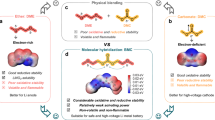
Abstract
The cost of fuel cell systems can be largely reduced by developing hydroxide exchange membrane fuel cells (HEMFCs) based on platinum group metal-free (PGM-free) catalysts. However, the sluggish hydrogen oxidation reaction (HOR) in alkaline electrolytes forces HEMFCs to use higher PGM loadings at the anode than proton exchange membrane fuel cells to sustain the desired power densities. Here we report nickel–molybdenum–niobium metallic glasses as PGM-free HOR catalysts. The optimal Ni52Mo13Nb35 metallic glass exhibits an intrinsic exchange current density of 0.35 mA cm−2, outperforming that of a Pt disk catalyst (0.30 mA cm−2). This catalyst also shows remarkable robustness in alkaline electrolyte with a wide stability window up to 0.8 V versus the reversible hydrogen electrode. When used as the anode, this catalyst enables power densities of 390 mW cm−2 in H2/O2 fuel cells and 253 mW cm−2 in H2/air fuel cells, and shows negligible performance degradation over 50 h and 30 h, respectively.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are presented in the article and Supplementary Information. Source data are provided with this paper. Any other relevant data are also available from the corresponding authors upon reasonable request.
References
Sekine, S. & Kojima, K. Progress and challenges in Toyota’s fuel cell vehicle development. In 16th Asia Pacific Automotive Engineering Conference https://doi.org/10.4271/2011-28-0061 (The Automotive Research Association of India, 2011).
Shinji Jomori, K. K., Nonoyama, N. & Manabu Kato, T. Y. Experimental study of the activity change due to operation history in PEMFC. Electrochem. Soc. 58, 1457–1469 (2013).
Chattot, R. et al. Surface distortion as a unifying concept and descriptor in oxygen reduction reaction electrocatalysis. Nat. Mater. 17, 827–833 (2018).
Setzler, B. P., Zhuang, Z., Wittkopf, J. A. & Yan, Y. Activity targets for nanostructured platinum-group-metal-free catalysts in hydroxide exchange membrane fuel cells. Nat. Nanotechnol. 11, 1020–1025 (2016).
Strmcnik, D. et al. Improving the hydrogen oxidation reaction rate by promotion of hydroxyl adsorption. Nat. Chem. 5, 300–306 (2013).
Sheng, W., Gasteiger, H. A. & Shao-Horn, Y. Hydrogen oxidation and evolution reaction kinetics on platinum: acid vs alkaline electrolytes. J. Electrochem. Soc. 157, B1529–B1536 (2010).
Tatus-Portnoy, Z. et al. A low-loading Ru-rich anode catalyst for high-power anion exchange membrane fuel cells. Chem. Commun. 56, 5669–5672 (2020).
Rheinländer, P. J., Herranz, J., Durst, J. & Gasteiger, H. A. Kinetics of the hydrogen oxidation/evolution reaction on polycrystalline platinum in alkaline electrolyte reaction order with respect to hydrogen pressure. J. Electrochem. Soc. 161, F1448–F1457 (2014).
Zhuang, Z. et al. Nickel supported on nitrogen-doped carbon nanotubes as hydrogen oxidation reaction catalyst in alkaline electrolyte. Nat. Commun. 7, 10141 (2016).
Davydova, E. S., Mukerjee, S., Jaouen, F. & Dekel, D. R. Electrocatalysts for hydrogen oxidation reaction in alkaline electrolytes. ACS Catal. 8, 6665–6690 (2018). 2018.
Dekel, D. R. Review of cell performance in anion exchange membrane fuel cells. J. Power Sources 375, 158–169 (2018).
Cherstiouk, O. V. et al. Electrocatalysis of the hydrogen oxidation reaction on carbon-supported bimetallic NiCu particles prepared by an improved wet chemical synthesis. J. Electroanal. Chem. 783, 146–151 (2016).
