Abstract
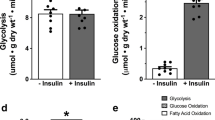
Cardiac glucose metabolism is critical to hypoxic cardiac function and hypoxia is known to stimulate glucose metabolism. This increases generation of ATP when mitochondrial respiration is inhibited. In diabetes, cardiac glucose metabolism declines and this may contribute to diabetic cardiomyopathy. The first step in committing glucose to metabolism is glucose phosphorylation catalyzed by hexokinase. But the potential role of hexokinase in the hypoxic or diabetic heart is uncertain. This study is designed to assess the ability of hexokinase and elevated ATP to protect cardiomyocyte contractility from hypoxia and diabetes. We used cardiomyocytes from the transgenic mouse Mh, which has cardiac specific expression of yeast hexokinase, to investigate the importance of glucose phosphorylation in the myocyte response to hypoxia and diabetes. Cardiomyocytes were isolated from FVB control and Mh hearts to assess the effects of 2h of hypoxia on myocyte contractility and ATP conent. The protective effect of hexokinase on diabetes was assessed in myocytes from the OVE26 Type I diabetic mouse and in OVE26Mh diabetic mice that carry the hexokinase gene. Overexpression of hexokinase had no effect during aerobic culture, but during hypoxia, hexokinase improved ATP content by 44% and this restored contractility almost to normal levels. In myocytes from diabetic mice, tested under both aerobic and hypoxic conditions, the hexokinase gene significantly improved ATP content and this significantly improved contractility. These results demonstrate that elevating hexokinase activity can be beneficial to hypoxic or diabetic cardiomyocytes secondary to improving myocyte ATP levels.
Similar content being viewed by others
References
Manchester, J., Kong, X., Nerbonne, J., Lowry, O.H., and Lawrence, J.C. Jr. (1994). Glucose transport and phosphorylation in single cardiac myocytes: rate-limiting steps in glucose metabolism. Am. J. Physiol. 266:E326-E333.
Feldhaus, L.M. and Liedtke, A.J. (1998). mRNA expression of glycolytic enzymes and glucose transporter proteins in ischemic myocardium with and without reperfusion. J. Mol. Cell. Cardiol. 30:2475–2485.
McFalls, E.O., Murad, B., Liow, J.S., Gannon, M.C., Haspel, H.C., Lange, A., et al. (2002). Glucose uptake and glycogen levels are increased in pig heart after repetitive ischemia. Am. J. Physiol. Heart Circ. Physiol. 282:H205-H211.
Annane, D., Duboc, D., Mazoyer, B., Merlet, P., Fiorelli, M., Eymard, B., et al. (1994). Correlation between decreased myocardial glucose phosphorylation and the DNA mutation size in myotonic dystrophy. Circulation 90:2629–2634.
Stanley, W.C., Lopaschuk, G.D., and McCormack, J.G. (1997). Regulation of energy substrate metabolism in the diabetic heart. Cardiovasc. Res. 34:25–33.
Rodrigues, B. and McNeill, J.H. (1992). The diabetic heart: metabolic causes for the development of a cardiomyopathy. Cardiovasc. Res. 26: 913–922.
Rubler, S., Dlugash, J., Yuceoglu, Y.Z., Kumral, T., Branwood, A.W., and Grishman, A. (1972). New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am. J. Cardiol. 30:595–602.
Kannel, W.B., Hjortland, M., and Castelli, W.P. (1974). Role of diabetes in congestive heart failure: the Framingham study. Am. J. Cardiol. 34:29–34.
Belke, D.D., Larsen, T.S., Gibbs, E.M., and Severson, D.L. (2000). Altered metabolism causes cardiac dysfunction in perfused hearts from diabetic (db/db) mice. Am. J. Physiol. Endocrinol. Metab. 279:E1104-E1113.
Feuvray, D. and Lopaschuk, G.D. (1997). Controversies on the sensitivity of the diabetic heart to ischemic injury: the sensitivity of the diabetic heart to ischemic injury is decreased. Cardiovasc. Res. 34:113–120.
Liang, Q., Donthi, R.V., Kralik, P.M., and Epstein, P.N. (2002). Elevated hexokinase increases cardiac glycolysis in transgenic mice. Cardiovasc. Res. 53:423–430.
Epstein, P.N., Overbeek, P.A., and Means, A. P. (1989). Calmodulin-induced early-onset diabetes in transgenic mice. Cell 58:1067–1073.
Liang, Q., Carlson, E.C., Donthi, R.V., Kralik, P.M., Shen, X., and Epstein, P.N. (2002). Overexpression of metallothionein reduces diabetic cardiomyopathy. Diabetes 51:174–181.
Bedoya, F.J., Meglasson, M.D., Wilson, J.M., and Matschinsky, F.M. (1985). Radiometric oil well assay for glucokinase in microscopic structures. Anal. Biochem. 144:504–513.
Ye, G., Metreveli, N.S., Donthi, R.V., Xia, S., Xu, M., Carlson, E.C., et al. (2004). Catalase protects cardiomyocyte function in models of type 1 and type 2 diabetes. Diabetes 53:1336–1343.
Zheng, S., Noonan, W.T., Metreveli, N.S., Coventry, S., Kralik, P.M., Carlson, E.C., et al. (2004). Development of late-stage diabetic nephropathy in OVE26 diabetic mice. Diabetes 53:3248–3257.
Ye, G., Metreveli, N.S., Ren, J., and Epstein, P.N. (2003). Metallothionein prevents diabetes-induced deficits in cardiomyocytes by inhibiting reactive oxygen species production. Diabetes 52:777–783.
Steenbergen, C., Murphy, E., Watts, J.A., and London, R.E. (1990). Correlation between cytosolic free calcium, contracture, ATP and irreversible ischemic injury in perfused rat heart. Circ. Res. 66:135–146.
Meno, H., Kanaide, H., Okada, M., and Nakamura, M. (1984). Total adenine nucleotide stores and sarcoplasmic reticular Ca transport in ischemic rat heart. Am. J. Physiol. 247:H380-H386.
Ren, J. and Davidoff, A.J. (1997). Diabetes rapidly induces contractile dysfunctions in isolated ventricular myocytes. Am. J. Physiol. 272:H148-H158.
Ren, J., Sowers, J.R., Walsh, M.F., and Brown, R.A. (2000). Reduced contractile response to insulin and IGF-I in ventricular myocytes from genetically obese Zucker rats. Am. J. Physiol. Heart Circ. Physiol. 279:H1708-H1714.
Noda, C., Masuda, T., Sato, K., Ikeda, K., Shimohama, T., Matsuyama, N., et al. (2003). Vanadate improves cardiac function and myocardial energy metabolism in diabetic rat hearts. Jpn. Heart J. 44:745–757.
Rights and permissions
About this article
Cite this article
Ye, G., Donthi, R.V., Metreveli, N.S. et al. Overexpression of hexokinase protects hypoxic and diabetic cardiomyocytes by increasing ATP generation. Cardiovasc Toxicol 5, 293–300 (2005). https://doi.org/10.1385/CT:5:3:293
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/CT:5:3:293