Abstract
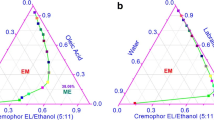
This project was carried out to investigate the feasibility of using microemulsions for transdermal delivery of lapachol. From the screening of surfactants and oils, a range of microemulsions were developed using oleic acid, a mixture of Cremophor EL and Tween 20 and water. The solubility of lapachol was determined in these ingredients and in the formulated microemulsions. The microemulsions were characterised using cross-polarising light microscopy, their electrical conductivity, pH, zeta potential and rheology were analysed, and they were also investigated using small-angle X-ray scattering and differential scanning calorimetry. Ex vivo studies were performed using porcine ear skin and Franz diffusion cells to investigate the permeation and retention of lapachol. Systems containing different concentrations of Cremophor EL (8.4–41.6%), Tween 20 (5.4–41.6%) and oleic acid (12–31.9%) are able to form microemulsions. Lapachol was delivered more effectively through the skin from all of the microemulsions tested than by the control (oleic acid). These studies indicated that microemulsions incorporating lapachol were formed successfully and that these enhanced drug delivery and retention in the skin. Microemulsion systems may, therefore, provide promising vehicles for percutaneous delivery of lapachol.





Similar content being viewed by others
Abbreviations
- DSC:
-
Differential scanning calorimetry
- HLB:
-
Hydrophilic-lipophilic balance
- HPLC:
-
High-performance liquid chromatography
- PEG 400:
-
Polyethylene glycol-400
- SAXS:
-
Small-angle X-ray scattering
References
Hussain H, Krohn K, Ahmad VU, Miana GA, Green IR. Lapachol: an overview. ARKIVOC. 2007;2:145–71.
Almeida ERD. Preclinical and clinical studies of lapachol and beta-lapachone. Open Nat Prod J. 2009;2(1):42–7.
Ferreira SB, Gonzaga DTG, Santos WC, Araújo KG, Ferreira VF. beta-Lapachona: Sua Importância em Química Medicinal e Modificações Estruturais. Rev Virtual Quím. 2010;2(2):140–60.
Tabosa MAM, de Melo EKS, de Souza Dantas IMF, de Santana DP, Leal LB. Physicochemical characterization of lapachol. Afr J Pharm Pharmacol. 2015;9(5):131–8.
de Almeida ER, da Silva Filho AA, Dos Santos ER, Lopes CAC. Antiinflammatory action of lapachol. J Ethnopharmacol. 1990;29(2):239–41.
Guerra MO, Mazoni A, Brandão M, Peters V. Toxicology of lapachol in rats: embryolethality. Rev Bras Biol. 2001;61(1):171–4.
de Andrade-Neto VF, Goulart MO, da Silva Filho JF, da Silva MJ, Pinto Mdo C, Pinto AV, et al. Antimalarial activity of phenazines from lapachol, beta-lapachone and its derivatives against Plasmodium falciparum in vitro and Plasmodium berghei in vivo. Bioorg Med Chem Lett. 2004;14(5):1145–9. https://doi.org/10.1016/j.bmcl.2003.12.069.
Peres RS, Santos GB, Cecilio NT, Jabor VA, Niehues M, Torres BG, et al. Lapachol, a compound targeting pyrimidine metabolism, ameliorates experimental autoimmune arthritis. Arthritis Res Ther. 2017;19(1):47.
Pisani DE, Elliot AJ, Hinman DR, Aaronson LM, Pardini RS. Relationship between inhibition of mitochondrial respiration by naphthoquinones, their antitumor activity, and their redox potential. Biochem Pharmacol. 1986;35(21):3791–8.
Lira AA, Sester EA, Carvalho AL, Strattmann RR, Albuquerque MM, Wanderley AG, et al. Development of lapachol topical formulation: anti-inflammatory study of a selected formulation. AAPS PharmSciTech. 2008;9(1):163–8. https://doi.org/10.1208/s12249-007-9002-z.
Lima NM, Correia CS, Leon LL, Machado G, Madeira MF, Santana AEG, et al. Antileishmanial activity of lapachol analogues. Mem Inst Oswaldo Cruz. 2004;99(7):757–61.
Shakeel F, Ramadan W. Transdermal delivery of anticancer drug caffeine from water-in-oil nanoemulsions. Colloids Surf B: Biointerfaces. 2010;75(1):356–62. https://doi.org/10.1016/j.colsurfb.2009.09.010.
