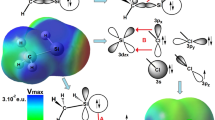
Abstract
This study is performed to detect the pentavalent silicon center in the structure of pentavalent-halosiliconate R–O–Si(CH3)3X– and halotrimethylsilyloxyfurane structures (X–TMSOF), (X = F–; Cl–; Br–) and (R = CH3–; CH2–CH3; –CH(CH3)2; –CH=CH2; C6H5–). DFT calculations at the B3LYP/6-31G(d) level are caried out to understand their structures and charge delocalization. These intermediates are obtained by attacking the silicon center in trimethyl-alkoxysilanes and trimethyl-silyloxyfurane with halogen ions X–. The results obtained show that the attack by F– generates more stable structures because of the Si–F bond strong. In the case of Br– and Cl–, the structure of intermediates appears as an interaction between the ions and the silicon center. NBO analysis shows that one of F– lone pairs takes part in the Si–F bond formation. However, the lone pairs of Br– and Cl– do not contribute to generate a real bonding.





Similar content being viewed by others
REFERENCES
M. A. Pigaliva, V. Elmanovich, M. Timnikov, and A. M. Muzafarov. Polym. Sci., Ser. B, 2016, 58, 270.
C. Zhang, P. Patschinski, S. D. Stephenson, R. Panisch, J. H. Wender, M. C. Holthausen, and H. Zipse. Phys. Chem. Chem. Phys., 2014, 16, 16650.
I. Mohamed and K. Subramani. E-J. Chem., 2008, 5, 136-143.
S. S. Sen and W. H. Roesky. Chem. Commun., 2018, 54, 5057.
V. V. Negrebetsky, S. N. Tandura, and Yu. I. Baukov. Russ. Chem. Rev., 2009, 78, 51.
S. R. Ghadwal, P. Kevin, D. Birger, G. P. Jones, and W. H. Roesky. Inorg. Chem., 2011, 50, 364.
F. Bitto, K. Kraushaar, U. Böhme, E. Brendler, and J. Wagler. Eur. J. Inorg. Chem., 2013, 35, 2962.
D. K. Onan, T. A. McPhail, H. C. Yoder, and W. R. Hillyard. J. Chem. Soc., Chem. Commun., 1978, 5, 210.
A. R. Bassindale, P. G. Taylor. In: The Chemistry of Organic Silicon Compounds / Eds. S. Patai, Z. Rappoport. John Wiley & Sons: Chichester, UK, 1989, Part 1, 839-892.
C. Marsden. J. Inorg. Chem., 1983, 22, 3178.
N. Kocher, J. Henn, B. Gostevskii, D. Kost, and I. Kalikhman. J. Am. Chem. Soc., 2004, 126, 5568.
C. W. So, H. W. Roesky, J. Magull, and R. B. Oswald. Angew. Chem., Int. Ed. Engl., 2006, 45(24), 3948.
C. A. Burkhard, E. G. Rochow, and H. S. Booth, J. Hartt. Chem. Rev., 1947, 41,149.
H. Gilman and G. E. Dunn. Chem. Rev., 1953, 52, 115.
W. P. Weber. Silicon Reagents for Organic Synthesis. Springer-Verlag: New York, 1983.
I. Fleming, A. Barbero, and D. Walter. Chem. Rev., 1997, 97, 2192.
C. Chuit, R. J. Corriu, C. Reye, and J. C. Young. Chem. Rev., 1993, 93, 1448.
D. Kost and I. Kalikhman. Hypervalent Silicon Compounds. In: The Chemistry of Organic Silicon Compounds / Eds. Z. Rappoport, Z. Rappoport and Y. Apeloig. Wiley: Chichester, 1998.
I. H. Krouse and P. G. Wenthold. J. Am. Soc. Mass Spectrom., 2005, 16, 697-707.
D. J. Hjdasz and R. R. Squires. J. Am. Chem. Soc., 1986, 108, 3139-3140.
R. R. Holmes. Chem. Rev., 1990, 90, 17-31.
M. S. Gordon, L. P. Davis, L. W. Burggraf, and R. Damrauer. J. Am. Chem. Soc., 1986, 108, 7889-7893.
J. A. Deiters and R. R. Holmes. J. Am. Chem. Soc., 1987, 109, 1686-1692.
M. L. P. da Silva and J. M. Reveros. J. Mass Spectrom., 1995, 30, 733-740.
J. C. Sheldon, R. N. Hayes, and J. H. Bowie. J. Am. Chem. Soc., 1984, 106, 7711-7715.
T. Veszpremi, Y. Harada, K. Ohno, and H. J. Mutoh. J. Organomet. Chem., 1984, 266, 9-16.
N. Lühmann, R. Panisch, and T. Müller. Appl. Organomet. Chem., 2010, 24, 537.
T. Ziegler. Chem. Rev., 1991, 91, 667.
X. Shu-Hong and Z. Ming-yu. Chem. Phys. Lett., 2006, 421, 447.
K. Wolinski, J. F. Hinton, and P. Pulay. J. Am. Chem. Soc., 1990, 112, 8260.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox. Gaussian 09. Gaussian, Inc.: Wallingford, CT, 2009.
A. E. Reed, R. B. Weinstock, and F. Weinhold. J. Chem. Phys., 1985, 83, 746.
Valency and Bonding: A Natural Bond Orbital Donor-Acceptor Perspective / Eds. F. Weinhold and C. R. Landis. Cambridge University Press, 2005.
E. D. Glendening, C. R. Landis, and F. Weinhold. J. Comput. Chem., 2013, 34, 1437.
F. Weinhold. Natural Bond Orbital Methods. In: Encyclopedia of Computational Chemistry / Eds. P. von Ragué Schleyer, N. L. Allinger, T. Clark, J. Gasteiger, P. A. Kollman, H. F. Schaefer, and P. R. Schreiner. Wiley, 2002.
G. L. Larson. Recent Synthetic Applications of Organosilanes. In: Organic Silicon Compounds / S. Patai, and Z. Rappoport. Wiley: Chichester, UK, 1989.
A. Sekiguchi, R. Kinjo, and M. Ichinohe. Science, 2004, 305, 1757.
H. Ottosson and A. M. Eklöf. Coord. Chem. Rev., 2007, 252, 1314.
E. C. Lee, D. Kim, P. Jurecka, P. Tarakeshwar, and P. Hobza. J. Phys. Chem. A, 2007, 111, 3457.
M. El Idrissi, A. Zeroual, and A. El Hajbi. Int. J. Innovation Appl. Stud., 2014, 5, 260.
Essentials of Computational Chemistry, 2nd ed. / Ed. C. J. Cramer. John Wiley & Sons, 2004.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interests.
Additional information
Text © The Author(s), 2021, published in Zhurnal Strukturnoi Khimii, 2021, Vol. 62, No. 6, pp. 892-902.https://doi.org/10.26902/JSC_id72849
Rights and permissions
About this article
Cite this article
Aichi, M., Hafied, M. & Dibi, A. THEORETICAL STUDY OF PENTAVALENT HALOSILICONATES: STRUCTURE AND CHARGE DELOCALIZATION. J Struct Chem 62, 824–834 (2021). https://doi.org/10.1134/S0022476621060020
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476621060020