Abstract
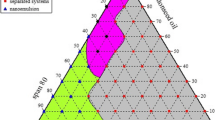
The objective of the present study was to formulate and evaluate microemulsion systems for topical delivery of clotrimazole (CTM). The solubility of CTM in various oils was determined to select the oil phase of the microemulsion systems. Pseudoternary phase diagrams were constructed to identify the area of microemulsion existence. Five CTM microemulsion formulations (M1–M5) were prepared and evaluated for their thermodynamic stability, pH, refractive index, droplet size, viscosity, and in vitro release across cellulose membrane. Among the prepared microemulsion formulations, M3 (lemon oil/Tween 80/n-butanol/water) and M4 (isopropyl myristate/Tween 80/n-butanol/water) microemulsion systems were found to be promising according to their physical properties and CTM cumulative percentage release. Gel form of M3 and M4 were prepared using 1% Carbopol 940 as the hydrogel matrix. Both formulations were evaluated in the liquid and gel forms for drug retention in the skin in comparison to the marketed CTM topical cream and their stability examined after storage at 40°C for 6 months. Microemulsion formulations achieved significantly higher skin retention for CTM over the CTM cream. Stability studies showed that M4 preparations were more stable than M3. The in vitro anti-fungal activity of M4 against Candida albicans was higher than that of the conventional cream. Moreover, clinical evaluation proved the efficacy and tolerability of this preparation in the treatment of various topical fungal infections.





Similar content being viewed by others
REFERENCES
Ritter W, Patzschke K, Krause U, Stettendorf S. Pharmacokinetic fundamentals of vaginal treatment with clotrimazole. Chemotherapy. 1982;28:37–42.
Pedersen M, Bjerregaard S, Jacobsen J, Sørensen AM. Agenuine clotrimazole γ-cyclodextrin inclusion complex—isolation, antimycotic activity, toxicity and an unusual dissolution rate. Int J Pharm. 1998;176:121–31.
Chang JY, Oh YK, Kong HS, Kim EJ, Jang DD, Nam KT, et al. Prolonged antifungal effects of clotrimazole-containing mucoadhesive thermosensitive gels on vaginitis. J Control Release. 2002;82:39–50.
Memişoğlu E, Bochot A, Ozalp M, Sen M, Duchêne D, Hincal AA. Direct formation of nanospheres from amphiphilic beta-cyclodextrin inclusion complexes. Pharm Res. 2003;20(1):117–25.
Souto EB, Wissing SA, Barbosa CM, Müller RH. Development of a controlled release formulation based on SLN and NLC for topical clotrimazole delivery. Int J Pharm. 2004;278:71–7.
Ning M, Gu Z, Pan H, Yu H, Xiao K. Preparation and in vitro evaluation of liposomal/niosomal delivery systems for antifungal drug clotrimazole. Indian J Exp Biol. 2005;43:150–7.
Pavelic Z, Skalko-Basnet N, Jalsenjak I. Characterization and in vitro evaluation of bioadhesive liposome gels for local therapy of vaginitis. Int J Pharm. 2005;301:140–8.
Parbagar B, Yoo BK, Woo JS, Kim JA, Rhee JD, Piao MG, et al. Enhanced bioavailability of poorly water-soluble clotrimazole by inclusion with β cyclodextrin. Arch Pharm Res. 2007;30(2):249–54.
Comelles F, Pascual A. Microemulsions with butyl lactate as cosurfactant. J Dispers Sci Tech. 1997;18:161–75.
Park KM, Kim CH. Preparation and evaluation of flurbiprofen-loaded microemulsion for parenteral delivery. Int J Pharm. 1999;181:173–9.
Lawrence MJ, Rees GD. Microemulsion-based media as novel drug delivery systems. Adv Drug Del Rev. 2000;45:89–121.
Osborne DW, Ward AJI, O’Neil KJ. Microemulsions as topical drug delivery vehicles: in vitro transdermal studies of a model hydrophilic drug. J Pharm Pharmacol. 1991;43:451–4.
Baroli B, López-Quintela MA, Delgado-Charro MB, Fadda AM, Blanco-Méndez J. Microemulsions for topical delivery of 8-methoxsalen. J Control Release. 2000;69:209–18.
Sintov AC, Shapiro L. New microemulsion vehicle facilitates percutaneous penetration in vitro and cutaneous drug bioavailability in vivo. J Control Release. 2004;95:173–83.
Delgado-Charro MB, Iglesias-Vilas G, Blanco-Mendez J, López-Quintela MA, Marty JP, Guy RH. Delivery of a hydrophilic solute through the skin from novel microemulsion systems. Eur J Pharm Biopharm. 1997;43:37–42.
