Abstract
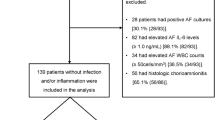
Because relevant biochemical changes are known to begin at the choriodecidual interface some weeks before actual clinical onset of labor, we hypothesized that the preterm choriodecidua may display gene and protein expression patterns specific to preterm labor. Transcriptomic (microarray) and proteomic (2-dimensional gel electrophoresis [2DGE]) profiling methodologies were used to compare changes in choriodecidual tissue collected from women who delivered before 35 weeks of gestation following spontaneous preterm labor (n = 12) and gestation-matched nonlaboring controls (n = 7). Additionally, 2DGE was used to compare differences in protein expression during term and preterm labor and to construct a choriodecidual proteome map. Overall, expressed transcripts and proteins indicated active tissue remodeling independent of labor status and an association with inflammatory processes during labor. Spontaneous, infection-induced and abruption-associated preterm deliveries were each defined by distinct transcriptional profiles. Proteins osteoglycin and progesterone receptor component 2 (PGRMC2) were upregulated during term and preterm labor while galectin 1, annexin 3, annexin 5, and protein disulfide isomerase (PDI) were upregulated only during preterm labor, suggesting a probable association with the underlying pathology. Together, these results represent novel data that warrant further investigations to elucidate plausible causal relationships of these molecules with spontaneous preterm delivery.
Similar content being viewed by others
References
Romero R, Espinoza J, Kusanovic JP, et al. The preterm parturition syndrome. BJOG. 2006;113(suppl 3):17–42.
Bell SC, Malak TM. Structural and Cellular Biology of the Fetal Membranes. In: Elder MG, Romero R, Lamont RF, eds. Preterm Labour. New York: Churchill Livingstone; 1997: 401–428.
Goldenberg RL, Iams JD, Mercer BM, et al. The Preterm Prediction Study: toward a multiple-marker test for spontaneous preterm birth. Am J Obstet Gynecol. 2001;185(3):643–651.
Haddad R, Tromp G, Kuivaniemi H, et al. Human spontaneous labor without histologic chorioamnionitis is characterized by an acute inflammation gene expression signature. Am J Obstet Gynecol. 2006;195(2):394 e391–324.
Ramagli LS. Quantifying protein in 2D PAGE Solubilization Buffers. In: Link Andrew, ed. Methods in Molecular Biology Vol 112: 2D Proteome Analysis Protocols. New Jersey: Humana Press Inc; 1999:99–103.
Kaplanski G, Marin V, Montero-Julian F, Mantovani A, Farnarier C. IL-6: a regulator of the transition from neutrophil to monocyte recruitment during inflammation. Trends Immunol. 2003;24(1):25–29.
Croker BA, Krebs DL, Zhang JG, et al. SOCS3 negatively regulates IL-6 signaling in vivo. Nat Immunol. 2003;4(6): 540–545.
Ognjanovic S, Bryant-Greenwood GD. Pre-B-cell colony-enhancing factor, a novel cytokine of human fetal membranes. Am J Obst Gynecol. 2002;187(4):1051–1058.
Yasukawa H, Ohishi M, Mori H, et al. IL-6 induces an anti-inflammatory response in the absence of SOCS3 in macro-phages. Nat Immunol. 2003;4(6):551–556.
Laham N, Brennecke SP, Bendtzen K, Rice GE. Differential release of interleukin-6 from human gestational tissues in association with labour and in vitro endotoxin treatment. J Endocrinol. 1996;149(3):431–439.
Salmon JE, Girardi G, Holers VM. Complement activation as a mediator of antiphospholipid antibody induced pregnancy loss and thrombosis. Ann Rheum Dis. 2002;61(suppl 2):ii46–ii50.
Takeuchi O, Sato S, Horiuchi T, et al. Cutting edge: role of Toll-like receptor 1 in mediating immune response to microbial lipoproteins. J Immunol. 2002;169(1):10–14.
Lockwood CJ, Toti P, Arcuri F, et al. Mechanisms of abruption-induced premature rupture of the fetal membranes: thrombin-enhanced interleukin-8 expression in term decidua. Am J Pathol. 2005;167(5):1443–1449.
Hayashi F, Means TK, Luster AD. Toll-like receptors stimulate human neutrophil function. Blood. 2003;102(7): 2660–2669.
Fortunato SJ, Menon R. Distinct molecular events suggest different pathways for preterm labor and premature rupture of membranes. Am J Obstet Gynecol. 2001;184(7):1399–1405; discussion 1405–1396.
Huang Y, Zhao Q, Chen GQ. Phospholipid scramblase 1. Sheng Li Xue Bao. 2006;58(6):501–510.
Hart SP, Dougherty GJ, Haslett C, Dransfield I. CD44 regulates phagocytosis of apoptotic neutrophil granulocytes, but not apoptotic lymphocytes, by human macrophages. J Immunol. 1997;159(2):919–925.
Gottlieb RA, Kitsis RN. Seeing death in the living. Nat Med. 2001;7(12):1277–1278.
