Abstract
Background: Laboratory-based research with community samples has suggested changes in affective, behavioural and cognitive processes as possible explanations for the effects of serotonergic medications. Examining the effects of serotonergic medications using an ecological momentary measure (such as event-contingent recording) in the daily lives of people with social anxiety disorder would contribute to establishing the effects of these medications on affect, behaviour and one form of cognition: perception of others’ behaviour.
Methods: The present study assessed changes in affect, interpersonal behaviour and perception of others’ behaviour in adults with social anxiety disorder using ecological momentary assessment at baseline and over 4 months of a single-arm, uncontrolled, open-label trial of treatment with the selective serotonin reuptake inhibitor paroxetine.
Results: Anxiety and concurrent depressive symptoms decreased. Participants also reported increased positive and decreased negative affect; increased agreeable and decreased quarrelsome behaviour; increased dominant and decreased submissive behaviour; and increased perception that others behaved agreeably toward them. Moreover, participants demonstrated reduced intraindividual variability in affect, interpersonal behaviour and perception of others’ behaviour.
Limitations: Limitations included the lack of a placebo group, the inability to identify the temporal order of changes and the restricted assessment of extreme behaviour.
Conclusion: The results of the present study demonstrate changes during pharmacotherapy in the manifestation of affect, interpersonal behaviour and interpersonal perception in the daily lives of people with social anxiety disorder. Given the importance of interpersonal processes to social anxiety disorder, these results may guide future research seeking to clarify mechanisms of action for serotonergic medications.
Introduction
Social anxiety disorder (SAD), also known as social phobia, describes anxiety-related physiologic, cognitive and affective responses to social stimuli, such as interpersonal interactions. 1 Within the cognitive–behavioural formulation, fears about perceived or anticipated negative social outcomes drive physiologic and affective distress. This distress negatively reinforces interpersonal behaviours (such as submissive2,3 and quarrelsome behaviours4) that avoid the feared outcome by limiting social interaction.5 The resulting behaviours6 may be regarded negatively by others and result in negative social outcomes.7 Individuals with SAD report expecting negative evaluations from others, low expectations of their performance in social situations5 and a tendency to construe social situations as threatening,8,9 including perceiving less closeness in interpersonal relationships and perceiving themselves as inferior and of lower social status.10
Serotonergic medications (e.g., selective serotonin reuptake inhibitors [SSRIs])11 have demonstrated effectiveness in treating symptoms of SAD. As well, SSRIs may modulate characteristic affect, interpersonal behaviour and interpersonal perceptions among people with SAD. For example, enhanced serotonergic function through the administration of tryptophan, 12 SSRIs13 or serotonin and norepinephrine reuptake inhibitors (SNRIs)14,15 augments warm-agreeableness13–18 and dominance,13,15,19 decreases within-person variability20 and reduces aggression in nonhuman primates21 and humans.22 Additionally, administration of SSRIs and tryptophan to healthy and depressed individuals21,22 increases awareness of and attention to positive social cues, such as perceiving ambiguous stimuli as positive.
Affect, behaviour and perceptions related to SAD may contribute to its maintenance, and modification of these patterns may indicate a mechanism of action to explain the anxiety-reducing effect of serotonergic treatments. The Social Interaction Model proposed by Young and colleagues23 focuses on depression and suggests that serotonergic medication increases prosocial behaviour and reduces instability, thereby improving social interactions that reduce symptomatology.24 Among healthy volunteers, research has documented changes in interpersonal behaviour13–15,18,25 and participant-reported interpersonal problems26 during the administration of serotonergic medications. Harmer and colleagues27,28 summarized evidence supporting a cognitive neuropsychological model, in which serotonergic medication facilitates changes in attention and memory that reduce cognitive biases. Changes in attention and memory on laboratory-based facial emotion recognition tasks has been found among healthy volunteers29 and clinical samples.23 Empirical support for these proposals is indicated from research with healthy volunteers but is limited in clinical samples.
While both self-report and laboratory-based research have unique strengths, they also have limitations, including retrospective recall bias for self-reports completed long (e.g., weeks) after the event of interest and limited generalizability of laboratory tasks to the range of situations encountered in daily life.30 For example, Tse and Bond13,15 provided critical initial indications that, among healthy volunteers, SSRI or SNRI administration may increase agreeable–cooperative behaviour and decrease submissive behaviour in participants’ daily lives. This work relied on flatmate reports of participant behaviour, which improved ecological validity but relied on retrospection. Moreover, this kind of method focuses on how the person behaves in general, but is unable to examine temporal patterns such as intraindividual variability. To further examine whether changes in affect, cognition and interpersonal behaviour are potential mechanisms of action for serotonergic pharmacotherapy of SAD, investigation is needed with interventions using clinical samples.
