Abstract
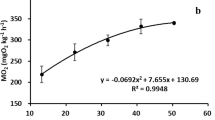
The effects of temperature changes on the endogenous rhythm of oxygen consumption in the Japanese flounder Paralichthys olivaceus which were acclimated to 3, 14 or 20°C in advance were investigated. The oxygen consumption rate (OCR) of individuals (total length >28 cm) that were acclimated to 3°C, maintained a relatively low value of 6.9±2.3 mL (mean±SD) O2/kg ww (wet weight)/h till 5.8°C, which is considered a ‘minimum’ rate. Thereafter, the OCR gradually increased up to 18.7 mL O2/kg ww/h at the temperature of 10.6°C. The OCR of individuals acclimated to 14°C stabilized with 124.2±24.1 mL O2/kg ww/h, when tested at the relatively constant temperature of 14.4±0.17°C. Maximum entropy spectral analysis revealed that the peak OCR occurred at 23.9 h intervals, which could be a circadian rhythm. The mean OCR showed a peak at 06.00–08.00 hours. For the temperature above 25°C in which the fish were acclimated to 20°C, the rhythmic patterns of OCR were observed until the experimental temperature reached 26.4°C. For the temperature above 28.4°C, the metabolic activity rhythm of the OCR was dampened and then the OCR abruptly rose again. It is assumed that the oscillator of the endogenous rhythm lost the governing of normal metabolic activity in Japanese flounders above 28.4°C. These results suggest that the temperatures of around 5.8, 26.4 and 28.4°C are the ‘lower limit, temperature’, ‘upper incipient lethal temperature’, and the ‘critical thermal maximum’, respectively, for the endogenous rhythm of oxygen consumption in the Japanese flounder.
Similar content being viewed by others
References
Masuda H, Amaoka K, Araga C, Uyeno T, Yoshino K. The Fishes of the Japanese Archipelago. Tokai University Press, Tokyo, Japan, 1984; 437.
Chyung MK. The Fishes of Korea. Il-Ji Sa Press. Seoul. 1977; 559–560.
Yasunaga Y, Koshiishi Y. Basic studies of problems on the propagation of plaice, Paralichthysolivaceus. I. Acclimation to low salinity, feeding and gathering behaviour. Bull. Jap. Sea Reg. Fish. Res. Lab. 1980; 31: 17–31.
Kubosho T, Ecological data of marine organisms. Nippon Suisan Shigenhohokyogai (Asso Jap Fish Res Protect), 1981; 190–195.
Iwata N, Honda H, Kiyono M. Effects of temperature on growth of Japanese flounder (Paralichthys. oilvaceus). In: Carrillo M, Dahle L, Morales J, Sorgeloos, P, Svennevig N, Wyban J (eds). From-Discovery-to-Commercialization. Oostende Belgium European Aquacult. Soc., Belgium, 1993; 19: 391.
Iwata N, Kikuchi K, Honda H, Kiyono M, Kurokura H. Effects of temperature on the growth of Japanese flounder. Fish. Sci. 1994; 60: 527–531.
Iwata N, Kikuchi K, Kurokura H. Growth of Japanese flounder Paralichthys olivaceus at different temperatures. Isr. J. Aquacult. Bamidgeh 1995; 47: 178–184.
Liu H, Sakurai Y, Munehara H, Shimazaki K. Diel rhythms of oxygen consumption and activity level of juvenile flounder Paralichthys olivaceus. Fish. Sci. 1997; 63: 655–658.
Kim WS, Jeon JK, Lee SH, Huh HT. Effects of pentachlorophenol (PCP) on the oxygen consumption rate of the river puffer fish, Takifugu obscurus. Mar. Ecol. Prog. Series 1996; 143: 9–14.
Kim WS, Kim JM, Kim MS, Park CW, Huh HT. Effects of sudden changes in salinity on endogenous rhythm of the spotted sea bass Lateolabrax sp. Mar. Biol. 1998; 131: 219–225.
Kim WS, Huh HT, Huh SH, Lee TW. Effect of salinity on endogenous rhythm of the Manila clam Ruditapes philippinarum (Bivalvia: Veneridae), Mar. Biol. 2001; 138: 157–162.
Kim WS, Yoon SJ, Moon HT, Lee TW. Effects of water temperature changes on the endogenous and exogenous rhythms of oxygen consumption in glass eels Anguilla japonica. Mar. Ecol. Prog. Series 2002; 243: 209–216.
Brett JR. Energetic responses of salmon to temperature. A study of some thermal relations in the physiology and fresh water ecology of sockeye salmon (O. nerka). Am. Zool. 1971: 11: 99–113.
Reynolds WW, Casterlin ME. The role of temperature in the environmental physiology of fishes. In: Ali MA (ed). Environmental Physiology of Fishes. Plenum Press, New York. 1980; 497–518.
Van Dijk PLM, Tesch C, Hardewig I, Pörtner HO. Physiological disturbances at critically high temperatures: a comparison between stenothermal Antarctic and eurythermal temperate eelpouts (Zoarcidae). J. Exp. Biol. 1999; 202: 3611–3621.
Morgan MJ. Low temperature tolerance of American plaice in relation to declines in abundance. Trans. Am. Fish. Soc. 1992; 121: 399–402.
Evans DO. Metabolic thermal compensation by rainbow trout: effects on standard metabolic rate and potential usable power. Trans. Am. Fish. Soc. 1990; 119: 585–600.
