Abstract
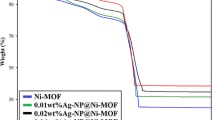
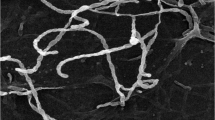
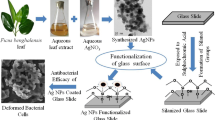
In the current work, silver nanoparticles (AgNPs) were green synthesized using starch and modified with anionic dye (acid fuchsin) and aldehyde (salicylaldehyde and cinnamaldehyde). Thus in situ formation of Schiff base stabilized AgNPs were characterized by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), scanning electron microscope (SEM) and transmission electron microscope (TEM) analysis. The size and the stability of the Schiff base modified AgNPs were explored using DLS and Zeta potential analysis. TEM analyses showed the size of the Schiff base (cinnamaldehyde) modified AgNPs with an average particle size of 5–10 nm. The synthesized Schiff base modified AgNPs were tested for their antibacterial efficacy against gram-negative bacteria such as Escherichia coli (MTCC733), Pseudomonas aeruginosa (MTCC1688) and gram-positive bacteria such as Bacillus subtilis (MTCC41) and Enterococcus faecalis (MTCC96). The antifungal activity of the synthesized materials has been studied against Candida albicans along with standard fluconazole. Higher antibacterial and antifungal activities were observed for the Schiff base (SB) capped AgNPs due to the combined effect of SB and AgNPs. The mechanism of the bactericidal activity of nanoparticles is suggested as due to the interaction of AgNPs with the cell membrane. The results of the present study confirm that SB stabilized AgNPs have significant potential as an antimicrobial agent in treating infectious diseases.









Similar content being viewed by others
REFERENCES
A. Ahmad, P. Mukherjee, S. Senapati, et al., Colloids Surf., B 28, 313 (2003).
S. Iravani, H. Korbekandi, S. V. Mirmohammadi, and B. Zolfaghari, Res. Pharm. Sci. 9, 385 (2014).
S. A. Jones, P. G. Bowler, M. Walker, and D. Parsons, Wound Rep. Regen. 12, 288 (2004).
S. Mirsadeghi, M. F. Koudehi, H. R. Rajabi, and S. M. Pourmortazavi, Curr. Pharm. Biotechnol. 10, 2174 (2019)
H. Jang, Y. K. Kim, H. Huh, and D. H. Min, ACS Nano 8, 467 (2014).
Gao Xianghua, Liqiao Wei, Jing Wang, and Bingshe Xu, Adv. Mater. Res. 236, 1945 (2011).
S. Honary, K. Ghajar, P. Khazaeli, and P. Shalchia, Trop. J. Pharm. Res. 10, 69 (2011).
N. Vigneshwaran, R. P. Achane, R. H. Balasubramanya, and P. V. Varadarajan, Carbohydr. Res. 341, 2012 (2006).
S. Patil, S. D. Jadhav, and S. K. Shinde, Org. Chem. Int., No. 153519, 1 (2012).
Suba Kannaiyan, Easwaramoorthi, and V. Andal, Int. J. Chem. Tech. Res. 8, 54 (2015).
Abiola Azeez Jimoh, Aasif Helal, M. Nasiruzzaman Shaikh, et al., J. Nanomater., No. 101694, 1 (2015).
Pengcheng Huang, Jianfang Li, Xin Liu, and Fangying Wu, Microchim. Acta 183, 863 (2016).
J. Balachandramohan and T. Sivasankar, J. Microencapsul. 37, 29 (2020).
M. Ganguly, C. Mondal, J. Jana, et al., Langmuir 30, 4120 (2014).
A. Abbasi, H. Khojasteh, M. Hamadanian, and M. Salavati-Niasari, J. Mater. Sci.: Mater. Electron. 27, 4972 (2016).
Z. Shiri-Yekta, M. Reza Yaftian, and A. Nilchi, Korean J. Chem. Eng. 30, 1644 (2013).
E. E. Elemikea, E. O. Dareb, I. D. Samuel, and J. Ch. Onwukab, J. Appl. Res. Technol. 14, 38 (2016).
M. Shakouri-Arani and M. Salavati-Niasari, Spectrochim. Acta, A 133, 463 (2014).
S. Agnihotri, S. Mukherjee, and S. Mukherjee, Nanoscale 5, 7328 (2013).
S. A. Khan, A. A. Siddiqui, and B. Shibeer, Asian J. Chem. 14, 1117 (2002)
C. Perez, M. Pauli, and P. Bazerque, Acta Biol. Med. Exp. 15, 113–115 (1990).
R. M. Issa, A. M. Khedr, and H. F. Rizk, J. Chin. Chem. Soc. 55, 875 (2008).
M. Zobir bin Hussein, A. Hj Yahaya, M. Shamsul, et al., Mater. Lett. 58, 329 (2004)
M. R. Silverstein, G. C. Bassler, and T. C. Morril, Spectrometric Identification of Organic Compounds, 4th ed. (Wiley, New York, 1981), pp. 111, 130.
A. K. Anandalakshmi, J. V. Venugobal, and Ramasamy, Appl. Nanosci. 6, 399 (2016).
V. dal Lago, L. F. de Oliveira, K. de Almeida Goncalves, et al., J. Mater. Chem. 21, 12267 (2011).
S. Pal, Y. K. Tak, and J. M. Song, Appl. Environ. Microbiol. 73, 1712 (2007).
S. Ashraf, N. Akhtar, M. Afzal Ghauri, et al., Nanoscale Res. Lett. 267, 1 (2012)
A. M. El Badawy, R. G. Silva, B. Morris, et al., Environ. Sci. Technol. 45, 283 (2011).
P. K. Khanna, N. Singh, K. Deepti, et al., Mater. Lett. 61, 3366 (2007).
M. Cleiton, Da Silva, D. L. Da Silva, et al., J. Adv. Res. 2, 1 (2011).
Y. Xie, L. Chen, X. Zhang et al., J. Colloid Interface Sci. 510, 308 (2018).
T. Dai, C. Wang, Y. Wang, et al., ACS Appl. Mater. Interfaces 10, 15163 (2018).
K. J. Kim, W. S. Sung, B. K. Suh, et al., Biometals 22, 235 (2009).
H. H. Lara, D. G. Romero-Urbina, Ch. Pierce, et al., J. Nanobiotechnol. 13, 91 (2015).
E. M. Ali and B. M. Abdallah, Nanomaterials 10, 422 (2020).
S. Paul, K. Mohanram, and I. Kannan, Pharmacol. Study 39, 182 (2018).
R. Lali Raveendran, N. Kumar Sasidharan, and S. Devaki, J. Bioconjug. Chem. 28, 1005 (2017).
ACKNOWLEDGMENTS
The authors would like to express heartfelt thanks to KCG College of Technology for providing lab facilities and for their support during their research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kannaiyan, S., Easwaramoorthy, Kannan, K. et al. Synthesis, Characterisation, and Antimicrobial Efficacy of Acid Fuchsin Schiff Base-Modified Silver Nanoparticles. Nanotechnol Russia 15, 828–836 (2020). https://doi.org/10.1134/S1995078020060208
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995078020060208