Abstract
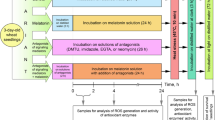
Leaves of 4-week-old (juvenile) and 9-week-old (adult) plants of the halophyte Mesembryanthemum crystallinum L. (the common ice plant), cultured under controlled conditions in the phytotron, were treated with paraquat (0.1 μM), which produces superoxide radical, and (or) paraquat combined with introduction of NaCl (100 mM) or proline (5 mM) into nutrient medium. After a 20-h dark period (23°C), plants were transferred into light (4 h at 54.1 W/m2 of photosynthetically active radiation) for stimulation of O° −2 formation in plastids. Activities of antioxidant enzymes, the contents of MDA, H2O2, chlorophyll, and free proline were measured in leaves. Plant responses in two age groups, which differed in the type of photosynthesis (juvenile plants had C3 type of photosynthesis, whereas adult plants were at the transition stage to Crassulacean Acid Metabolism (CAM) photosynthesis), differed in the levels of constitutive proline and proline, induced by NaCl and paraquat, as well as in activities of superoxide dismutase (SOD) and catalase. Changes in SOD activity and proline accumulation in response to paraquat treatment combined with NaCl revealed opposite dependence to accumulation of proline: the more proline accumulated in leaves, the lower activity of the enzyme. In response to paraquat treatment, the content of chlorophylls a and b most drastically declined in juvenile plants. Negative effect of salinity on the content of chlorophylls was lower than that of paraquat and was almost the same in plants of both age groups. Protective effect of exogenous proline was most profound in the case of paraquat treatment. Exogenous proline decreased the rate of lipid peroxidation, the content of superoxide radical and, consequently, SOD activity (almost fivefold), and increased the content of chlorophylls (a and b) in leaves of adult plants. The obtained data suggest that stress-induced accumulation of proline in the common ice plant has both osmoprotectory and antioxidant functions.
Similar content being viewed by others
Abbreviations
- CAM:
-
crassulacean acid metabolism
- MDA:
-
malondialdehyde
- POL:
-
peroxidation of lipids
- ROS:
-
reactive oxygen species
- SOD:
-
superoxide dismutase
References
Hernandes, J.A., Ferrer, M.A., and Jiménez, A., Los Barceló A., Sevilla F. Antioxidant Systems and O° −2 /H2O2 Production in the Apoplast of Pea Leaves. Its Relation with Salt-Induced Necrotic Lesions in Minor Veins, Plant Physiol., 2001, vol. 127, pp. 817–831.
Stadtman, E.R., Protein Oxidation and Aging, Science, 1992, vol. 257, pp. 1220–1224.
Asada, K., Production and Action of Active Oxygen Species in Photosynthetic Tissues, Causes of Photosynthetic Stress and Amelioration of Defense Systems in Plants, Foyer, C.H. and Mullineaux, P.M., Eds., London: CPC, 1994, pp. 77–104.
Smirnoff, N. and Cumbes, Q.J., Hydroxyl Radicals Scavenging Activity of Compatible Solutes, Phytochemistry, 1989, vol. 28, pp. 1057–1059.
Shen, B., Jensen, G., and Bohnert, H.J., Mannitol Protects against Oxidation by Hydroxyl Radicals, Plant Physiol., 1997, vol. 115, pp. 527–532.
Taiczynski, M.C., Jensen, R.G., and Bonnert, H.J., Stress Protection of Transgenic Tobacco by Production of the Osmolyte Mannitol, Science, 1993, vol. 259, pp. 508–510.
Radyukina, N.L., Shashukova, A.V., Shevyakova, N.I., and Kuznetsov, Vl.V., Proline Involvement in the Common Sage Antioxidant System in the Presence of NaCl and Paraquat, Russ. J. Plant Physiol., 2008, vol. 55, pp. 721–730.
Miszalski, Z., Slesak, I., Niewiadomska, E., Baczek-Kwita, R., Luttge, U., and Ratajczak, R., Subcellular Localization and Stress Responses of Superoxide Dismutase Isoforms from Leaves in the C3-CAM Intermediate Halophyte Mesembryanthemum crystallinum L., Plant Cell Environ., 1998, vol. 21, pp. 169–179.
Bodriquez-Serrano, M., Romero-Puertas, M.C., Corpas, F.J., del Rio, L.A., and Palma, J.M., Characterization of Mitochondrial and Peroxisomal Mn-Superoxide Dismutase in Germinating Watermelon Cotyledons, Free Radic. Res., 2003, vol. 37,suppl. 2, p. 30.
Kant, S., Kant, P., Raven, E., and Barak, S., Evidence That Differential Gene Expression between the Halophytes, Thellungiella halophila, and Arabidopsis thaliana, Is Responsible for Higher Levels of Compatible Osmolyte Proline and Higher Control of Na+ Uptake in T. halophila, Plant, Cell Environ., 2006, vol. 29, pp. 1220–1234.