Gao, L. et al. A nickel nanocatalyst within a h-BN shell for enhanced hydrogen oxidation reactions. Chem. Sci. 8, 5728–5734 (2017).
Kiros, Y., Majari, M. & Nissinen, T. A. Effect and characterization of dopants to Raney nickel for hydrogen oxidation. J. Alloys Compd. 360, 279–285 (2003).
Ni, W. et al. Ni3N as an active hydrogen oxidation reaction catalyst in alkaline medium. Angew. Chem. Int. Ed. 58, 7445–7449 (2019).
Yang, Y. et al. Enhanced electrocatalytic hydrogen oxidation on Ni/NiO/C derived from a Ni-based MOF. Angew. Chem. Int. Ed. 131, 10754–10759 (2019).
Kabir, S. et al. Platinum group metal-free NiMo hydrogen oxidation catalysts: high performance and durability in alkaline exchange membrane fuel cells. J. Mater. Chem. A 5, 24433–24443 (2017).
Sheng, W. et al. Non-precious metal electrocatalysts with high activity for hydrogen oxidation reaction in alkaline electrolytes. Energy Environ. Sci. 7, 1719–1724 (2014).
Yang, F. et al. Boosting hydrogen oxidation activity of Ni in alkaline media through oxygen-vacancy-rich CeO2/Ni heterostructures. Angew. Chem. Int. Ed. 58, 14179–14183 (2019).
Lu, S., Pan, J., Huang, A., Zhuang, L., & Lu, J. Alkaline polymer electrolyte fuel cells completely free from noble metal catalysts. Proc. Natl Acad. Sci. USA 105, 20611–20614 (2008).
Song, F. et al. Interfacing nickel nitride and nickel boosts both electrocatalytic hydrogen evolution and oxidation reactions. Nat. Commun. 9, 4531 (2018).
Duan, Y. et al. Bimetallic nickel-molybdenum/tungsten nanoalloys for high-efficiency hydrogen oxidation catalysis in alkaline electrolytes. Nat. Commun. 11, 4789 (2020).
Davydova, E. S., Speck, F. D., Paul, M. T. Y., Dekel, D. R. & Cherevko, S. Stability limits of Ni-based hydrogen oxidation electrocatalysts for anion exchange membrane fuel cells. ACS Catal. 9, 6837–6845 (2019).
Qin, S. et al. Ternary nickel–tungsten–copper alloy rivals platinum for catalyzing alkaline hydrogen oxidation. Nat. Commun. 12, 2686 (2021).
Oshchepkov, A. G. et al. Nanostructured nickel nanoparticles supported on vulcan carbon as a highly active catalyst for the hydrogen oxidation reaction in alkaline media. J. Power Sources 402, 447–452 (2018).
Davydova, E., Zaffran, J., Dhaka, K., Toroker, M. & Dekel, D. Hydrogen oxidation on Ni-based electrocatalysts: the effect of metal doping. Catalysts 8, 454 (2018).
Winsel, E. W. & Ja, A. W. The DSK system of fuel cell electrodes. J. Electrochem. Soc. 108, 1073–1079 (1961).
Roy, A. et al. Nickel–copper supported on a carbon black hydrogen oxidation catalyst integrated into an anion-exchange membrane fuel cell. Sustain. Energy Fuels 2, 2268–2275 (2018).
Klement, W., Willens, R. H. & Duwez, P. Non-crystalline structure in solidified gold-silicon alloys. Nature 187, 869–870 (1960).
Greer, A. L. Metallic glasses…on the threshold. Mater. Today 12, 14–22 (2009).
Escudero-Escribano, M. et al. Tuning the activity of Pt alloy electrocatalysts by means of the lanthanide contraction. Science 352, 73–76 (2016).
Zhang, X. et al. Lithiation-induced amorphization of Pd3P2S8 for highly efficient hydrogen evolution. Nat. Catal. 1, 460–468 (2018).
Zhang, B. et al. Homogeneously dispersed multimetal oxygen-evolving catalysts. Science 352, 333–337 (2016).
