Abstract
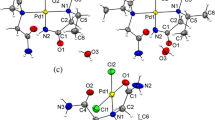
The crystal structure of a palladium(II) complex of imidazolidine-2-thione (Imt), [Pd(Imt)4]Cl2 (I) was determined by X-ray crystallography (CIF file CCDC no. 1946290). The structure of complex I is ionic consisting of [Pd(Imt)4]2+ cation and two chloride counter ions. The palladium atom in the complex ion assumes a distorted square planar geometry. The complex cation and non-coordinated chloride ions are connected to each other through electrostatic and hydrogen bonding interactions. To compare the stability of I with its corresponding non-ionic species [Pd(Imt)2Cl2] . 2Imt (Ia), DFT calculations were performed in the gas phase as well as in the DMSO solvent. The IR spectra of the ligand Imt and complex I were reliably interpreted theoretically. The structures of two related cyanide complexes; [Pd(Imt)2(CN)2]2 (II) and [Pd(Imt)4][Pd(CN)4] (IIa) were also predicted by DFT calculations in terms of geometries and stability. The DFT results reveal that in the gas phase, the structure I is less stable in comparison to the calculated structure Ia by 11.26 kcal/mol (ΔG) but reverse is true when the DMSO solvent is included in the quantum chemical calculations. For the cyanide analogues, the ionic dinuclear form IIa is more stable than the dimer II of the mononuclear complex [Pd(Imt)2(CN)2] in gas phase by 13.37 kcal/mol but is less stable in calculations with DMSO by 6.86 kcal/mol.



Similar content being viewed by others
REFERENCES
Garoufis, A., Hadjikakou, S.K., and Hadjiliadis, N., Coord. Chem. Rev., 2009, vol. 253, p. 1384.
Moro, A.C., Mauro, A.E., Netto, A.V.G., et al., Eur. J. Med. Chem., 2009, vol. 44, p. 4611.
Nadeem, S., Bolte, M., Ahmad, S., et al., Inorg. Chim. Acta, 2010, vol. 363, p. 261.
Zhang, L.-M., Li, H.-Y., Li, H.-X., et al., Inorg. Chem., 2017, vol. 56, p. 11230.
Tyson, G.E., Tokmic, K., Oian, C.S., et al., Dalton Trans., 2015, vol. 44, p. 14475.
Nadeem, S., Rauf, M.K., Ahmad, S., et al., Transition Met. Chem., 2009, vol. 34, p. 197.
Nadeem, S., Rauf, M.K., Ebihara, M., et al., Acta Crysrtallogr., Sect. E: Struct. Rep. Online, 2008, vol. 64, р. m698.
Fuks, L., Sadlej-Sosnowska, N., Samochocka, K., and Starosta, W., J. Mol. Struct., 2005, vol. 740, p. 229.
Jolley, J., Cross, W.I., Pritchard, R.G., et al., Inorg. Chim. Acta, 2001, vol. 315, p. 36.
Wang, Y.-L., Bi, W.-H., Li, X., and Cao, R., Acta Crysrtallogr., Sect. E: Struct. Rep. Online, 2004, vol. 60, р. m699.
De Munno, G., Gabriele, B., and Salerno, G., Inorg. Chim. Acta, 1995, vol. 234, p. 181.
Kahn, E.S., Rheingold, A.L., and Shupack, S.I., J. Chem. Crystallogr., 1993, vol. 23, p. 697.
Butler, L.M., Creighton, J.R., Oughtred, R.E., and Raper, E.S., Inorg. Chim. Acta, 1983, vol. 75, p. 149.
Ahmad, S., Rüffer, T., Lang, H., et al., Russ. J. Coord. Chem., 2010, vol. 36, p. 520. https://doi.org/10.1134/S1070328410070079
de Moura, T.R., Cavalcanti, S.L., de Godoy, P.R.D.V., et al., Transition Met. Chem., 2017, vol. 42, p. 565.
Umakoshi, K., Ichimura, A., Kinoshita, I., and Ooi, S., Inorg. Chem., 1990, vol. 29, p. 4005.
