Abstract
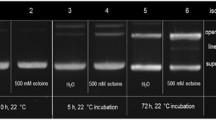
Copper [Cu(II)] is an ubiquitous transition and trace element in living organisms. It increases reactive oxygen species (ROS) and free-radical generation that might damage biomolecules like DNA, proteins, and lipids. Furthermore, ability of Cu(II) greatly increases in the presence of oxidants. ROS, like hydroxyl (·OH) and superoxide (·O2) radicals, alter both the structure of the DNA double helix and the nitrogen bases, resulting in mutations like the AT→GC and GC→AT transitions. Proteins, on the other hand, suffer irreversible oxidations and loss in their biological role. Thus, the aim of this investigation is to characterize, in vitro, the structural effects caused by ROS and Cu(II) on bacteriophage λ DNA or proteins using either hydrogen peroxide (H2O2) or ascorbic acid with or without Cu(II). Exposure of DNA to ROS-generating mixtures results in electrophoretic (DNA breaks), spectrophotometric (band broadening, hypochromic, hyperchromic, and bathochromic effects), and calorimetric (denaturation temperature [T d], denaturation enthalpy [ΔH], and heat capacity [C p] values) changes. As for proteins, ROS increased their thermal stability. However, the extent of the observed changes in DNA and proteins were distinct, depending on the efficiency of the systems assayed to generate ROS. The resulting effects were most evident when Cu(II) was present. In summary, these results show that the ROS, ·O2 and ·OH radicals, generated by the Cu(II) systems assayed deeply altered the chemical structure of both DNA and proteins. The physiological relevance of these structural effects should be further investigated.
Similar content being viewed by others
References
I. Fridovich, Quantitative aspects of the production of superoxide anion radical by xanthine oxidase, J. Biol. Chem. 245, 4053–4057 (1970).
J. M. C. Gutteridge, Biological origin of free radicals, and mechanisms of antioxidant protection, Chem.-Biol. Interact. 91, 133–140 (1994).
O. I. Aruoma, B. Halliwell, E. Gajewski, and M. Dizdaroglu, Copper-iron dependent damage to the bases in DNA in the presence of hydrogen peroxide, Biochem. J. 273, 601–604 (1991).
M. Dizdaroglu, O. I. Aruoma, and B. Halliwell, Modification of bases in DNA by copper ion-1,10-phenantroline complexes, Biochemistry 29(36), 8447–8451 (1990).
A. J. F. Griffiths, J. H. Miller, D. T. Suzuki, R. C. Lewontin, and R. C. Gelbart, Genetic Analysis, 5th ed., W. H. Freeman, New York, (1993).
J. R. Wagner, J. E. van Lier, C. Decarroz, M. Berger, and J. Cadet, Photodynamic methods for oxyradical-induced DNA damage, in P. Lester and Al. Glazer (eds.), Oxygen Radicals in Biological Systems, Part B, Academic, Chichester (1990).
K. Kubo, H. Ide, S. S. Wallace, and Y. W. Kow A novel sensitive and specific assay for abasic sites, the most commonly produced DNA lesion, Biochemistry 31(14), 3703–3708 (1992).
R. I. Salganik, I. G. Shabalina, N. A. Solovyova, N. G. Kolosova, V. N. Solovyov, and A. R. Kolpakov, Impairment of respiratory functions in mitochondria of rats with an inherited hyperproduction of free radicals, Biochem. Biophys. Res. Commun. 205(1), 180–185 (1994).
R. Krishnamurthy, R. D. Madurawe, K. D. Bush, and J. A. Lumpkin, Conditions promoting metal-catalized oxidations during immobilized Cu-iminodiacetic acid metal affinity chromatography, Biotechnol. Prog. 11(6), 643–650 (1995).
B. Geierstanger, T. F. Kagawa, S. L. Chen, G. J. Quigley, and P. S. Ho, Base specific binding of copper (II) to Z-DNA, J. Biol. Chem. 266, 20,185–20,191 (1991).
D. M. Miller, G. R. Buettner, and S. D. Aust, Transition metals as catalyst of “autoxidation” reactions, Free Radical Res. Biol. Med. 8, 95–108 (1990).
R. P. Hertzberg and P. B. Dervan, Cleavage of DNA with methydiumpropyl-EDTA-iron(II): reaction conditions and product analyses, Biochemistry 23(17), 3934–3945 (1984).
Y. Wang, and B. Van Ness, Site-specific cleavage of supercoiled DNA by ascorbate/Cu(II), Nucleic Acids Res. 17(17), 6915–6926 (1989).
G. J. Quinlan, T. W. Evans, and J. M. C. Gutteridge, Oxidative damage to plasma proteins in adult respiratory distress syndrome, Free Radical Res. 20(5), 289–298 (1994).