Chaiyana W, Rades T, Okonogi S. Characterization and in vitro permeation study of microemulsions and liquid crystalline systems containing the anticholinesterase alkaloidal extract from Tabernaemontana divaricata. Int J Pharm. 2013;452(1–2):201–10. https://doi.org/10.1016/j.ijpharm.2013.05.005.
Hathout RM, Woodman TJ, Mansour S, Mortada ND, Geneidi AS, Guy RH. Microemulsion formulations for the transdermal delivery of testosterone. Eur J Pharm Sci. 2010;40(3):188–96. https://doi.org/10.1016/j.ejps.2010.03.008.
Fouad SA, Basalious EB, El-Nabarawi MA, Tayel SA. Microemulsion and poloxamer microemulsion-based gel for sustained transdermal delivery of diclofenac epolamine using in-skin drug depot: in vitro/in vivo evaluation. Int J Pharm. 2013;453(2):569–78. https://doi.org/10.1016/j.ijpharm.2013.06.009.
Zhao JH, Ji L, Wang H, Chen ZQ, Zhang YT, Liu Y, et al. Microemulsion-based novel transdermal delivery system of tetramethylpyrazine: preparation and evaluation in vitro and in vivo. Int J Nanomedicine. 2011;6:1611–9. https://doi.org/10.2147/IJN.S23597.
Nikumbh KV, Sevankar SG, Patil MP. Formulation development, in vitro and in vivo evaluation of microemulsion-based gel loaded with ketoprofen. Drug Delivery. 2015;22(4):509–215. https://doi.org/10.3109/10717544.2013.859186.
Friberg SE. Micelles, microemulsions, liquid crystals, and the structure of stratum corneum lipids. J Soc Cosmet Chem. 1990;41(3):155–71.
Yu A, Wang H, Wang J, Cao F, Gao Y, Cui J, et al. Formulation optimization and bioavailability after oral and nasal administration in rabbits of puerarin-loaded microemulsion. J Pharm Sci. 2011;100(3):933–41. https://doi.org/10.1002/jps.22333.
Hosny KM, Hassan AH. Intranasal in situ gel loaded with saquinavir mesylate nanosized microemulsion: preparation, characterization, and in vivo evaluation. Int J Pharm. 2014;475(1–2):191–7. https://doi.org/10.1016/j.ijpharm.2014.08.064.
Moser K, Kriwet K, Naik A, Kalia YN, Guy RH. Passive skin penetration enhancement and its quantification in vitro. Eur J Pharm Biopharm. 2001;52(2):103–12.
Azeem A, Ahmad FJ, Khar RK, Talegaonkar S. Nanocarrier for the transdermal delivery of an antiparkinsonian drug. AAPS PharmSciTech. 2009;10(4):1093–103.
Boelsma E, Tanojo H, Boddé H, Ponec M. Assessment of the potential irritancy of oleic acid on human skin: evaluation in vitro and in vivo. Toxicol in Vitro. 1996;10(6):729–42.
Martins M, Veiga F. Promotores de permeação para a liberação transdérmica de fármacos: uma nova aplicação para as ciclodextrinas. Braz J Pharm Sci. 2002;38(1):33–54.
Talegaonkar S, Azeem A, Ahmad FJ, Khar RK, Pathan SA, Khan ZI. Microemulsions: a novel approach to enhanced drug delivery. Recent Pat Drug Deliv Formul. 2008;2(3):238–57.
Moghassemi S, Hadjizadeh A. Nano-niosomes as nanoscale drug delivery systems: an illustrated review. J Control Release. 2014;185:22–36.
Rege BD, Kao JP, Polli JE. Effects of nonionic surfactants on membrane transporters in Caco-2 cell monolayers. Eur J Pharm Sci. 2002;16(4):237–46.
Bhatia G, Zhou Y, Banga AK. Adapalene microemulsion for transfollicular drug delivery. J Pharm Sci. 2013;102(8):2622–31. https://doi.org/10.1002/jps.23627.
Patel HK, Barot BS, Parejiya PB, Shelat PK, Shukla A. Topical delivery of clobetasol propionate loaded microemulsion based gel for effective treatment of vitiligo: ex vivo permeation and skin irritation studies. Colloids Surf B: Biointerfaces. 2013;102:86–94. https://doi.org/10.1016/j.colsurfb.2012.08.011.
Baroli B, López-Quintela MA, Delgado-Charro MB, Fadda AM, Blanco-Méndez J. Microemulsions for topical delivery of 8-methoxsalen. J Control Release. 2000;69(1):209–18.
