Abstract
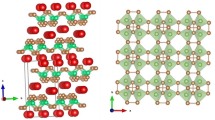
The crystal structure of the low-temperature form of K3PO4 has been determined for the first time using neutron diffraction (Rietveld method) and Raman spectroscopy: orthorhombic cell (sp. gr. Pnma, Z = 4), lattice parameters a = 1.12377(2) nm, b = 0.81046(1) nm, c = 0.59227(1) nm. The structure is made up of isolated [PO4] tetrahedra, with the potassium ions in between.
Similar content being viewed by others
References
Kolsi, A.W., Stabilisation des formes haute temperature des phosphates Na3PO4 et K3PO4 par des cations divalents, Rev. Chim. Miner., 1976, vol. 13, no. 5, pp. 416–421.
Jungowska, W. and Znamierowska, T., The System LaPO4-K3PO4, J. Solid State Chem., 1991, vol. 95, no. 2, pp. 265–269.
Hoppe, R. and Seyfert, H.M., Zur Kenntnis wasserfreier Orthophosphate der hoheren Alkalimetalle: K3PO4, Rb3PO4, Cs3PO4, Z. Naturforsch., B: Anorg. Chem., Org. Chem., 1973, vol. 28, no. 2, pp. 507–508.
Norbert, A., Contribution à la chimie des orthophosphates de sodium et de potassium, Rev. Chim. Miner., 1966, vol. 3, no. 1, pp. 1–17.
Mosin, D.N., Marks, E.A., Burmakin, E.I., et al., Electrical Conductivity of Potassium, Rubidium, and Cesium Orthophosphates, Elektrokhimiya, 2001, vol. 37, no. 8, pp. 1005–1007.
Burmakin, E.I. and Shekhtman, G.Sh., Electrical Conductivity of K3−2x MxPO4 (M = Ca, Sr, Ba) Solid Solutions, Elektrokhimiya, 2004, vol. 40, no. 2, pp. 238–241.
Burmakin, E.I. and Shekhtman, G.Sh., Electrical Conductivity of Potassium Orthophosphate Modified with Tetravalent Cations, Elektrokhimiya, 2002, vol. 38, no. 12, pp. 1447–1451.
Burmakin, E.I., Mosin, D.N., and Shekhtman, G.Sh., Potassium-Ion Conduction in the Systems K3−x P1−x AxO4 (A = S, Sr, Mo, W), Elektrokhimiya, 2001, vol. 37, no. 11, pp. 1392–1396.
Nord, A. and Kierkegaard, P., Crystal Chemistry of Some Anhydrous Divalent-Metal Phosphates, Chem. Scr., 1980, vol. 15, no. 1, pp. 27–39.
Altomare, A., Burla, M.C., Carrozzini, B., et al., EXPO: A Program for Full Powder Pattern Decomposition and Crystal Structure Solution, J. Appl. Crystallogr., 1999, vol. 32, no. 3, pp. 339–340.
Rietveld, H.M., A Profile Refinement Method for Nuclear and Magnetic Structures, J. Appl. Crystallogr., 1969, vol. 2, no. 2, pp. 65–71.
Larson, A.C. and Von Dreele, R.B., General Structure Analysis System (GSAS), Los Alamos National Laboratory LAUR, 2004.
Rodriges-Carvajal, J., The Programs for Rietveld Refinement, Physica. B (Amsterdam), 1993, vol. 192, no. 1, p. 55.
Zlokazov, V.B. and Chernyshev, V.V., MRIA—A Program for a Full Profile Analysis of Powder Multiphase Neutron Diffraction Time-of-Flight (Direct and Fourier) Spectra, J. Appl. Crystallogr., 1992, vol. 25, no. 3, pp. 447–452.
Shannon, R.D. and Prewitt, C.T., Effective Ionic Radii in Oxides and Fluorides, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1969, vol. 25, no. 5, pp. 925–946.
Author information
Authors and Affiliations
Additional information
Oiginal Russian Text © V.I. Voronin, Yu.S. Ponosov, I.F. Berger, N.V. Proskurnina, V.G. Zubkov, A.P. Tyutyunnik, S.N. Bushmeleva, A.M. Balagurov, D.V. Sheptyakov, E.I. Burmakin, G.Sh. Shekhtman, E.G. Vovkotrub, 2006, published in Neorganicheskie Materialy, 2006, Vol. 42, No. 8, pp. 1001–1006.
Rights and permissions
About this article
Cite this article
Voronin, V.I., Ponosov, Y.S., Berger, I.F. et al. Crystal structure of the low-temperature form of K3PO4 . Inorg Mater 42, 908–913 (2006). https://doi.org/10.1134/S0020168506080206
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1134/S0020168506080206