-
PDF
- Split View
-
Views
-
Cite
Cite
Anne Sacré, François Jouret, Daniel Manicourt, Olivier Devuyst, Topiramate induces type 3 renal tubular acidosis by inhibiting renal carbonic anhydrase, Nephrology Dialysis Transplantation, Volume 21, Issue 10, October 2006, Pages 2995–2996, https://doi.org/10.1093/ndt/gfl251
Close - Share Icon Share
Sir,
We report a case of mixed (type 3) renal tubular acidosis (RTA) associated with the anti-convulsant drug topiramate used for migraine prophylaxis. A 47-year-old woman treated with topiramate (150 mg/day) since 12 months for invalidating migraine was referred for a metabolic acidosis evidenced in a routine blood sampling. She complained of muscle weakness and bone pain. Clinical examination showed weight loss (−6 kg in 4 months) and joint sensitivity. Blood analyses confirmed a mild hyperchloraemic metabolic acidosis (plasma 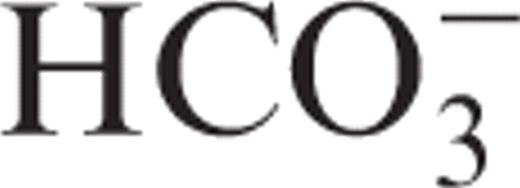 , 19.0 mEq/l; plasma anion gap, 8 mEq/l) with hypokalaemia (3.2 mEq/l) and normal renal function. Plasma
, 19.0 mEq/l; plasma anion gap, 8 mEq/l) with hypokalaemia (3.2 mEq/l) and normal renal function. Plasma 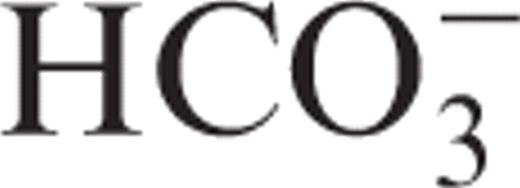 before topiramate administration was normal. The urine pH was 6.0 with a positive urinary anion gap (UAG, +39 mEq/day), positive β 2 -microglobulinuria (2.1 mg/l) and a low urinary citrate excretion (0.7 mM/24 h). Serology and auto-immune markers were negative. Echography showed no kidney calcifications. The hyperchloraemic metabolic acidosis with normal glomerular filtration rate (GFR) and positive UAG indicates RTA, associated with both proximal (β 2 -microglobulinuria) and distal (hypocitraturia) tubular dysfunction. The normal blood analyses before the introduction of topiramate suggests that these disorders result from tubular dysfunction caused by inhibition of carbonic anhydrase (CA).
before topiramate administration was normal. The urine pH was 6.0 with a positive urinary anion gap (UAG, +39 mEq/day), positive β 2 -microglobulinuria (2.1 mg/l) and a low urinary citrate excretion (0.7 mM/24 h). Serology and auto-immune markers were negative. Echography showed no kidney calcifications. The hyperchloraemic metabolic acidosis with normal glomerular filtration rate (GFR) and positive UAG indicates RTA, associated with both proximal (β 2 -microglobulinuria) and distal (hypocitraturia) tubular dysfunction. The normal blood analyses before the introduction of topiramate suggests that these disorders result from tubular dysfunction caused by inhibition of carbonic anhydrase (CA).
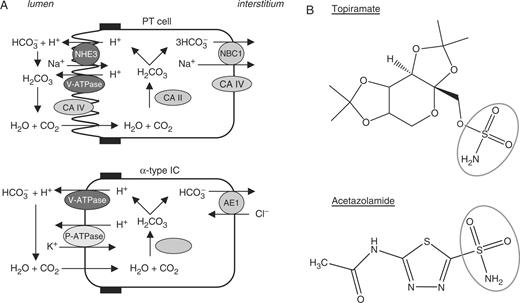
Type II CA (CAII) plays an essential role in the reabsorption of ultrafiltered 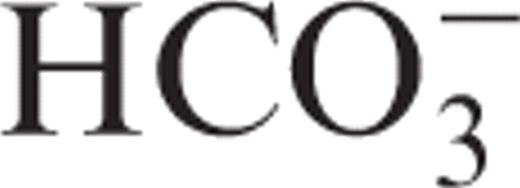 by the proximal tubule (PT) and the net urinary acidification by α-type intercalated cells of the distal nephron ( Figure 1 ). It also provides the H + for extracellular acidification during bone resorption by osteoclasts. Deficiency in CAII, either inherited (marble brain disease; OMIM #259730) or acquired (e.g. acetazolamide-induced), is associated with a mixed proximal and distal RTA (type 3 RTA), without evidence for generalized PT dysfunction [ 1 , 2 ]. Topiramate is a sulfamate-substituted monosaccharide that is structurally related to acetazolamide ( Figure 1 ). As such, it inhibits CAII more effectively than other isoforms [ 3 ]. Long-term inhibition of CAII by topiramate may thus disturb the homeostasis of both the proximal and distal tubules, leading to a mixed form of RTA. By extension, it may also affect bone resorption, leading to bone pain [ 4 ].