Tirmizi, S.A., Nadeem, S., Hameed, A., et al., Spectroscopy, 2009, vol. 23, p. 299.
Nadeem, S., Rauf, M.K., Bolte, M., et al., Transition Met. Chem., 2010, vol. 35, p. 555.
Ahmad, S., Nadeem, S., Anwar, A., et al., J. Mol. Struct., 2017, vol. 1141, p. 204.
Nadeem, S., Sirajuddin, M., Ahmad, S., et al., Pharmaceut. Chem. J., 2017, vol. 51, p. 793.
Ahmad, S., Isab, A.A., and Perzanowski, H.P., Can. J. Chem., 2002, vol. 80, p. 1279.
Isab, A.A., Al-Arfaj, A.R., Arab, M., and Hassan, M.M., Transition Met. Chem., 1994, vol. 19, p. 87.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, p. 112.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, p. 3.
Spek, A.L., Acta Crystallogr., Sect. D: Biol. Crystallogr., 2009, vol. 65, p. 148.
Becke, A.D., J. Chem. Phys., 1993, vol. 98, p. 5648.
Lee, C., Yang, W., and Parr, R.G., Phys. Rev., B, 1988, vol. 37, p. 785.
Grimme, S., Antony, J., Ehrlich, S., and Krieg, H., J. Chem. Phys., 2010, vol. 132, p. 154104.
Tomasi, J., Mennucci, B., and Cammi, R., Chem. Rev., 2005, vol. 105, p. 2999.
Boys, S.F. and Bernardi, F., Mol. Phys., 1970, vol. 19, p. 553.
Hay, P.J. and Wadt, W.R., J. Chem. Phys., 1985, vol. 82, p. 299.
Dunning, T.H., Jr. and Hay, P.J., in Modern Theoretical Chemistry, Schaefer H.F. III, Ed., New York: Plenum, vol. 3, 1976.
Weinhold, F., Landis, C.R., and Glendening. E.D., Inter. Rev. Phys. Chem., 2016, vol. 35, p. 399.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., et al., Wallingford: Gaussian, Inc., 2016.
Orysyk, S.I., Bon, V.V., Zholob, O.A., et al., Russ. J. Coord. Chem., 2014, vol. 40, p. 160. https://doi.org/10.1134/S1070328414030063
Burlov, A.S., Uraev, A.I., Lysenko, K.A., et al., Russ. J. Inorg. Chem., 2015, vol. 60, p. 1481. https://doi.org/10.1134/S0036023615120062
Jomaa, M.Y., Altaf, M., Ahmad, S., et al., Polyhedron, 2018, vol. 141, p. 360.
Mahmood, R., Ahmad, S., Fettouhi, M., et al., J. Mol. Struct., 2018, vol. 1156, p. 235.
Ahmad, S., Seerat-ur-Rehman, Arshad, M.N., et al., Z. Naturforsch., B, 2019, vol. 74, p. 565.
Isab, A.A., Fettouhi, M., Malik, M.R., et al., Russ. J. Coord. Chem., 2011, vol. 37, p. 180. https://doi.org/10.1134/S1070328411030055
Mahmood, R., Sadaf, S., Is, A.A., et al., Russ. J. Coord. Chem., 2012, vol. 38, p. 456. https://doi.org/10.1134/S1070328412060085
Ahmad, T., Mahmood, R., Lee, S.C., and Ahmad, S., Russ. J. Coord. Chem., 2014, vol. 40, p. 125. https://doi.org/10.1134/S1070328414020018
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Rights and permissions
About this article
Cite this article
Sadaf, H., Zierkiewicz, W., Michalczyk, M. et al. Crystal Structure of [Pd(Imt)4]Cl2 and DFT Studies of [Pd(Imt)4]Cl2 and [Pd(Imt)2(CN)2] (Imt = Imidazolidine-2-Thione). Russ J Coord Chem 47, 81–87 (2021). https://doi.org/10.1134/S107032842101005X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107032842101005X