Epifano F, Genovese S, Fiorito S, Mathieu V, Kiss R. Lapachol and its congeners as anticancer agents: a review. Phytochem Rev. 2013;13(1):37–49. https://doi.org/10.1007/s11101-013-9289-1.
Hashem FM, Shaker DS, Ghorab MK, Nasr M, Ismail A. Formulation, characterization, and clinical evaluation of microemulsion containing clotrimazole for topical delivery. AAPS PharmSciTech. 2011;12(3):879–86. https://doi.org/10.1208/s12249-011-9653-7.
Chen Y, Liu J, Yang X, Zhao X, Xu H. Oleanolic acid nanosuspensions: preparation, in-vitro characterization and enhanced hepatoprotective effect. J Pharm Pharmacol. 2005;57(2):259–64. https://doi.org/10.1211/0022357055407.
Meagher MJ, Leone B, Turnbull TL, Ross RD, Zhang Z, Roeder RK. Dextran-encapsulated barium sulfate nanoparticles prepared for aqueous dispersion as an X-ray contrast agent. J Nanopart Res. 2013;15:1246. https://doi.org/10.1007/s11051-013-2146-8.
Ge S, Lin Y, Lu H, Li Q, He J, Chen B, et al. Percutaneous delivery of econazole using microemulsion as vehicle: formulation, evaluation and vesicle-skin interaction. Int J Pharm. 2014;465(1–2):120–31. https://doi.org/10.1016/j.ijpharm.2014.02.012.
Podlogar F, Gasperlin M, Tomsic M, Jamnik A, Rogac MB. Structural characterisation of water-Tween 40/Imwitor 308-isopropyl myristate microemulsions using different experimental methods. Int J Pharm. 2004;276(1–2):115–28. https://doi.org/10.1016/j.ijpharm.2004.02.018.
Mitra RK, Paul BK. Physicochemical investigations of microemulsification of eucalyptus oil and water using mixed surfactants (AOT+ Brij-35) and butanol. J Colloid Interface Sci. 2005;283(2):565–77.
Boonme P, Krauel K, Graf A, Rades T, Junyaprasert VB. Characterization of microemulsion structures in the pseudoternary phase diagram of isopropyl palmitate/water/Brij 97: 1-butanol. AAPS PharmSciTech. 2006;7(2):E99–E104.
Podlogar F, Bester Rogac M, Gasperlin M. The effect of internal structure of selected water-Tween 40-Imwitor 308-IPM microemulsions on ketoprofene release. Int J Pharm. 2005;302(1–2):68–77. https://doi.org/10.1016/j.ijpharm.2005.06.023.
Mouri A, Diat O, El Ghzaoui A, Bauer C, Maurel JC, Devoisselle JM, et al. Phase behavior of reverse microemulsions based on Peceol((R)). J Colloid Interface Sci. 2014;416:139–46. https://doi.org/10.1016/j.jcis.2013.10.058.
Regev O, Ezrahi S, Aserin A, Garti N, Wachtel E, Kaler E, et al. A study of the microstructure of a four-component nonionic microemulsion by cryo-TEM, NMR, SAXS, and SANS. Langmuir. 1996;12(3):668–74.
Sintov AC, Greenberg I. Comparative percutaneous permeation study using caffeine-loaded microemulsion showing low reliability of the frozen/thawed skin models. Int J Pharm. 2014;471(1):516–24.
Peltola S, Saarinen-Savolainen P, Kiesvaara J, Suhonen TM, Urtti A. Microemulsions for topical delivery of estradiol. Int J Pharm. 2003;254(2):99–107. https://doi.org/10.1016/s0378-5173(02)00632-4.
Lane ME. Skin penetration enhancers. Int J Pharm. 2013;447(1–2):12–21. https://doi.org/10.1016/j.ijpharm.2013.02.040.
Acknowledgements
The authors thank the Laboratório Nacional de Luz Síncroton (LNLS, Campinas SP, Brazil). LNLS is acknowledged through the projects D11A-SAXS1-14558. We especially thank SAXS staff for the support. The authors also thank FECDA/NUDFAC for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tabosa, M.A.M., de Andrade, A.R.B., Lira, A.A.M. et al. Microemulsion Formulations for the Transdermal Delivery of Lapachol. AAPS PharmSciTech 19, 1837–1846 (2018). https://doi.org/10.1208/s12249-018-0995-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-0995-2