by the proximal tubule (PT) and the net urinary acidification by α-type intercalated cells of the distal nephron ( Figure 1 ). It also provides the H + for extracellular acidification during bone resorption by osteoclasts. Deficiency in CAII, either inherited (marble brain disease; OMIM #259730) or acquired (e.g. acetazolamide-induced), is associated with a mixed proximal and distal RTA (type 3 RTA), without evidence for generalized PT dysfunction [ 1 , 2 ]. Topiramate is a sulfamate-substituted monosaccharide that is structurally related to acetazolamide ( Figure 1 ). As such, it inhibits CAII more effectively than other isoforms [ 3 ]. Long-term inhibition of CAII by topiramate may thus disturb the homeostasis of both the proximal and distal tubules, leading to a mixed form of RTA. By extension, it may also affect bone resorption, leading to bone pain [ 4 ].
The main adverse effects of topiramate include ataxia, impaired concentration, confusion, dizziness, fatigue and weight loss. An increased incidence of nephrolithiasis has been reported, in relation with defective urinary acidification and hypocitraturia [ 5 ]. Although non-anion gap metabolic acidosis was initially considered as a rare complication of topiramate, it may actually be more frequent than expected, particularly in children [ 5 , 6 ]. The plasma 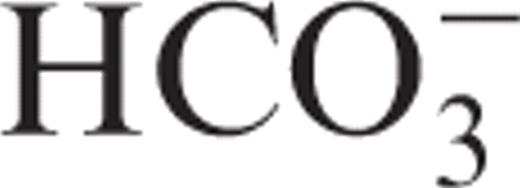 levels return to normal when the drug is discontinued [ 5 ]. Since the use of topiramate has recently been extended to the treatment of migraine, clinicians should be aware of its potential effect on renal tubular functions and monitor plasma
levels return to normal when the drug is discontinued [ 5 ]. Since the use of topiramate has recently been extended to the treatment of migraine, clinicians should be aware of its potential effect on renal tubular functions and monitor plasma 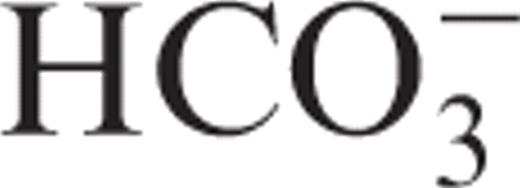 and K + concentrations as long as the drug is administered.
and K + concentrations as long as the drug is administered.
Conflict of interest statement. None declared.
Role of CA isoforms in the kidney and structure of topiramate. ( A ) Pathways involved in 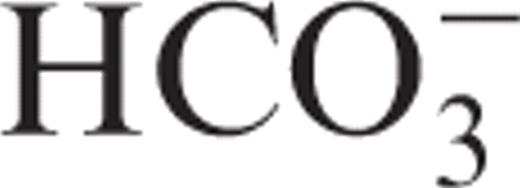 reabsorption and H + secretion in PT cells and α-type intercalated cells of the collecting ducts, respectively. Cytosolic type II CA is distributed in both cell types. Note that PT cells also express type IV CA in the apical brush border and basolateral membrane. ( B ) Comparative structure of topiramate and acetazolamide. Both molecules present a sulfonamide group responsible for the inhibition of CA activity.
reabsorption and H + secretion in PT cells and α-type intercalated cells of the collecting ducts, respectively. Cytosolic type II CA is distributed in both cell types. Note that PT cells also express type IV CA in the apical brush border and basolateral membrane. ( B ) Comparative structure of topiramate and acetazolamide. Both molecules present a sulfonamide group responsible for the inhibition of CA activity.
References
DuBose TD. (
Dodgson SJ, Shank RP, Maryanoff BE. (
Lehenkari P, Heutunen TA, Laitala-Leinonen T, Tuukkanen J, Vaananen HK. (
Lamb EJ, Stevens PE, Nashef L. (





Comments