tunneling zone
Scientific Reports (2022)
-
Effect of contact resistance on the electrical conductivity of polymer graphene nanocomposites to optimize the biosensors detecting breast cancer cells
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.