J. M. C. Gutteridge, Invited review. Free radicals in disease processes: a compilation of cause and consequence, Free Radical Res. Commun. 19(3), 141–158 (1993).
X. Huang, C. S. Atwood, M. A. Hartshorn, et al., The Aβ peptide of Alzheimer's disease directly produces hydrogen peroxide through metal ion reduction, Biochemistry 38(24), 7609–7616 (1999).
T. D. Tullius, and B. A. Dombroski, Iron(II) used to measure the helical twist along any DNA molecule, Science 230, 679–681 (1985).
W. Xu, S. Rao, B. Jampani, H. Deng, and W. H. Braunlin, Chiral recognition of deoxynucleotides by Δ- and Λ-Tris (ethylenediamine)cobalt(III), Biochemistry 34 14,059–14,065 (1995).
H. Rodríguez, G. P. Holmquist, R. D'Agostino, Jr., J. Séller, and S. A. Akman, Metal ion-dependent hydrogen peroxide-induced DNA damage is more sequence specific than metal specific, Cancer Res. 57(12), 2394–2403 (1997).
I. Smirnov and R. H. Shafer, Lead is unusually effective in sequence-specific folding of DNA, J. Mol. Biol. 296, 1–5 (2000).
M. S. Torres, Efecto protector del ácido ascórbico-Cu++ contra la infección del bacteriófago lambda sobre Escherichia coli 837. BSc Thesis. ENCB-IPN, México City (1996).
J. L. Muñoz, P. A. Loyola, and D. Sandoval, Bacteriophage inactivation by ascorbic acid and thiol reducing agents: synergic effect with copper and iron, Ann. Esc. Nac. Cien. Biol. 37, 121–131 (1992).
A. Murata, K. Kitagawa, H. Inmaru, and R. Saruno, Inactivation of single-stranded DNA and RNA phages by ascorbic acid and thiol reducing agents, Agric. Biol. Chem. 36(13), 2597–2599 (1972).
A. Murata, H. Suenega, S. Hideshima, Y. Tanaka, and F. Kato, Hydroxyl radicals as the reactive species in the inactivation of phages by ascorbic acid, Agric. Biol. Chem. 50(6), 1481–1487 (1986).
C. Winterbourn, Oxidative reactions of hemoglobin, Methods Enzymol. 186, 265–278 (1990).
R. J. Henry, et al., Clinical Chemistry. Principles and Techniques, 2nd ed., Harper and Row, New York, p. 1288 (1974).
H. Kaur and B. Halliwell, Salicylic acid and phenilalanine as probes to detect hydroxyl radicals, in Free Radicals. A Practical Approach, N. A. Punchard and F. J. Kelly, eds., Oxford University Press, New York, pp. 101–116 (1996).
B. Perbal, A Practical Guide to Molecular Cloning, 2nd ed., Wiley, New York (1988).
C. N. Rao, Ultra-violet and Visible Spectroscopy. Chemical Applications, 3rd ed., Butterworths, Boston (1975).
Anonymous. Shimadzu Manual for the Stand-alone Thermal Analysis Instrument. DSC-50, Differential Scanning Calorimeter.
S. H. Chiou, DNA—and protein-scission activities of ascorbate in the presence of copper ion and a copper-peptide complex, J. Biochem. (Tokyo) 94(4), 1259–1267 (1983).
F. R. Klenner, Observations on the dose and administration of ascorbic acid when employe beyond the range of A vitamin inhuman pathology, J. Appl. Nutr. 23(3–4), 61–88 (1971).
J. L. Sagripanti and K. H. Kraemer, Site-specific DNA damage at polyguanosines produced by copper plus hydrogen peroxide, J. Biol. Chem. 264, 1729–1734 (1989).
J. Howard-Parish, Principles and Practice of Experiments with Nucleic Acids. Longman, London (1972).
I. Jelesarov, C. Crane-Robinson, and P. L. Privalov, The energetics of HMG Box interactions with DNA: thermodynamic description of the target DNA duplexes, J. Mol. Biol. 294, 981–995 (1999).
B. Chowdhry and S. C. Cole, Differential scanning calorimetry: applications in biotechnology, TiBtech. 7, 11–18 (1989).
G. Vogt, S. Woell, and P. Argos, Protein thermal stability, hydrogen bonds and ion pairs, J. Mol. Biol. 269, 631–643 (1997).
K. L. Malisza, A. R. McIntosh, E. Sveinson, and B. B. Hasinoff, Semiquinone free radical formation by daunorubicin aglycone incorporated into cellular membranes of intact chinese hamster ovary cells, Free Radical Res. 24(1), 9–18 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cervantes-Cervantes, M.P., Calderón-Salinas, J.V., Albores, A. et al. Copper increases the damage to DNA and proteins caused by reactive oxygen species. Biol Trace Elem Res 103, 229–248 (2005). https://doi.org/10.1385/BTER:103:3:229
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:103:3:229