Abstract
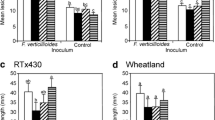
Volatiles released from glucosinolates (predominantly 2-propenyl glucosinolate) in tissues of Brassica juncea and Wasabi japonica are toxic to the blackleg fungus, Leptosphaeria maculans, in vitro. However, there is no relationship between blackleg resistance (in stems or cotyledons) and the level of 2-propenyl glucosinolate in seed or shoot tissue of B. juncea. A representative isolate from each of two classes of Australian isolates of L. maculans was tested: those which cause stem cankers on both B. napus and B. juncea (e.g. isolate MI), and those which cause stem cankers on B. napus but not on B. juncea (e.g, isolate C13). Even in lines of B. juncea in which levels of 2-propenyl glucosinolate were as low as those in B. napus, isolate C13 was still unable to cause stem cankers. This suggests that either 2-propenyl glucosinolate does not play a major role in blackleg disease resistance, or that isolates of L. maculans able to attack B. juncea can detoxify or evade the toxic effects of hydrolysis products from this glucosinolate.
Similar content being viewed by others
References
Angus, J.F., Gardner, P.A., Kirkegaard, J.A, and Desmarchelier J.M. (1994) — Biofumigation: Isothiocyanates released from Brassicn roots inhibit growth of the take-all fungus. Plant and Soil 162: 107–112.
Appelqvist, L-A. and Josefsson, E. (1967) — Quantitative determination of isothiocyanates and oxazolidinethiones in rape and turnip rape. Journal of the Science of Food and Agriculture 18: 510–519.
Ballinger, D.J. and Salisbury, P.A. (1996) — Seedling and adult plant evaluation of race variability in Leptosphaeria maculans on Brassica species in Australia. Australian Journal of Experimenta1 Agriculture 36: 485–488.
Bodnaryk, R.P. (1997) — Will low-glucosinolate lines of the mustards Brassica juncea and Sinapis alba be vulnerable to insect pests. Canadian Journal of plant Science 77: 283–287.
Bones, A.M. and Rossiter, J.T. (1996) — The myrosinaseglucosinolate system, its organisation and biochemistry. Physiologia Plantarum 97: 194–208.
Bowyer, P., Clarke, B.R., Lunness, P., Daniels, M.J. and Osbourn, A. (1995) — Host range of a plant pathogenic fungus determined by a saponin detoxifying enzyme. Science 267: 371 -374.
Brown. P.D. and Morra, M.J. (1997) — Control of soilborne plant pests using glucosinolate-containing plants. Advances in Agronomy 61: 167–231.
Chen. C.-Y. and Howlett, B.J. (1996) — Rapid necrosis of guard cells is associated with the arrest of fungal growth in the leaves of Indian mustard (Brassica juncea) inoculated with avirulent isolates of Leptosphaeria maculans. Physiological and Molecular Plant Pathology 48: 73–81.
Chen, C.-Y.. Plummer, K.M. and Howlett, B.J. (1996) — Ability of an Leptosphaeria maculans isolate to form stem cankers on Indian mustard (Brassica juncea) segregates as a single locus. European Journal of Plant Pathology 102: 349–352.
Fenwick, G.R.. Heaney, R.K. and Mullin, W.J. (1983) — Glucosinolates and their breakdown products in food and food plants. CRC Critical Reviews in Food Science and Nutrition 18: 123–201.
Giamoustaris, A, and Mithen, R. (1997) — Glucosinolates and disease resistance in oilseed rape (Brassica napus ssp. oleifera). Plant Pathology 46: 271–275.
Gugel, R.K. and Petrie, G.A. (1992) — History, occurrence, impact, and control of blackleg of rapeseed. Canadian Journal of plant Pathology 14: 36–45.
Kirkegaard: J.A. and Sarwar, M. (1998) — Biofumigation potential of brassicas. I. Variation in glucosinolate profiles of diverse field-grown brassicas. Plant and Soil 201: 71–89,
Kirkegaard, J.A., Wong, P.T.W. And Desmarchelier, J.M. (1996) — vitro suppression of fungal root pathogens of cereals by Brassica tissues. Plant Pathology 45: 593–603
Manici, L.M., Lazzeri, L. and Palmieri, S. (1997) — In vitro fungitoxic activity of some glucosinolates and their enzyme-derived products to ward plant pathogenic fungi. Journal of Agricultural and Food Chemistry 45: 2768–2773.
Mayton. H.S., Olivier, C., Vaughn, S.F. and Loria, R. (1996) — Correlation of fungicidal activity of Brassica species with ally1 isothiocyanate production in macerated leaf tissue. Phytopathology 86: 267–271.
Mithen, R.F., Lewis, B.G. and Fenwick, G.R. (1986) — In vitro activity of glucosinolates and their products against Leptosphaeria maculans. Transactions of the British Mycological Society 87: 433–440.
Pedras, M.S.C. and Khan, A.Q. (1996) — Biotransformation of the Brassica phytoalexin brassicanal A by the blackleg fungus. Journal of Agriculture and Food Chemistry 44: 3403–3407.
Pinkerton, A., Randall, P.J. and Wallace, P.A. (1993) — Determination of total glucosinolates in oilseed rape by X-ray spectrometric analysis for oxidised sulphur (S6+). Journal of the Science of Food and Agriculture 61: 79–86.
Plummer, K.M., Dunse, K., and Howlett, B.J. (1994) — Non-aggressive strains of the blackleg fungus Leptosphaeria maculans are present in Australia and can be distinguished from aggressive strains by molecular analysis. Australian Journal of Botany 42: 1–8.
Rimmer, S.R. and van den Berg, C.G.J. (1992) — Resistance of oilseed Brassica spp. to blackleg caused by Leptosphaeria maculans. Canadian Journal of plant Pathology 14: 56–66.
Sarwar, M., Kirkegaard, J.A., Wong. P.T.W., and Desmarchelier, J.M. (1998) — Biofumigation potential of brassicas. III. In vitro toxicity of isothiocyanates to soil-borne fungal pathogens. Plant and Soil 201: 103–112.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sexton, A.C., Kirkegaard, J.A. & Howlett, B.J. Glucosinolates in Brassica juncea and resistance to Australian isolates of Leptosphaeria maculans, the blackleg fungus. Australasian Plant Pathology 28, 95–102 (1999). https://doi.org/10.1071/AP99017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1071/AP99017