Carmo, M. et al. Bulk Metallic glass nanowire architecture for electrochemical applications. ACS Nano 5, 2979–2298 (2011).
Li, J. et al. Combinatorial screening of Pd-based quaternary electrocatalysts for oxygen reduction reaction in alkaline media. J. Mater. Chem. A 5, 67–72 (2017).
Sekol, R. C. et al. Bulk metallic glass micro fuel cell. Small 9, 2081–2085 (2013) 2026.
Schilter, D., Fuller, A. L. & Gray, D. L. Nickel-molybdenum and nickel-tungsten dithiolates: hybrid models for hydrogenases and hydrodesulfurization. Eur. J. Inorg. Chem. 2015, 4638–4642 (2015).
Inoue, A. & Ta, A. Classification of bulk metallic glasses by atomic size difference, heat of mixing and period of constituent elements and its application to characterization of the main alloying element. Mater. Trans. 46, 2817–2829 (2005).
Wang, Y., Brezesinski, T., Antonietti, M. & Smarsly, B. Ordered mesoporous Sb-, Nb-, and Ta-doped SnO2 thin films with adjustable doping levels and high electrical conductivity. ACS Nano 3, 1373–1378 (2009).
Wei, X. et al. In situ visualizing atomic structural evolution during crystallization in ternary Zr–Cu–Al bulk metallic glasses. Intermetallics 105, 173–178 (2019).
Fernandez Garrillo, P. A., Grevin, B., Chevalier, N. & Borowik, L. Calibrated work function mapping by Kelvin probe force microscopy. Rev. Sci. Instrum. 89, 043702 (2018).
Ashby, M. & Greer, A. Metallic glasses as structural materials. Scr. Mater. 54, 321–326 (2006).
Johnson, W. L. Bulk glass-forming metallic alloys: science and technology. MRS Bull. 24, 42–56 (1999).
Schroers, J. Bulk metallic glasses. Phys. Today 66, 32–37 (2013).
Lan, S. et al. A medium-range structure motif linking amorphous and crystalline states. Nat. Mater. 20, 1347–1352 (2021).
Lan, S. et al. Hidden amorphous phase and reentrant supercooled liquid in Pd-Ni-P metallic glasses. Nat. Commun. 8, 14679 (2017).
Kube, S. A. et al. Compositional dependence of the fragility in metallic glass forming liquids. Nat. Commun. 13, 3708 (2022).
Zhukov, A. et al. Trends in optimization of giant magnetoimpedance effect in amorphous and nanocrystalline materials. J. Alloys Compd. 727, 887–901 (2017).
Inoue, A. & Nishiyama, N. New bulk metallic glasses for applications as magnetic-sensing, chemical, and structural materials. MRS Bull. 32, 651–658 (2007).
Glasscott, M. W. et al. Electrosynthesis of high-entropy metallic glass nanoparticles for designer, multi-functional electrocatalysis. Nat. Commun. 10, 2650 (2019).
Tan, Y. et al. Noble‐metal‐free metallic glass as a highly active and stable bifunctional electrocatalyst for water splitting. Adv. Mater. Interfaces 4, 1601086 (2017).
Bard, A. J. & Faulkner, L. R. Electrochemical Methods: Fundamentals and Applications (Wiley, 2001).
Sun, Y., Dai, Y., Liu, Y. & Chen, S. A rotating disk electrode study of the particle size effects of Pt for the hydrogen oxidation reaction. Phys. Chem. Chem. Phys. 14, 2278–2285 (2012).
Cong, Y., Yi, B. & Song, Y. Hydrogen oxidation reaction in alkaline media: from mechanism to recent electrocatalysts. Nano Energy 44, 288–303 (2018).
Camara, G. A., Ticianelli, E. A., Mukerjee, S., Lee, J. & McBreen, J. The CO poisoning mechanism of the hydrogen oxidation reaction in proton exchange membrane fuel cells. J. Electrochem. Soc. 149, A748–A753 (2002).
