Abstract
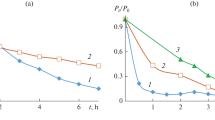
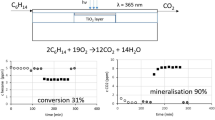
Supercritical CO2 (scCO2) has been used as a reaction medium for the photocatalytic oxidative degradation of n-octanol on a partially desilanized hydrophobic suspension of TiO2 as photocatalyst. Hydrophobic sites on the catalyst surface are necessary to maintain a sustained suspension, and hence surface-mediated interfacial electron exchange, in this non-polar medium. The reaction rates for photooxidative degradation, ultimately to complete mineralization, depend only weakly on temperature and pressure of the supercritical fluid near the critical point. Product distributions were monitored in situ by on-line gas chromatographic analysis, which provides a convenient and rapid method for comparisons and optimization of the reaction conditions.
Similar content being viewed by others
REFERENCES
M. A. Fox, D. F. Ollis and H. Al-Ekabi (Eds), Photocatalytic Purification and Treatment of Water and Air, in: Proceedings of the First International Conference on the TiO 2 Photocatalytic Purification and Treatment of Water and Air, p. 163. Elsevier, Amsterdam (1993).
M. A. Fox and M. T. Dulay, Chem. Rev. 93, 341 (1993).
A. L. Pruden and D. F. Ollis, Environ. Sci. Tech. 17, 628 (1983). (b) M. Graetzel, Energy Resources through Photochemistry and Catalysis. Academic Press, New York (1983).
M. R. Hoffmann, S. T. Martin, W. Y. Choi and D. W. Bahnemann, Chem. Rev. 95, 69 (1995).
N. R. Rusu and J. T. Yates, Jr., J. Phys. Chem. B 104, 12299 (2000).
(a) C. Erkey, G. Madras, M. Orenjuela and A. Akgerman, Envir. Sci. Technol. 27, 1225 (1993). (b) M. A. McHugh and V. J. Krukonis, Supercritical Fluid Extractions: Principles and Practice, 2nd edn. Butterworth-Heinerman,Boston, MA (1994).
T. J. Bruno and J. F. Ely (Eds), in: Supercritical Fluid Technology: Reviews in Modern Theory and Applications, p. 593. CRC Press, Boston, MA (1991).
J.F. Brennecke and C. A. Eckert, in: Supercritical Fluid Science and Technology, K. P. Johnston and J. M. L. Penninger (Eds), p. 14. American Chemical Society, Washington, DC (1989).
A. Baiker, Chem. Rev. 99, 453 (1999).
J. M. Becnel and K. M. Dooley, Ind. Eng. Chem. Res. 37, 584 (1998).
K. Hartonen, S. Bøwadt, S. B. Hawthorne and M.-L. Riekkola, J. Chromat. A 774, 229 (1997).
C. S. Eskilsson and L. Mathiasson, J. Agric. Food Chem. 48, 5159 (2000).
K. Chihara, K. Oomori, T. Oono and Y. Mochizuki,Water Sci. Technol. 35, 261 (1997).
G. Musie, M. Wei, B. Subramaniamand D. H. Busch, Coord. Chem. Rev. 219-221, 789 (2001).
S. Kaneco, H. Kurimoto, Shimizu, K. Ohta and T. Mizuno, Energy 24, 21 (1999).
Degussa Tech. Bull. No. 56, 5th edn. A. G. Degussa, Frankfurt, Germany (1990).
C. Minero, V. Maurino and E. Pelizzetti, Marine Chem. 58, 361 (1997).
C. Minero, V. Maurino and E. Pelizzetti, Langmuir 11, 4440 (1995).
T. Rhodes and M. A. Fox, J. Phys. Chem. 100, 17931 (1996).
T. Rhodes and M. A. Fox, Appl. Spectrosc. 51, 358 (1997).
B. Erdem, R. A. Hunsicker, G. W. Simmons, E. D. Sudol, V. L. Dimonie and M. S. El-Aasser, Langmuir 17, 2664 (2001).
Rights and permissions
About this article
Cite this article
Resmi, M.R., Whitesell, J.K. & Fox, M.A. Photocatalytic oxidation of n-octanol in aerated supercritical CO2 on hydrophobic TiO2 . Research on Chemical Intermediates 28, 711–718 (2002). https://doi.org/10.1163/15685670260469375
Issue Date:
DOI: https://doi.org/10.1163/15685670260469375