Abstract
Background
Several cohort studies have reported that post-esophagectomy morbidities may worsen prognosis. Smoking cessation is an effective prophylactic measure for reducing post-esophagectomy morbidity; however, whether smoking cessation can contribute to the improvement of prognosis is unknown due to the absence of reliable databases covering the cessation period. This study aimed to elucidate whether sufficient preoperative smoking cessation can improve prognosis after esophageal cancer surgery by reducing post-esophagectomy morbidity.
Methods
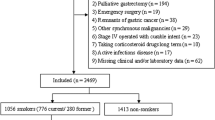
This study included 544 consecutive patients who underwent curative McKeown and Ivor–Lewis esophagectomies for esophageal cancer between May 2011 and June 2021. Data on smoking status and cessation period were prospectively accumulated. Survival data were finally updated on 30 January 2022. Receiver operating characteristic curve analysis for the cut-off value of appropriate cessation period in reducing post-esophagectomy respiratory morbidity as well as analyses for the association of cessation period with short- and long-term outcomes were performed.
Results
Post-esophagectomy morbidity significantly diminished overall survival (OS) after esophagectomy (p = 0.0003). A short preoperative smoking cessation period of ≤ 2 months was associated with frequent post-esophagectomy morbidity of Clavien–Dindo classification ≥IIIb (p = 0.0059), pneumonia (p = 0.016), respiratory morbidity (p = 0.0057), and poor OS in clinical stages II and III (p = 0.0015). Moreover, it was an independent factor for poor OS (hazard ratio 1.85, 95% confidence interval 1.068–3.197; p = 0.028), along with body mass index <18.5 and R1 resection.
Conclusions
Sufficient preoperative smoking cessation > 2 months may be effective in improving not only short-term outcomes but also prognosis after esophagectomy for locally advanced esophageal cancer.



Similar content being viewed by others
References
Yoshida N, Yamamoto H, Baba H, et al. Can minimally invasive esophagectomy replace open esophagectomy for esophageal cancer? Latest analysis of 24,233 esophagectomies from the Japanese national clinical database. Ann Surg. 2020;272:118–24.
Yoshida N, Kosumi K, Tokunaga R, et al. Clinical importance of mean corpuscular volume as a prognostic marker after esophagectomy for esophageal cancer: a retrospective study. Ann Surg. 2020;271:494–501.
Baba Y, Yoshida N, Shigaki H, et al. Prognostic impact of postoperative complications in 502 patients with surgically resected esophageal squamous cell carcinoma: a retrospective single-institution study. Ann Surg. 2016;264:305–11.
Yoshida N, Baba Y, Hiyoshi Y, et al. Duration of smoking cessation and postoperative morbidity after esophagectomy for esophageal cancer: how long should patients stop smoking before surgery? World J Surg. 2016;40:142–7.
Kamarajah SK, Madhavan A, Chmelo J, et al. Impact of smoking status on perioperative morbidity, mortality, and long-term survival following transthoracic esophagectomy for esophageal cancer. Ann Surg Oncol. 2021;28:4905–15.
Rice TW, Ishwaran H, Ferguson MK, Blackstone EH, Goldstraw P. Cancer of the esophagus and esophagogastric junction: an eighth edition staging primer. J Thorac Oncol. 2017;12:36–42.
Kuwano H, Nishimura Y, Oyama T, et al. Guidelines for diagnosis and treatment of carcinoma of the esophagus April 2012 edited by the Japan Esophageal Society. Esophagus. 2015;12:1–30.
Yoshida N, Baba Y, Shigaki H, et al. Preoperative nutritional assessment by controlling nutritional status (CONUT) is useful to estimate postoperative morbidity after esophagectomy for esophageal cancer. World J Surg. 2016;40:1910–7.
Yoshida N, Nakamura K, Kuroda D, et al. Preoperative Smoking Cessation is integral to the prevention of postoperative morbidities in minimally invasive esophagectomy. World J Surg. 2018;42:2902–9.
Mazaki T, Ishii Y, Murai I. Immunoenhancing enteral and parenteral nutrition for gastrointestinal surgery: a multiple-treatments meta-analysis. Ann Surg. 2015;261:662–9.
Yamana I, Takeno S, Hashimoto T, et al. Randomized controlled study to evaluate the efficacy of a preoperative respiratory rehabilitation program to prevent postoperative pulmonary complications after esophagectomy. Dig Surg. 2015;32:331–7.
