Abstract
Background
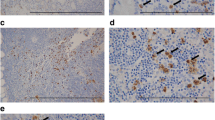
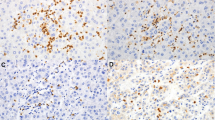
Tumor-infiltrating neutrophils (TINs) indicate poor prognosis for patients with pancreatic ductal adenocarcinoma (PDAC). Activated neutrophils can generate neutrophil extracellular traps (NETs). Little is known about the presence and prognostic significance of tumor-infiltrating NETs in PDAC.
Methods
This study enrolled 317 patients, in two independent sets (training and validation), who underwent curative pancreatectomy for PDAC in Shanghai Cancer Center. TINs and NETs were identified by immunohistochemical staining for CD15 and citrullinated histone H3, respectively. The relationship between clinicopathological features and outcomes was analyzed. Accuracy of prognostic prediction models was evaluated using concordance index (C-index) and Akaike information criterion (AIC).
Results
NETs were associated with OS (both, P < 0.001) and RFS (both, P < 0.001) in the training and validation sets. Tumor-infiltrating NETs predicted poor postsurgical survival of patients with PDAC. Moreover, multivariate analysis identified NETs and AJCC TNM stage as two independent prognostic factors for OS and RFS. Combination of NETs with the 8th edition TNM staging system (C-index, 0.6994 and 0.6669, respectively; AIC, 1067 and 1126, respectively) generated a novel model that improved the predictive accuracy for survival in both sets (C-index, 0.7254 and 0.7117, respectively; AIC, 1047 and 1102, respectively). The model combining presence of NETs with the 7th edition AJCC TNM staging system also had improved predictive accuracy.
Conclusions
NETs were an independent prognostic factor in PDAC and incorporation of NETs along with the standard TNM stating system refined risk-stratification and predicted survival in PDAC with improved accuracy.


Similar content being viewed by others
References
Wolfgang CL, Herman JM, Laheru DA, et al. Recent progress in pancreatic cancer. Ca Cancer J Clin 2013;63(5):318.
Kamisawa T, Wood LD, Itoi T, Takaori K. Pancreatic cancer. Lancet. 2016;388(10039):73.
Diakos CI, Charles KA, Mcmillan DC, Clarke SJ. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15(11):e493–503.
Wculek SK, Malanchi I. Neutrophils support lung colonization of metastasis-initiating breast cancer cells. Nature. 2015;528(7582):413–7.
Spiegel A, Brooks MW, Houshyar S, et al. Neutrophils suppress intraluminal NK cell-mediated tumor cell clearance and enhance extravasation of disseminated carcinoma cells. Cancer Discovery. 2016;6(6):630.
Granot Z, Henke E, Comen EA, King TA, Norton L, Benezra R. Tumor entrained neutrophils inhibit seeding in the premetastatic lung. Cancer Cell. 2011;20(3):300–14.
Yang JJ, Hu ZG, Shi WX, Deng T, He SQ, Yuan SG. Prognostic significance of neutrophil to lymphocyte ratio in pancreatic cancer: a meta-analysis. World J Gastroenterol. 2015;21(9):2807–15.
Cheng H, Luo G, Lu Y, et al. The combination of systemic inflammation-based marker NLR and circulating regulatory T cells predicts the prognosis of resectable pancreatic cancer patients. Pancreatology. 2016;16(6):1080–4.
Pulze L, Bassani B, Gini E, et al. NET amyloidogenic backbone in human activated neutrophils. Clin Exp Immunol. 2016;183(3):469–79.
Korabecna M, Tesar V. NETosis provides the link between activation of neutrophils on hemodialysis membrane and comorbidities in dialyzed patients. Inflamm Res. 2017;66(5):1–10.
Bonaventura A, Liberale L, Carbone F, et al. The pathophysiological role of neutrophil extracellular traps in inflammatory diseases. Thromb Haemost. 2018;118(1):6.
Berger-Achituv S, Brinkmann V, Abed UA, et al. A proposed role for neutrophil extracellular traps in cancer immunoediting. Frontiers Immunol. 2013;4:48.
Tohme S, Yazadani HO, Al-Khafaji AB, et al. Neutrophil extracellular traps promote the development and progression of liver metastases after surgical stress. Cancer Res. 2016;76(6):1367.
Merza M, Hartman H, Rahman M, et al. Neutrophil extracellular traps induce trypsin activation, inflammation, and tissue damage in mice with severe acute pancreatitis. Gastroenterology. 2015;149(7):1920–31.
Yang C, Sun W, Wu C, et al. Procoagulant role of neutrophil extracellular traps in patients with gastric cancer. Int J Clin Exp Pathol. 2015;8(11):14075–86.
Cools-Lartigue J, Spicer J, Najmeh S, Ferri L. Neutrophil extracellular traps in cancer progression. Cell Molec Life Sci. 2014;71(21):4179–94.
Zhao J, Pan K, Wang W, et al. The prognostic value of tumor-infiltrating neutrophils in gastric adenocarcinoma after resection. Plos One. 2015;7(3):e33655.
Perisanidis C, Kornek G, Pöschl PW, et al. High neutrophil-to-lymphocyte ratio is an independent marker of poor disease-specific survival in patients with oral cancer. Med Oncol. 2013;30(1):1–8.