Wu, G. et al. A general synthesis approach for amorphous noble metal nanosheets. Nat. Commun. 10, 4855 (2019).
Sheng, H. W., Luo, W. K., Alamgir, F. M., Bai, J. M. & Ma, E. Atomic packing and short-to-medium-range order in metallic glasses. Nature 439, 419–425 (2006).
Gross, O. et al. Signatures of structural differences in Pt–P- and Pd–P-based bulk glass-forming liquids. Commun. Phys. 2, 83 (2019).
Ding, J., Ma, E., Asta, M. & Ritchie, R. O. Second-nearest-neighbor correlations from connection of atomic packing motifs in metallic glasses and liquids. Sci. Rep. 5, 17429 (2015).
Miracle, D. B. A structural model for metallic glasses. Nat. Mater. 3, 697–702 (2004).
Ma, D., Stoica, A. D. & Wang, X. L. Power-law scaling and fractal nature of medium-range order in metallic glasses. Nat. Mater. 8, 30–34 (2008).
Giles, S. A. et al. Recent advances in understanding the pH dependence of the hydrogen oxidation and evolution reactions. J. Catal. 367, 328–331 (2018).
Zheng, J., Nash, J., Xu, B. & Yan, Y. Towards establishing apparent hydrogen binding energy as the descriptor for hydrogen oxidation/evolution reactions. J. Electrochem. Soc. 165, H27–H29 (2018).
Cheng, T., Wang, L., Merinov, B. V. & Goddard, W. A. III Explanation of dramatic pH-dependence of hydrogen binding on noble metal electrode: greatly weakened water adsorption at high pH. J. Am. Chem. Soc. 140, 7787–7790 (2018).
Yang, X., Nash, J., Oliveira, N., Yan, Y. & Xu, B. Understanding the pH dependence of underpotential deposited hydrogen on platinum. Angew. Chem. Int. Ed. 58, 17718–17723 (2019).
Ledezma-Yanez, I. et al. Interfacial water reorganization as a pH-dependent descriptor of the hydrogen evolution rate on platinum electrodes. Nat. Energy 2, 17031 (2017).
Stevens, M. B. et al. Ternary Ni-Co-Fe oxyhydroxide oxygen evolution catalysts: intrinsic activity trends, electrical conductivity, and electronic band structure. Nano Res. 12, 2288–2295 (2019).
Zaffran, J., Nagli, M., Shehadeh, M. & Toroker, M. C. Efficient cationic agents for exfoliating two-dimensional nickel oxide sheets. Theor. Chem. Acc. 137, 3 (2018).
Zaffran, J. & Toroker, M. C. Understanding the oxygen evolution reaction on a two-dimensional NiO2 catalyst. ChemElectroChem 4, 2764–2770 (2017).
Zaffran, J. et al. Influence of electrolyte cations on Ni(Fe)OOH catalyzed oxygen evolution reaction. Chem. Mater. 29, 4761–4767 (2017).
Zaffran, J. & Caspary Toroker, M. Benchmarking density functional theory based methods to model NiOOH material properties: Hubbard and van der Waals corrections vs hybrid functionals. J. Chem. Theory Comput. 12, 3807–3812 (2016).
Fidelsky, V., Butera, V., Zaffran, J. & Toroker, M. C. Three fundamental questions on one of our best water oxidation catalysts: a critical perspective. Theor. Chem. Acc. 135, (2016).
Yayapao, O., Phuruangrat, A., Thongtem, T. & Thongtem, S. Synthesis, characterization and electrochemical properties of α-MoO3 nanobelts for Li-ion batteries. Russ. J. Phys. Chem. A 90, 1224–1230 (2016).
Duan, Y. et al. Scaled-up synthesis of amorphous NiFeMo oxides and their rapid surface reconstruction for superior oxygen evolution catalysis. Angew. Chem. Int. Ed. 58, 15772–15777 (2019).
