Abstract
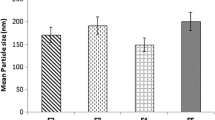
The purpose of this research was to study the effect of the lipid matrix on the entrapment of olanzapine (OL). OL-loaded solid lipid nanoparticles (SLNs) were prepared using lipids like glyceryl monostearate (GMS), Precirol ATO 5 (PRE), glyceryl tristearate (GTS), and Witepsol E85 (WE 85)—and poloxamer 407 and hydrogenated soya phosphatidylcholine as stabilizers—using a hot melt emulsification high-pressure homogenization technique, and then characterized by particle size analysis, zeta potential, differential scanning calorimetry (DSC), and powder X-ray diffraction (pXRD). Homogenization at 10 000 psi for 3 cycles resulted in the formation of SLNs with a mean particle size of ∼190 nm for the 4 lipids investigated. The highest partition coefficient for OL between the melted lipid and pH 7.4 phosphate buffer (pH 7.4 PB) was obtained with GTS. The entrapment efficiency was in the following order: GTS SLNs>PRE SLNs>WE 85 SLNs>GMS SLNs. DSC and pXRD showed that much of the incorporated fraction of OL existed in the amorphous state after incorporation into SLNs. A sharp increase in the flocculation of the SLN dispersions was observed upon addition of 0.6 M aqueous sodium sulfate solution. Nanoparticle surface hydrophobicity was in the following order: GTS SLNs>PRE SLNs>WE 85 SLNs>GMS SLNs. A significant increase in size and zeta potential was observed for GTS SLN and WE 85 SLN dispersions stored at 40°C. Release of OL from the SLNs was sustained up to 48 hours in pH 7.4 PB and obeyed Higuchi’s release kinetics.
Similar content being viewed by others
References
Heiati H, Tawashi R, Phillips NC. Drug retention and stability of solid lipid nanoparticles containing azidothymidine palmitate after autoclaving, storage and lyophilization.J Microencapsul. 1998;15:173–184.
Bunjes H, Westesen K, Koch MHJ. Crystallization tendency and polymorphic transitions in triglyceride nanoparticles.Int J Pharm. 1996;129:159–173.
Westesen K, Bunjes H, Koch MHJ. Physicochemical characterization of lipid nanoparticles and evaluation of their drug loading and sustained release potential.J Control Release. 1997;48:223–236.
Cavalli R, Gasco MR, Morel S. Behaviour of timolol incorporated in lipospheres in the presence of a series of phosphate esters.STP Pharma Sci. 1992;2:514–518.
Mehnert W, Mader K. Solid lipid nanoparticles production, characterization and applications.Adv Drug Deliv Rev. 2001;47:165–196.
Siekmann B, Westesen K. Submicron-sized parenteral carrier systems based on solid lipids.Pharm Pharmacol Lett. 1992;1:123–126.
Bever KA, Perry PJ. Olanzapine: a serotonin-dopamine receptor antagonist for antipsychotic therapy.Am J Health Syst Pharm. 1998;55:1003–1016.
Venkateswarlu V, Manjunath K. Preparation, characterization and in vitro release kinetics of clozapine solid lipid nanoparticles.J Control Release. 2004;95:627–638.
Reddy LH, Vivek K, Bakshi N, et al. Tamoxifen citrate loaded solid lipid nanoparticles (SLN™): preparation, characterization, in vitro drug release and pharmacokinetic evaluation.Pharm Dev Technol. 2006;11:167–177.
Subramanian N, Murthy RSR. Use of electrolyte induced flocculation technique for an in vitro steric stability study of steric stabilized liposome formulations.Pharmazie. 2003;59:74–76.
Gessner A, Waicz R, Lieske A, et al. Nanoparticles with decreasing surface hydrophobicities: influence on plasma protein adsorption.Int J Pharm. 2000;196:245–249.
Muller RH, Mader K, Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery—a review of the state of the art.Eur J Pharm Biopharm. 2000;50:161–177.
Muller RH, Keck CM. Challenges and solutions for the delivery of biotech drugs—a review of drug nanocrystal technology and lipid nanoparticles.J Biotechnol. 2004;113:151–170.
Freitas C, Muller RH. Correlation between long-term stability of solid lipid nanoparticles (SLN™) and crystallinity of the lipid phase.Eur J Pharm Sci. 1999;47:125–132.
Huang SK, Martin FJ, Jay G, et al. Extravasation and transcytosis of liposomes in Kaposi’s sarcoma—like dermal lesions of transgenic mice bearing HIV Tat gene.Am J Pathol. 1993;143:10–14.
Reddy LH, Murthy RSR. Etoposide-loaded nanoparticles made from glyceride lipids: formulation, characterization, in vitro drug release, and stability evaluation.AAPS PharmSciTech [serial online]. 2005;6:Article 24.
Carstensen H, Muller BW, Muller RH. Adsorption of ethoxylated surfactants on nanoparticles, I: characterization by hydrophobic interaction chromatography.Int J Pharm. 1991;67:29–37.
Muller RH, Radtke M, Wissing SA. Nanostructured lipid matrices for improved microencapsulation of drugs.Int J Pharm. 2002;242:121–128.
Jenning V, Gohla S. Comparison of wax and glyceride solid lipid nanoparticles (SLN®).Int J Pharm. 2000;196:219–222.
Freitas C, Muller RH. Effect of light and temperature on zeta potential and physical stability in solid lipid nanoparticle (SLN™) dispersions.Int J Pharm. 1998;168:221–229.
Sato K. Crystallization of fats and fatty acids. In: Garti N, Sato K, eds.Crystallization and Polymorphism of Fats and Fatty Acids. New York, NY: Marcel Dekker; 1988:227–266.
Vivek K, Harivardhan Reddy L, Murthy RSR. Comparative study of some biodegradable polymers on the entrapment efficiency, release behavior of etoposide from microspheres.Pharm Dev Technol. In press.
Author information
Authors and Affiliations
Corresponding author
Additional information
Themed Issue: Transforming Pharmaceutical Manufacturing: Benefits and Challenges
Guest Editor — Robin Bogner
Published: October 12, 2007
Rights and permissions
About this article
Cite this article
Vivek, K., Reddy, H. & Murthy, R.S.R. Investigations of the effect of the lipid matrix on drug entrapment, in vitro release, and physical stability of olanzapine-loaded solid lipid nanoparticles. AAPS PharmSciTech 8, 83 (2007). https://doi.org/10.1208/pt0804083
Received:
Revised:
Accepted:
DOI: https://doi.org/10.1208/pt0804083