Abstract
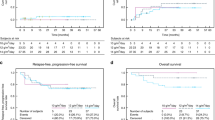
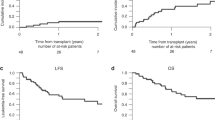
Relapse of the primary disease remains the predominant cause of death following bone marrow transplantation for high-risk haematological malignancies. Improved supportive care and patient selection have resulted significant improvements in toxicity with standard conditioning regimens. Further dose intensification to reduce the risk of relapse may therefore be feasible. We determined the maximal tolerated dose (MTD) of a 5-day continuous infusion (CI) of etoposide when added to oral busulphan 16 mg/kg and intravenous cyclophosphamide 120 mg/kg (Bu/Cy) as conditioning in 44 autograft and 18 allograft recipients at high risk of relapse. The major toxicity of escalating doses of etoposide was oral and gastro-intestinal mucositis, reflected by a statistically significant increase in the requirement for total parenteral nutrition in both autografts and allograft recipients. Time to neutrophil and platelet recovery, opiate analgesia requirements, and duration of hospitalization were not affected by etoposide dose escalation. The MTD in autograft recipients was 300 mg/m2/day (1500 mg/m2 total dose), and 100 mg/m2/day (500 mg/m2 total dose) for allograft recipients. Mucositis and hepatotoxicity were more frequent in allograft recipients, suggesting that methotrexate may have contributed to the lower tolerable dose in these patients. As a consequence, further dose escalation may not be possible in heavily pre-treated patients undergoing allogeneic transplantation. Conversely, high dose CI etoposide can be added with relative safety to Bu/Cy in autograft recipients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tutschka PJ . Copelan EA. Klein JP. Bone marrow transplantation for leukemia following a new busulfan and cyclophosphamide regimen Blood 1987 70: 1382 1388
Bandini G, Belardinelli A, Rosti G et al. Toxicity of high-dose busulphan and cyclophophamide as conditioning therapy for allogeneic bone marrow transplantation in adults with haematological malignancies Bone Marrow Transplant 1994 13: 577 581
Rosenthal MA, Grigg AP, Sheridan WP . High dose busulphan/cyclophosphamide for autologous bone marrow transplantation is associated with minimal non-hemopoietic toxicity Leuk Lymph 1994 14: 279 283
Vaughan WP, Dennison JD, Reed EC et al. Improved results of allogeneic bone marrow transplantation for advanced hematologic malignancy using busulfan, cyclophosphamide and etoposide as cytoreductive and immunosuppressive therapy Bone Marrow Transplant 1991 8: 489 495
Zander AR, Berger C, Kroger N et al. High dose chemotherapy with busulphan, cyclophosphamide, and etoposide as conditioning regimen for allogeneic bone marrow transplantation for patients with acute myeloid leukemia in first complete remission Clin Cancer Res 1997 3: 2671 2675
Crilley P, Topolsky D, Styler MJ et al. Extramedullary toxicity of a conditioning regimen containing busulfan, cyclophosphamide and etoposide in 84 patients undergoing autologous and allogeneic bone marrow transplantation Bone Marrow Transplant 1995 15: 361 365
Spitzer TR, Cottler-Fox M, Torrisi J et al. Escalating doses of etoposide with cyclphosphamide and fractionated total body irradiation or busulfan as conditioning for bone marrow transplantation Bone Marrow Transplant 1989 4: 559 565
Ratanatharathorn V, Karanes C, Lum LG et al. Allogeneic bone marrow transplantation in high-risk myeloid disorders using busulfan, cytosine arabinoside and cyclophosphamide (BAC) Bone Marrow Transplant 1992 9: 49 55
Geller RB, Myers S, Devine S et al. Phase I study of busulfan, cyclophosphamide, and timed sequential escalating doses of cytarabine followed by bone marrow transplantation Bone Marrow Transplant 1992 9: 41 47
Jerjis S, Roovers E, Muus P et al. Idarubicin to intensify the conditioning of autologous bone marrow transplantation for patients with acute myeloid leukemia in first complete remission Bone Marrow Transplant 1998 22: 13 19
Kroger N, Zabelina T, Sonnenberg S et al. Dose-dependent effect of etoposide in combination with busulfan plus cyclophosphamide as conditioning for stem cell transplantation in patients with acute myeloid leukemia Bone Marrow Transplant 2000 26: 711 716
Kanda Y, Akiyama H, Tanikawa S et al. Etoposide with/without G-CSF with busulfan and cyclophosphamide as conditioning for bone marrow transplantation. The BMT Team Am J Hematol 1996 51: 265 268
Jones RJ, Santos GW . New conditioning regimens for high risk marrow transplants Bone Marrow Transplant 1989 4: (Suppl. 4) 15 17
Rosenfeld CS, Przepiorka D, Schwinghammer TL et al. Autologous bone marrow transplantation following high-dose busulfan and VP-16 for advanced non-Hodgkin's lymphoma and Hodgkin's disease Exp Hematol 1991 19: 317 321
Mross K, Bewermeier P, Reifke J et al. Pharmacokinetics of high dose VP-16: 6-hour infusion versus 34-hour infusion Bone Marrow Transplant 1994 13: 423 430
Herzig RH . High-dose etoposide and marrow transplantation Cancer 1990 67: (Suppl.) 292 298
Thompson DS, Hainsworth JD, Hande KR et al. Prolonged administration of low-dose, infusional etoposide in patients with etoposide-sensitive neoplasms: a phase I/II study J Clin Oncol 1993 11: 1322 1328
Ahmed T, Engelking C, Szalyga J et al. Propantheline prevention of mucositis from etoposide Bone Marrow Transplant 1993 12: 131 132
Bearman SI, Appelbaum FR, Back A et al. Regimen-related toxicity and early posttransplant survival in patients undergoing marrow transplantation for lymphoma J Clin Oncol 1989 7: 1288 1294
Hoyt R, Szer J, Grigg A . Neurological events associated with the infusion of cryopreserved bone marrow and/or peripheral blood progenitor cells Bone Marrow Transplant 2000 25: 1285 1287
Essell JH, Thompson JM, Harman GS et al. Marked increase in veno-occlusive disease of the liver associated with methotrexate use for graft-versus-host disease prophylaxis in patients receiving busulphan/cyclophosphamide Blood 1992 79: 2784 2788
Holden SA, Teicher BA, Robinson MF et al. Antifolates can potentiate topoisomerase II inhibitors in vitro and in vivo Cancer Chemother Pharmacol 1995 36: 165 171
Workman DL, Clancy J Jr . Phenotypic analysis of pulmonary perivascular mononuclear infiltrates that occur as a direct result of acute lethal graft-versus-host disease describes the onset of interstitial pneumonitis Am J Pathol 1995 147: 1350 1360
Porter D, Boddy A, Thomas H et al. Etoposide phosphate infusion with therapeutic drug monitoring in combination with carboplatin dosed by area under the curve: a cancer research campaign phase I/II committee study Semin Oncol 1996 23: 34 44
Soni N, Meropol NJ, Pendyala L et al. Phase I and pharmacokinetic study of etoposide phosphate by protracted venous infusion in patients with advanced cancer J Clin Oncol 1997 15: 766 772
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ritchie, D., Szer, J., Roberts, A. et al. A phase I dose-escalation study of etoposide continuous infusion added to busulphan/cyclophosphamide as conditioning prior to autologous or allogeneic stem cell transplantation. Bone Marrow Transplant 30, 645–650 (2002). https://doi.org/10.1038/sj.bmt.1703698
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703698