Suryanarayana, C. & Inoue, A. Bulk Metallic Glasses (CRC Press, 2017).
Yoon, Y., Yan, B. & Surendranath, Y. Suppressing ion transfer enables versatile measurements of electrochemical surface area for intrinsic activity comparisons. J. Am. Chem. Soc. 140, 2397–2400 (2018).
Egami, T. & Billinge, S. Underneath the Bragg Peaks: Structural Analysis of Complex Materials (Pergamon Press, 2003).
Dimitrov, D. A., Röde, H. & Bishop, A. R. Peak positions and shapes in neutron pair correlation functions from powders of highly anisotropic crystals. Phys. Rev. B 64, 014303 (2001).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Blochl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Nosé, S. A unified formulation of the constant temperature molecular dynamics methods. J. Chem. Phys. 81, 511–519 (1984).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Acknowledgements
We thank S. L. Chen at Wuhan University for many helpful discussions. This work was supported by the National Basic Research Program of China (grant no. 2018YFA0702001), the National Natural Science Foundation of China (grant nos. 22225901, 22175162, 21975237, 21521001, 21431006, 21225315, 21321002, 91645202 and 51871120), the Chinese Academy of Sciences (grant nos. KGZD-EW-T05 and XDA090301001), the Strategic Priority Research Program of the Chinese Academy of Sciences (grant no. XDA21000000), the Anhui Provincial Research and Development Program (grant no. 202004a05020073), the USTC Research Funds of the Double First-Class Initiative (grant no. YD2340002007), the Fundamental Research Funds for the Central Universities (grant nos. WK9990000101, 30919011107 and 30919011404), the Natural Science Foundation of Jiangsu Province (grant no. BK20200019), the National Key R&D Program of China (grant no. 2021YFB3802800) and Guangdong–Hong Kong–Macao Joint Laboratory for Neutron Scattering Science and Technology. This research used the resources of the Advanced Photon Source, a US Department of Energy (DOE), Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357.
Author information
Authors and Affiliations
Contributions
M.-R.G. and S.L. conceived the idea and directed the project. S.-H.Y. and Y.Y. advised on the research and contributed to the scientific interpretation. F.-Y.G., S.-N.L. and J.-C.G. fabricated the metallic glasses and collected and analysed the data. X.-L.Z., L.Z., Z.-G.D. and W.L. performed the DFT calculations. Y.-R.Z. and Y.D. carried out the XRD measurements. W.D. performed the DMA measurements. R.-C.B. and X.Y. assisted with the electrochemical measurements. S.Q., Z.-Z.N. and P.-P.Y. assisted with the structure analyses. F.-Y.G., S.-N.L., J.-C.G., S.L. and M.-R.G. wrote and edited the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Hamish Miller, Jan Schroers and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Discussion, Figs. 1–51 and Tables 1–5.
Supplementary Data
Atomic coordinates of the simulated Ni52Mo13Nb35 models.
Source data
Source Data Fig. 4
The data used to plot the figures.
Source Data Fig. 5
The data used to plot the figures.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, FY., Liu, SN., Ge, JC. et al. Nickel–molybdenum–niobium metallic glass for efficient hydrogen oxidation in hydroxide exchange membrane fuel cells. Nat Catal 5, 993–1005 (2022). https://doi.org/10.1038/s41929-022-00862-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00862-8
This article is cited by
-
Metal-support interaction boosts the stability of Ni-based electrocatalysts for alkaline hydrogen oxidation
Nature Communications (2024)
-
Unveiling the nature of Pt-induced anti-deactivation of Ru for alkaline hydrogen oxidation reaction
Nature Communications (2024)
-
Advances in bio-inspired electrocatalysts for clean energy future
Nano Research (2024)
-
Embedding oxophilic rare-earth single atom in platinum nanoclusters for efficient hydrogen electro-oxidation
Nature Communications (2023)
-
Optimized valence state of Co and Ni in high-entropy alloy for high active-stable OER
Rare Metals (2023)