Leroy MJ, Dallot E, Czerkiewicz I, Schmitz T, Breuiller-Fouche M. Inflammation of choriodecidua induces tumor necrosis factor alpha-mediated apoptosis of human myometrial cells. Biol Reprod. 2007;76(5):769–776.
Gaur U, Aggarwal BB. Regulation of proliferation, survival and apoptosis by members of the TNF superfamily. Biochem Pharmacol. 2003;66(8):1403–1408.
Liu H, Ma Y, Pagliari LJ, et al. TNF-alpha-induced apoptosis of macrophages following inhibition of NF-kappa B: a central role for disruption of mitochondria. J Immunol. 2004;172(3): 1907–1915.
Alexander JM, Bikkal HA, Zervas NT, Laws ER Jr, Klibanski A. Tumor-specific expression and alternate splicing of messenger ribonucleic acid encoding activin/transforming growth factor-beta receptors in human pituitary adenomas. J Clin Endocrinol Metab. 1996;81(2):783–790.
Krause CD, He W, Kotenko S, Pestka S. Modulation of the activation of Stat1 by the interferon-gamma receptor complex. Cell Res. 2006;16(1):113–123.
Lappas M, Permezel M, Georgiou HM, Rice GE. Regulation of proinflammatory cytokines in human gestational tissues by peroxisome proliferator-activated receptor-gamma: effect of 15-deoxy-Delta(12,14)-PGJ(2) and troglitazone. J Clin Endocrinol Metab. 2002;87(10):4667–4672.
Monick MM, Robeff PK, Butler NS, et al. Phosphatidylinositol 3-kinase activity negatively regulates stability of cyclooxygenase 2 mRNA. J Biol Chem. 2002;277(36):32992–33000.
Shanahan CM, Cary NR, Osbourn JK, Weissberg PL. Identification of osteoglycin as a component of the vascular matrix. Differential expression by vascular smooth muscle cells during neointima formation and in atherosclerotic plaques. Arterioscler Thromb Vasc Biol. 1997;17(11):2437–2447.
Schultz-Norton JR, McDonald WH, Yates JR, Nardulli AM. Protein disulfide isomerase serves as a molecular chaperone to maintain estrogen receptor alpha structure and function. Mol Endocrinol. 2006;20(9):1982–1995.
Winkler M, Kemp B, Classen-Linke I, et al. Estrogen receptor alpha and progesterone receptor A and B concentration and localization in the lower uterine segment in term parturition. J Soc Gynecol Investig. 2002;9(4):226–232.
Morrison JJ, Charnock-Jones DS, Smith SK. Messenger RNA encoding thiol protein disulphide isomerase in amnion, chorion and placenta in human term and preterm labour. Br J Obstet Gynaecol. 1996;103(9):873–878.
Hirai Y, Utsugi K, Takeshima N, et al. Putative gene loci associated with carcinogenesis and metastasis of endocervical adenocarcinomas of uterus determined by conventional and array-based CGH. Am J Obstet Gynecol. 2004;191(4):1173–1182.
Peluso JJ, Pappalardo A, Losel R, Wehling M. Progesterone membrane receptor component 1 expression in the immature rat ovary and its role in mediating progesterone’s antiapoptotic action. Endocrinology. 2006;147(6):3133–3140.
Pereira L, Reddy AP, Jacob T, et al. Identification of novel protein biomarkers of preterm birth in human cervical-vaginal fluid. J Proteome Res. 2007;6(4):1269–1276.
Gottlieb RA. Mitochondria and apoptosis. Biol Signals Recept. 2001;10(3–4):147–161.
Bondanza A, Zimmermann VS, Rovere-Querini P, et al. Inhibition of phosphatidylserine recognition heightens the immunogenicity of irradiated lymphoma cells in vivo. J Exp Med. 2004;200(9):1157–1165.
Rabinovich GA, Daly G, Dreja H, et al. Recombinant galectin-1 and its genetic delivery suppress collagen-induced arthritis via T cell apoptosis. J Exp Med. 1999;190(3):385–398.
Fortunato SJ, Menon RP, Swan KF, Menon R. Inflammatory cytokine (interleukins 1, 6 and 8 and tumor necrosis factor-alpha) release from cultured human fetal membranes in response to endotoxic lipopolysaccharide mirrors amniotic fluid concentrations. Am J Obstet Gynecol. 1996;174(6): 1855–1861; discussion 1861–1852.
Challis JRG, Matthews SG, Gibb W, Lye SJ. Endocrine and paracrine regulation of birth at term and preterm. Endocrine Reviews. 2000;21(5):514–550.
Lappas M, Permezel M, Georgiou HM, Rice GE. Nuclear factor kappa B regulation of proinflammatory cytokines in human gestational tissues in vitro. Biol Reprod. 2002;67(2):668–673.
Marvin KW, Keelan JA, Eykholt RL, Sato TA, Mitchell MD. Use of cDNA arrays to generate differential expression profiles for inflammatory genes in human gestational membranes delivered at term and preterm. Mol Hum Reprod. 2002;8(4):399–408.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shankar, R., Johnson, M.P., Williamson, N.A. et al. Molecular Markers of Preterm Labor in the Choriodecidua. Reprod. Sci. 17, 297–310 (2010). https://doi.org/10.1177/1933719109353454
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719109353454