Radyukina, N.L., Ivanov, Yu.V., Kartashov, A.V., Shevyakova, N.I., Rakitin, V.Yu., Khryanin, V.N., and Kuznetsov, Vl.V., Inducible and Constitutive Mechanisms of Salt Stress Resistance in Geum urbanum L., Russ. J. Plant Physiol., 2007, vol. 54, pp. 612–618.
Kuznetsov, Vl.V. and Shevyakova, N.I., Proline under Stress: Biological Role, Metabolism, and Regulation, Russ. J. Plant Physiol., 1999, vol. 46, pp. 274–289.
Dodge, A., Herbicide Action and Effects on Detoxification Processes, Causes of Photooxidative Stress and Amelioration of Defence Systems in Plants, Foyer, C.H. and Mullineaux, P.M., Eds., Boca Raton: CRC, 1994, pp. 219–236.
Winter, K. and Holtum, J.A.M., The Effects of Salinity, Crassulacean Acid Metabolism and Plant Age on the Carbon Isotope Composition of Mesembryanthemum crystallinum L., a Halophytic C3-CAM Species, Planta, 2005, vol. 222, pp. 201–209.
Winter, K., CO2-Fixierungreaktionen bei der Salzpflanze Mesembryanthemum crystallinum under Variierten Aussenbedingungen, Planta, 1973, vol. 114, pp. 75–85.
Adams, P., Nelson, D.E., Yamada, S., Chmara, W., Jensen, R.J., and Griffiths, H., Growth and Development of Mesembryanthemum crystallinum (Aizoaceae), New Phytol., 1998, vol. 138, pp. 171–190.
Bates, L.S., Waldren, R.P., and Teare, I.D., Rapid Determination of Free Proline for Water Stress Studies, Plant Soil, 1973, vol. 39, pp. 205–207.
Borrell, A., Carbonell, L., Farras, R., Puig-Perellada, P., and Tiburcio, A.F., Polyamines Inhibit Lipid Peroxidation in Senescing Oat Leaves, Physiol. Plant., 1997, vol. 99, pp. 385–390.
Heath, R.L. and Parker, L., Photoperoxidation in Isolated Chloroplasts. I. Kinetics and Stoichiometry of Fatty Acid Peroxidation, Arch. Biochem. Biophys., 1968, vol. 125, pp. 189–198.
Brennan, T. and Frenkel, C., Involvement of Hydrogen Peroxide in the Regulation of Senescence in Pear, Plant Physiol., 1997, vol. 59, pp. 411–416.
Beckett, R.P., Minibayeva, F.V., and Mika, A., Extracellular Superoxide Production in the Liverwort Dumortiera, Free Radic. Res., 2003, vol. 37,suppl. 2, p. 12.
Beauchamp, Ch. and Fridovich, I., Superoxide Dismutase Improved Assays and an Assay Applicable to Acrylamide Gels, Anal. Biochem., 1971, vol. 44, pp. 276–287.
Maehly, A.C. and Chance, B., The Assay of Catalases and Peroxidases, InterScience (New York), 1954, pp. 357–408.
Esen, A., A Simple Method for Quantitative, Semiquantitative, and Qualitative Assay of Protein, Anal. Biochem., 1978, vol. 89, pp. 264–273.
Kalinkina, L.G. and Strogonov, B.P., Growth and Biomass Accumulation in Marine and Freshwater Chlorella at Glycolate Pathway Inhibition by Salinity, Sov. Plant Physiol., 1986, vol. 33, pp. 746–753.
Foyer, C.H. and Noctor, G., Redox Sensing and Signaling Associated with Reactive Oxygen in Chloroplasts, Peroxisomes and Mitochondria, Physiol. Plant., 2003, vol. 119, pp. 355–364.
Hanson, A.D., May, A.M., Grunet, R.R., Bode, J., Jamieson, G.C., and Rhodes, D., Betaine Synthesis in Chenopods: Localization in Chloroplasts, Proc. Natl. Acad. Sci. USA, 1985, vol. 82, pp. 3678–3682.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.I. Shevyakova, E.A. Bakulina, Vl.V. Kuznetsov, 2009, published in Fiziologiya Rastenii, 2009, Vol. 56, No. 5, pp. 736–742.
Rights and permissions
About this article
Cite this article
Shevyakova, N.I., Bakulina, E.A. & Kuznetsov, V.V. Proline antioxidant role in the common ice plant subjected to salinity and paraquat treatment inducing oxidative stress. Russ J Plant Physiol 56, 663–669 (2009). https://doi.org/10.1134/S1021443709050124
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443709050124