Abstract
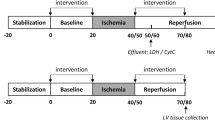
Effects of 10-(6′-plastoquinonyl) decyltriphenylphosphonium (SkQ1) and 10-(6′-plastoquinonyl) decylrhod-amine 19 (SkQR1) on rat models of H2O2- and ischemia-induced heart arrhythmia, heart infarction, kidney ischemia, and stroke have been studied ex vivo and in vivo. In all the models listed, SkQ1 and/or SkQR1 showed pronounced protective effect. Supplementation of food with extremely low SkQ1 amount (down to 0.02 nmol SkQ1/kg per day for 3 weeks) was found to abolish the steady heart arrhythmia caused by perfusion of isolated rat heart with H2O2 or by ischemia/reperfusion. Higher SkQ1 (125–250 nmol/kg per day for 2–3 weeks) was found to decrease the heart infarction region induced by an in vivo ischemia/reperfusion and lowered the blood levels of lactate dehydrogenase and creatine kinase increasing as a result of ischemia/reperfusion. In single-kidney rats, ischemia/reperfusion of the kidney was shown to kill the majority of the animals in 2–4 days, whereas one injection of SkQ1 or SkQR1 (1 μmol/kg a day before ischemia) saved lives of almost all treated rats. Effect of SkQR1 was accompanied by decrease in ROS (reactive oxygen species) level in kidney cells as well as by partial or complete normalization of blood creatinine and of some other kidney-controlled parameters. On the other hand, this amount of SkQ1 (a SkQ derivative of lower membrane-penetrating ability than SkQR1) saved the life but failed to normalize ROS and creatinine levels. Such an effect indicates that death under conditions of partial kidney dysfunction is mediated by an organ of vital importance other than kidney, the organ in question being an SkQ1 target. In a model of compression brain ischemia/reperfusion, a single intraperitoneal injection of SkQR1 to a rat (1 μmol/kg a day before operation) effectively decreased the damaged brain area. SkQ1 was ineffective, most probably due to lower permeability of the blood-brain barrier to this compound.
Similar content being viewed by others
Abbreviations
- AI:
-
area of infarction
- CK:
-
creatine kinase
- DCF:
-
5-(-6)-chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate
- ICF:
-
intensity of the (heart) contractile function
- LDH:
-
lactate dehydrogenase
- LV:
-
the heart left ventricle
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium
- RA:
-
risk area
- ROS:
-
reactive oxygen species
- SkQs:
-
cationic derivatives of plastoquinone or methyl plastoquinone
- SkQ1:
-
10-(6′-plastoquinonyl) decyltriphenylphosphonium
- SkQR1:
-
10-(6′-plastoquinonyl) decylrhodamine 19
- TMRE:
-
tetramethylrhodamine ethyl ester
- TTC:
-
2,3,5-triphenyltetrazolium chloride
References
Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L. E., Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Yu., Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A., Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A. A., Pletjushkina, O. Yu., Pustovidko, A. V., Roginsky, V. A., Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I., Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V., Sviryaeva, I. V., Tashlitsky, V. N., Vassiliev, J. M., Vyssokikh, M. Yu., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P. (2008) Biochemistry (Moscow), 73, 1273–1287.
Pisarenko, O. I., Studneva, I. M., Serebriakova, L. I., Tskitishvili, O. V., and Timoshin, A. A. (2005) Kardiologiya, 45, 37–44.
Watanabe, S., Hoffman, J. R., Craik, R. L., Hand, P. J., Croul, S. E., Reivich, M., and Greenberg, J. H. (2001) Stroke, 32, 2615–2623.
Kundrotiene, J., Wagner, A., and Liljequist, S. (2002) J. Neurotrauma, 19, 69–84.
Andersson, B., Bjelke, B., and Sykova, E. (2006) Physiol. Res., 55, 339–348.
Andersson, D., Wu, X., Bjelke, D., and Sekova, E. (2004) J. Neurosci. Res., 77, 901–912.
Andrews, R., and Muto, R. (1992) Neurol. Res., 14, 12–18.
Skulachev, V. P., Bakeeva, L. E., Chernyak, B. V., Domnina, L. V., Minin, A. A., Pletjushkina, O. Y., Saprunova, V. B., Skulachev, I. V., Tsyplenkova, V. G., Vasiliev, J. M., Yaguzhinsky, L. S., and Zorov, D. B. (2004) Mol. Cell. Biochem., 256/257, 341–358.
Plotnikov, E. Y., Kazachenko, A. V., Vyssokikh, M. Y., Vasileva, A. K., Tcvirkun, D. V., Isaev, N. K., Kirpatovsky, V. I., and Zorov, D. B. (2007) Kidney Int., 72, 1493–1502.
Serviddio, G., Romano, A. D., Gesualdo, L., Tamborra, R., Di Palma, A. M., Rollo, T., Altomare, E., and Vendemiale, G. (2008) Nephrol. Dial. Transplant., 23, 1504–1512.