Lee RM, Rinne JN. Critical thermal maxima of five trout species in the southwestern United States. Trans. Am. Fish. Soc. 1980; 109: 632–635.
Jobling M. Temperature tolerance and the final preferendum — rapid methods for the assessment of optimum growth temperatures. J. Fish Biol. 1980; 19: 439–455.
Rajaguru S. Critical thermal maximum of seven estuarine fishes. J. Thermal Biol. 2002; 27: 125–128.
Tsuchida S. The relationship between upper temperature tolerance and final preferendum of Japanese marine fish. J. Thermal Biol. 1995; 20: 35–41.
Fernandes MN, Rantin FT. Respiratory responses of Oreochromis niloticus (Pisces, Cichlidae) to environmental hypoxia under different thermal conditions. J. Fish Biol. 1989; 35: 509–519.
Gasca-Leyva JFE, Martinez-Palacios CA, Ross LG. The respiratory requirements of Macrobrachium acanthurus (Weigman) at different temperatures and salinities. Aquaculture 1991; 93: 191–197.
Mwangangi DW, Mutungi G. The effects of temperature acclimation on the oxygen consumption and enzyme activity of red and white muscle fibbers isolated from the tropical freshwater fish Oreochromis niloticus. J. Fish Biol. 1994; 44: 1033–1043.
Fernandes MN, Barrionuevo WR, Rantin FT. Effects of thermal stress on respiratory responses to hypoxia of a South American Prochilodontid fish. Prochilodus scrofat. J. Fish Biol. 1995; 46: 123–133.
Mark FC, Bock C, Pörtner HO. Oxygen-limited thermal tolerance in Antarctic fish investigated by MRI and 31P-MRS. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002; 283: R1254-R1262.
Palmer JD. The Biological Rhythms and Clocks of Intertidal Animals. Oxford University Press. Oxford. 1995.
Gibson RN. Hydrostatic pressure and the rhythmic behaviour of intertidal marine fishes. Trans. Am. Fish. Soc. 1984; 113: 479–483.
Jeon LK, Min KS, Lee JM, Kim KS, Son MH. Optimal stocking density for olive flounder, Paralichthys olivaceus, rearing in tanks. Bull. Natl. Fish. Res. Dev. Agency Korea 1993; 48: 57–70.
Cox DK. Effects of three heating rates on the critical thermal maximum of bluegill. In: Gibbons JW, Sharitz RR (eds). Thermal ecology. Proc. Symp. Augusta, Georgia May 3–5. 1973. Tech. Infor. Center, US Atomic Energy Commission. Virginia. 1974; 158–163.
Kim WS, Yoon SJ, Gil JW. Critical thermal maximum (CTM) of cultured black rockfish, Sebastes schegeli. J. Fish. Soc. Tec. 2003; 6: 59–63.
Weiss RF. The solubility of nitrogen. oxygen, and argon in water and seawater. Deep-Sea Res. 1970; 17: 721–735.
Forstner H, Gnaiger E. Polarographic Oxygen Sensors. Springer-Verlag. Berlin. 1983.
Kim WS, Kim JM, Yi SK, Huh HT. Endogenous circadian rhythm in the rate of oxygen consumption of the river puffer fish, Takifugu obscurus. Mar. Ecol. Prog. Series 1997; 153: 293–298.
Kim WS, Huh HT, Lee JH, Rumohr H, Koh CH. Endogenous circatidal rhuthm in the Manila clam Ruditapes philippinarum (Bivalvia: Veneridae). Mar. Biol. 1999; 134: 107–112.
Yoon SJ, Kim JK, Myung JG, Kim WS. Comparsion of oxygen consumption patterns between wild and cultured black rockfish. Sebastes schlegeli. Fish. Sci. 2003; 69: 43–49.
Dowse HB, Ringo JM. The search for hidden periodicities in biological time series revisited. J. Theor. Biol. 1989; 139: 487–515.
Schmidt-Nielsen K. Animal Physiology Adaptation and Environment. 5th edn. Cambridge University Press, Cambridge. 1997; 218–221.
Borsook D, Woolf CJ, Vellet AD. Temperature acclimation and learning in fish. Experientia 1978; 34: 70–71.
Davis KB, Parker NC. Physiological stress in stripped bass: effect of acclimation temperature. Aquaculture 1990; 91: 349–358.
Langford TEL. Ecological Effects of Thermal Discharges. Elsevier Applied Science. London. 1990; 101–137.
Itazawa Y, Matsumoto T, Kanda T. Group effects on physiological and ecological phenomena in fish-I. Group effect on the oxygen consumption of the Rainbow trout and the Medaka. Nippon Suisan Gakkaishi, 1978; 44: 965–969.
McCauley RW, Huggins NW. Ontogenetic and thermal seasonal effects on thermal prefernda of fish. Am. Zoo 1979; 19: 267–271.
Barrionuevo WR, Fernandes MN. Critical thermal maxima and minima for curimbata, Prochilodus scrofa Steindachner, of two different sizes. Aquacult. Res. 1995; 26: 447–450.
Woiwode JG, Adelman IR. Effects of starvation, oscillating temperature, and photoperiod on the critical thermal maximum of hybrid strippedx white bass. J. Thermal Biol. 1992; 17: 271–275.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, W.S., Yoon, SJ., Kim, J.M. et al. Effects of temperature changes on the endogenous rhythm of oxygen consumption in the Japanese flounder paralichthys olivaceus . Fish Sci 71, 471–478 (2005). https://doi.org/10.1111/j.1444-2906.2005.00990.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1444-2906.2005.00990.x