Akutsu Y, Matsubara H, Shuto K, et al. Pre-operative dental brushing can reduce the risk of postoperative pneumonia in esophageal cancer patients. Surgery. 2010;147:497–502.
Tsujimoto H, Kobayashi M, Sugasawa H, Ono S, Kishi Y, Ueno H. Potential mechanisms of tumor progression associated with postoperative infectious complications. Cancer Metastasis Rev. 2021;40:285–96.
Kauppila JH, Johar A, Lagergren P. Postoperative complications and health-related quality of life 10 years after esophageal cancer surgery. Ann Surg. 2020;271:311–6.
Watanabe M, Ishimoto T, Baba Y, et al. Prognostic impact of body mass index in patients with squamous cell carcinoma of the esophagus. Ann Surg Oncol. 2013;20:3984–91.
Yoshida N, Harada K, Baba Y, et al. Preoperative controlling nutritional status (CONUT) is useful to estimate the prognosis after esophagectomy for esophageal cancer. Langenbecks Arch Surg. 2017;402:333–41.
Klevebro F, Elliott JA, Slaman A, et al. Cardiorespiratory comorbidity and postoperative complications following esophagectomy: a European Multicenter Cohort Study. Ann Surg Oncol. 2019;26:2864–73.
van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–84.
Ando N, Kato H, Igaki H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19:68–74.
Demarest CT, Chang AC. The landmark series: multimodal therapy for esophageal cancer. Ann Surg Oncol. 2021;28:3375–82.
Yoshida N, Baba Y, Kuroda D, et al. Clinical utility of exhaled carbon monoxide in assessing preoperative smoking status and risks of postoperative morbidity after esophagectomy. Dis Esophagus. 2018; 31.
Triantafyllou T, Olson MT, Theodorou D, Schizas D, Singhal S. Enhanced recovery pathways vs standard care pathways in esophageal cancer surgery: systematic review and meta-analysis. Esophagus. 2020;17:100–12.
Kawata S, Hiramatsu Y, Shirai Y, et al. Multidisciplinary team management for prevention of pneumonia and long-term weight loss after esophagectomy: a single-center retrospective study. Esophagus. 2020;17:270–8.
Watanabe M, Mine S, Nishida K, et al. Improvement in short-term outcomes after esophagectomy with a multidisciplinary perioperative care team. Esophagus. 2016;13:337–42.
Chen Z, Huang K, Wei R, et al. Transcervical inflatable mediastinoscopic esophagectomy versus thoracoscopic esophagectomy for local early- and intermediate-stage esophageal squamous cell carcinoma: a propensity score-matched analysis. J Surg Oncol. 2022;125:839–46.
Mederos MA, de Virgilio MJ, Shenoy R, et al. Comparison of clinical outcomes of robot-assisted, video-assisted, and open esophagectomy for esophageal cancer: a systematic review and meta-analysis. JAMA Netw Open. 2021;4:e2129228.
Acknowledgments
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Naoya Yoshida and Takatsugu Ishimoto are members of the department supported by Chugai Pharmaceutical Co., Ltd and Yakuruto Honsya Co., Ltd, but have no conflicts of interest regarding this research. Yoshifumi Baba is a member of the department supported by Taiho Pharmaceutical Co., Ltd, but has no conflicts of interest regarding this research. Hideo Baba, Kojiro Eto, Tomo Horinouchi, Kazuto Harada, Hiroshi Sawayama, Katsuhiro Ogawa, Yohei Nagai, Masaaki Iwatsuki, Shiro Iwagami, and Yuji Miyamoto have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10434_2022_12433_MOESM2_ESM.pptx
Supplementary file 2. Fig. S1 Kaplan–Meier curves of overall survival between smokers and non-smokers in (a) all patients, (b) clinical stage IB, (c) clinical stage II and III, and (d) clinical stage IVA
10434_2022_12433_MOESM3_ESM.pptx
Supplementary file 3. Fig. S2 Kaplan–Meier curves of overall survival in clinical stage I in accordance with the appropriate duration of smoking cessation calculated by the Youden index
10434_2022_12433_MOESM4_ESM.pptx
Supplementary file 4. Fig. S3 Kaplan–Meier curves of overall survival in clinical stage IVa in accordance with the appropriate duration of smoking cessation calculated by the Youden index
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshida, N., Eto, K., Horinouchi, T. et al. Preoperative Smoking Cessation and Prognosis After Curative Esophagectomy for Esophageal Cancer: A Cross-Sectional Study. Ann Surg Oncol 29, 8172–8180 (2022). https://doi.org/10.1245/s10434-022-12433-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-12433-z