McCord, J. M. (1985) N. Engl. J. Med., 312, 159–163.
Baud, L., and Ardaillou, R. (1993) Br. Med. Bull., 49, 621–629.
Moro, M. A., Almeida, A., Bolanos, J. P., and Lizasoain, I. (2005) Free Rad. Biol. Med., 39, 1291–1304.
Webster, K. A. (2007) Trends Pharmacol. Sci., 28, 492–499.
Goldhaber, J. I., and Liu, E. (1994) J. Physiol., 477, 135–147.
Okabe, E., Tsujimoto, Y., and Kobayashi, Y. (2000) Antioxid. Redox. Signal, 2, 47–54.
Meissner, A., and Morgan, J. P. (1995) Am. J. Physiol., 268, 100–111.
Kojima, S., Wikman-Coffelt, J., Wu, S. T., and Parmley, W. W. (1994) Am. J. Physiol., 266, 1473–1484.
Camello-Almaraz, M. C., Pozo, M. J., Murphy, M. P., and Camello, P. J. (2006) J. Cell Physiol., 206, 487–494.
Adlam, V. J., Harrison, J. C., Porteous, C. M., James, A. M., Smith, R. A. J., Murphy, M. P., and Sammut, I. A. (2005) FASEB J., 19, 1088–1095.
Taggart, D. P., Jenkins, M., Hooper, J., Hadjinikolas, L., Kemp, M., Hue, D., and Bennett, G. (1996) Ann. Thorac. Surg., 61, 829–833.
Kalenikova, E. I., Gorodetskaya, E. A., Kolokolchikova, E. G., Shashurin, D. A., and Medvedev, O. S. (2007) Biochemistry (Moscow), 72, 332–338.
Verma, D. D., Hartner, W., Levchenko, T., and Torchilin, V. (2007) J. Mol. Cell. Cardiol., 42, 214–215.
Rokitskaya, T. I., Klishin, S. S., Severina, I. I., Fedorov, I. I., Tashlitsky, V. N., Antonenko, Y. N., and Skulachev, V. P. (2008) J. Membr. Biol., 222, 141–149.
Neroev, V. V., Archipova, M. M., Bakeeva, L. E., Fursova, A. Zh., Grigorian, E. N., Grishanova, A. Yu., Iomdina, E. N., Ivashchenko, Zh. N., Katargina, L. A., Khoroshilova-Maslova, I. P., Kilina, O. V., Kolosova, N. G., Kopenkin, E. P., Korshunov, S. S., Kovaleva, N. A., Novikova, Yu. P., Philippov, P. P., Pilipenko, D. I., Robustova, O. V., Saprunova, V. B., Senin, I. I., Skulachev, M. V., Sotnikova, L. F., Stefanova, N. A., Tikhomirova, N. K., Tsapenko, I. B., Shchipanova, A. I., Zinovkin, R. A., and Skulachev, V. P. (2008) Biochemistry (Moscow), 73, 1317–1328.
Anisimov, V. N., Bakeeva, L. E., Egormin, P. A., Filenko, O. F., Isakova, E. F., Manskikh, V. N., Mikhelson, V. M., Panteleeva, A. A., Pasyukova, E. G., Pilipenko, D. I., Piskunova, T. S., Popovich, I. G., Roshchina, N. V., Rybina, O. Yu., Samoylova, T. A., Saprunova, V. B., Semenchenko, A. V., Skulachev, M. V., Spivak, I. M., Tsybul’ko, E. A., Tyndyk, M. L., Vyssokikh, M. Yu., Yurova, M. N., Zabezhinsky, M. A., and Skulachev, V. P. (2008) Biochemistry (Moscow), 73, 1329–1342.
Skulachev, V. P. (2007) Biochemistry (Moscow), 72, 1385–1396.
Agapova, L. S., Chernyak, B. V., Domnina, L. V., Dugina, V. B., Efimenko, A. Yu., Fetisova, E. K., Ivanova, O. Yu., Kalinina, N. I., Khromova, N. V., Kopnin, B. P., Kopnin, P. B., Korotetskaya, M. V., Lichinitser, M. R., Lukashov, A. L., Pletjushkina, O. Yu., Popova, E. N., Skulachev, M. V., Shagieva, G. S., Stepanova, E. V., Titova, E. V., Tkachuk, V. A., Vasiliev, Y. M., and Skulachev, V. P. (2008) Biochemistry (Moscow), 73, 1300–1316.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2008, Vol. 73, No. 12, pp. 1607–1621.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Bakeeva, L.E., Barskov, I.V., Egorov, M.V. et al. Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 2. Treatment of some ROS- and Age-related diseases (heart arrhythmia, heart infarctions, kidney ischemia, and stroke). Biochemistry Moscow 73, 1288–1299 (2008). https://doi.org/10.1134/S000629790812002X
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S000629790812002X