Wang WQ, Liu L, Xu HX, et al. Infiltrating immune cells and gene mutations in pancreatic ductal adenocarcinoma. Br J Surg. 2016;103(9):1189–99.
Wang WQ, Liu L, Xu HX, et al. The combination of HTATIP2 expression and microvessel density predicts converse survival of hepatocellular carcinoma with or without sorafenib. Oncotarget. 2014;5(11):3895–906.
Wang WQ, Liu L, Xu HX, et al. Intratumoral α-SMA enhances the prognostic potency of CD34 associated with maintenance of microvessel integrity in hepatocellular carcinoma and pancreatic cancer. Plos One. 2013;8(8):e71189.
Wei J, Liao X, Lv Y, et al. MUC1 induces acquired chemoresistance by upregulating ABCB1 in EGFR-dependent manner. Cell Death Dis. 2017;8(8):e2980.
Arelaki S, Arampatzioglou A, Kambas K, et al. Gradient infiltration of neutrophil extracellular traps in colon cancer and evidence for their involvement in tumour growth. Plos One. 2016;11(5):e0154484.
Thålin C, Demers M, Blomgren B, et al. NETosis promotes cancer-associated arterial microthrombosis presenting as ischemic stroke with troponin elevation. Thromb Res. 2016;139:56.
Branzk N, Papayannopoulos V. Molecular mechanisms regulating NETosis in infection and disease. Sem Immunopathol. 2013;35(4):513.
Volker B, Arturo Z. Neutrophil extracellular traps: Is immunity the second function of chromatin? J Cell Biol. 2012;198(5):773–83.
Park J, Wysocki RW, Amoozgar Z, et al. Cancer cells induce metastasis-supporting neutrophil extracellularDNA traps. Sci Transl Med. 2016;8(361):361ra138.
Vallés J, Lago A, Santos MT, et al. Neutrophil extracellular traps are increased in patients with acute ischemic stroke: prognostic significance. Thromb Haemost. 2017;117(10):1919–29.
Chowdhury CS, Giaglis S, Walker UA, Buser A, Hahn S, Hasler P. Enhanced neutrophil extracellular trap generation in rheumatoid arthritis: analysis of underlying signal transduction pathways and potential diagnostic utility. Arthritis Res Ther. 2014;16(3):R122.
Stoikou M, Grimolizzi F, Giaglis S, et al. Gestational diabetes mellitus is associated with altered neutrophil activity. Frontiers Immunol. 2017;8.
Pan B, Alam HB, Wei C, et al. CitH3: a reliable blood biomarker for diagnosis and treatment of endotoxic shock. Scientific Rep. 2017;7(1):8972.
Cedervall J, Zhang Y, Huang H, et al. Neutrophil extracellular traps accumulate in peripheral blood vessels and compromise organ function in tumor-bearing animals. Cancer Res. 2015;75(13):2653–62.
Shak S, Capon DJ, Hellmiss R, Marsters SA, Baker CL. Recombinant Human DNase I Reduces the Viscosity of Cystic Fibrosis Sputum. Proc Natl Acad Sci U S A. 1990;87(23):9188.
Li YW, Qiu SJ, Fan J, et al. Intratumoral neutrophils: a poor prognostic factor for hepatocellular carcinoma following resection. J Hepatol. 2011;54(3):497.
Caruso RA, Bellocco R, Pagano M, Bertoli G, Rigoli L, Inferrera C. Prognostic value of intratumoral neutrophils in advanced gastric carcinoma in a high-risk area in northern Italy. Mod Pathol. 2002;15(8):831–7.
Fridlender ZG, Albelda SM. Tumor-associated neutrophils: friend or foe? Carcinogenesis. 2012;33(5):949–55.
Sionov RV, Fridlender ZG, Granot Z. The multifaceted roles neutrophils play in the tumor microenvironment. Cancer Microenviron. 2015;8(3):125–58.
Zhang X, Zhang W, Yuan X, Fu M, Qian H, Xu W. Neutrophils in cancer development and progression: Roles, mechanisms, and implications (Review). Int J Oncol. 2016;49(3):857.
Zhang H, Liu H, Shen Z, et al. Tumor-infiltrating neutrophils is prognostic and predictive for postoperative adjuvant chemotherapy benefit in patients with gastric cancer. Ann Surg. 2016:1.
Powell DR, Huttenlocher A. Neutrophils in the tumor microenvironment. Trends Immunol. 2016;37(1):41–52.
Sørensen OE, Borregaard N. Neutrophil extracellular traps—the dark side of neutrophils. J Clin Invest. 2016;126(5):1612–20.
Acknowledgments
This work was supported by grants from the National Science Foundation for Distinguished Young Scholars of China (81625016), the National Natural Science Foundation of China (81472670, 81871941, 81872366, 81802380, and 81802675), the Outstanding Academic Leader Program of the “Technological Innovation Action Plan” in Shanghai Science and Technology Commission (18XD1401200), and the Young Talented Specialist Training Program of Shanghai.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosure
The authors declare no conflicts of interest in this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jin, W., Xu, HX., Zhang, SR. et al. Tumor-Infiltrating NETs Predict Postsurgical Survival in Patients with Pancreatic Ductal Adenocarcinoma. Ann Surg Oncol 26, 635–643 (2019). https://doi.org/10.1245/s10434-018-6941-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6941-4