Peltola S, Saarinen-Savolainen P, Kiesvaara J, Suhonen TM, Urtti A. Microemulsions for topical delivery of estradiol. Int J Pharm. 2003;254:99–107.
Boonme P, Krauel K, Graf A, Rades T, Junyaprasert VB. Characterization of microemulsion structure in the pseudoternary phase diagram of isopropyl palmitate/water/Brij 97:1-butanol. AAPS PharmSciTech. 2006;7(2):Article 45.
Tiwari NG, Bajaj AN. Formulation development of eucalyptus oil microemulsion for intranasal delivery. Indian J Pharm Sci. 2007;69:731–3.
Brime B, Moreno MA, Frutos G, Ballesteros MA, Frutos P. Amphotericin B in oil–water lecithin-based microemulsions: formulation and toxicity evaluation. J Pharm Sci. 2002;91(4):1178–85.
Alam MA, Ahmad FJ, Khan ZI, Khar RK, Ali M. Development and evaluation of acid-buffering bioadhesive vaginal tablet for mixed vaginal infections. AAPS PharmSciTech. 2007;8(4):E109.
Hoogerheide JG, Strusiak SH, Taddei CR, Townley ER, Wyka BE. High performance liquid chromatographic determination of clotrimazole in pharmaceutical formulations. J Assoc Off Anal Chem. 1981;64(4):864–9.
Chen H, Mou D, Du D, Chang X, Zhu D, Liu J, et al. Hydrogel-thickened microemulsion for topical administration of drug molecule at an extremely low concentration. Int J Pharm. 2007;341:78–84.
Mou D, Chen H, Du D, Mao C, Wan J, Xu H, et al. Hydrogel-thickened nanoemulsion system for topical delivery of lipophilic drugs. Int J Pharm. 2008;353:270–6.
Baboota S, Shakeel F, Ahuj A, Shafiq S. Design, development and evaluation of novel nanoemulsion formulations for transdermal potential of celecoxib. Acta Pharm. 2007;57:315–32.
Panigrahi L, Ghosal SK, Snigdha P, Maharana L, Barik BB. Effect of permeation enhancers on the release and permeation kinetics of lincomycin hydrochloride gel formulations through mouse skin. Indian J pharm Sci. 2006;68:205–11.
Gupta S, Moulik SP. Biocompatible microemulsions and their prospective uses in drug delivery. J Pharm Sci. 2008;97:22–45.
Kawakami K, Yoshikawa T, Hayashi T, Nishihara Y, Masuda K. Microemulsion formulation for enhanced absorption of poorly soluble drugs. II. In vivo study. J Control Release. 2002;81:75–82.
Junyaprasert VB, Boonme P, Songkro S, Krauel K, Rades T. Transdermal delivery of hydrophobic and hydrophilic local anesthetics from o/w and w/o Brij 97-based microemulsions. J Pharm Pharm Sci. 2007;10(3):288–98.
Talegaonkar S, Azeem A, Ahmad FJ, Khar RK, Pathan SA, Khan ZI. Microemulsions: a novel approach to enhanced drug delivery. Recent Pat Drug Deliv Formul. 2008;2:238–57.
Lam AC, Schechter RS. The theory of diffusion in microemulsion. J Colloid Interface Sci. 1987;120:56–63.
Alany RG, Tucker IG, Davies NM, Rades T. Characterizing colloidal structures of pseudoternary phase diagrams formed by oil/water/amphiphile systems. Drug Dev Ind Pharm. 2001;27:31–8.
Djordjevic L, Primorac M, Stupar M. In vitro release of diclofenac diethylamine from caprylocaproyl macrogolglycerides based microemulsions. Int J Pharm. 2005;296:73–9.
Ammar H, Salama A, Ghorab M, Mahmoud A. Nanoemulsion as a potential ophthalmic delivery system for dorzolamide hydrochloride. AAPS PharmSciTech. 2009;10(3):808–19.
Bachhav YG, Patravale VB. Microemulsion-based vaginal gel of clotrimazole: formulation, in vitro evaluation, and stability studies. AAPS PharmSciTech. 2009;10(2):476–81.
Hajkova R, Sklenarova H, Matysova L, Svecova P, Solich P. Development and validation of HPLC method for determination of clotrimazole and its two degradation products in spray formulation. Talanta. 2007;73:483–9.
Kirbaslar FG, Tavman A, Dulger B, Turker G. Antimicrobial activity of Turkish citrus peel oils. Pak J Bot. 2009;41(6):3207–12.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hashem, F.M., Shaker, D.S., Ghorab, M.K. et al. Formulation, Characterization, and Clinical Evaluation of Microemulsion Containing Clotrimazole for Topical Delivery. AAPS PharmSciTech 12, 879–886 (2011). https://doi.org/10.1208/s12249-011-9653-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9653-7