Assessment of affect, interpersonal behaviour and perception in the daily lives of community and clinical samples has been refined over 30 years of research. Research on interpersonal behaviour and the perception of others has frequently conceptualized behaviour along the 2 dimensions (i.e., communal [agreeable–quarrelsome] and agentic [dominant–submissive]) of the interpersonal circumplex.31 Ecological momentary assessment (EMA) methods32 provide intensive repeated measures in naturalistic settings and are well-suited to examine changes in the naturalistic manifestation of affect, interpersonal behaviour and perception closely following interpersonal interactions during pharmacological treatment.33 The high temporal density of EMAs permits computing measures beyond mean level (e.g., intraindividual variability in affect, interpersonal behaviour and perception that indexes instability34 and confers risk for anxiety35,36 and interpersonal problems37,38). Because EMAs ask participants to self-report on current states (e.g., affect), reports are minimally retrospective and assess temporal dynamics outside of awareness. For example, one study demonstrated that tryptophan administration to a community sample decreased intraindividual variability of interpersonal behaviour for irritable individuals. 20 Testing whether SSRI administration reduces variability (i.e., instability) may clarify anxiety-reducing effects.
The present study
The present study sought to demonstrate change in mean level and intraindividual variability in affect, interpersonal behaviour and perception during serotonergic treatment of SAD. Given the importance of interpersonal situations for SAD, the present study assessed affect, interpersonal behaviour and perception during naturally occurring interpersonal situations using event-contingent recording (ECR), a specific form of EMA. Participants recorded their affect, interpersonal behaviour and perception of others’ behaviour following interpersonal interactions that lasted longer than 5 minutes.39
This study was conducted in Canada following the principles of the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans,40 which states that placebo-controlled clinical trials can be conducted when participants are randomly assigned to different groups, and when there is equipoise or genuine uncertainty on the part of the relevant expert community about which therapy is most effective for a given condition. Given that paroxetine is approved for the treatment of SAD, there was no equipoise between paroxetine and placebo. The use of a placebo group would not have conformed to the Tri-Council Policy Statement, so paroxetine was administered in an open-label design with no placebo or control condition.
This study provides a preliminary examination of changes in affect, interpersonal behaviour and perception during treatment with paroxetine, as manifested in the daily lives of people with SAD. Although further research is needed to examine affective, behavioural and cognitive processes as clinical outcomes of paroxetine treatment, the present study provides an initial examination of changes in both mean and intraindividual variability during paroxetine administration. Because paroxetine is frequently used to treat SAD in clinical practice,11 the present study indicates changes in affect, interpersonal behaviour and interpersonal perception that may occur during psychiatric practice.
Based on the literature in human and nonhuman primates, we hypothesized that during paroxetine treatment participants would demonstrate elevated positive and reduced negative affect; elevated agreeable and decreased quarrelsome behaviour; and elevated dominant and reduced submissive behaviour. Given evidence that SAD may be associated with a cognitive bias toward social threat8 that may decrease after SSRI administration,27,28 we hypothesized increased perception of warmth in others’ behaviour. We considered change in the perception of dominance in others’ behaviour to be exploratory. We further hypothesized that results would extend previous evidence20 of reduced intraindividual variability in affect, interpersonal behaviour and perception of others’ behaviour during paroxetine administration.
Methods
Participants
We recruited participants from a previous study of SAD3 and from community advertisements. After people had been screened for SAD symptoms by phone, they were invited to an initial meeting that included a diagnostic assessment. Inclusion criteria were the ability to understand study materials in English and a primary diagnosis of SAD. Exclusion criteria were a concurrent diagnosis of bipolar or psychotic disorder and a lack of suitability for paroxetine treatment due to hypersensitivity or current use of monoamine oxidase inhibitors.
Of 52 individuals who attended the initial session, 48 enrolled in the study, of whom 9 withdrew before beginning paroxetine administration.
Twenty-nine participants (60.4%) completed the baseline assessment and 4 months of treatment. Four (8.3%) participants discontinued the study after 2 months, 4 (8.3%) after 3 months and 2 (4.2%) after 4 months. Participants who withdrew indicated lower baseline anxiety on the Social Phobia Scale (SPS; t34.23 = −2.14, d = −0.73, p = 0.04) and Liebowitz Social Anxiety Scale (LSAS; t36.02 = −2.52, d = −0.84, p = 0.02). We reran analyses adjusting for baseline SPS and LSAS scores. Results were unchanged and are presented without adjustment. Withdrawal was not associated with baseline anxiety on the Social Interaction Anxiety Scale (SIAS), depression symptom severity, participant age, sex, language, level of education or employment status (all p > 0.10).
Measures
Psychiatric diagnosis and clinical assessment
We used the Mini-International Neuropsychiatric Interview (MINI, version 5)41 to establish diagnoses of SAD and comorbid disorders. We assessed the severity of SAD symptoms during treatment via clinician report on the LSAS42 and via participant report on the SPS and the SIAS.43 The SIAS includes 2 reverse-coded items. We conducted analyses with both items removed. We obtained identical results when we reran analyses with both items included (see Appendix 1, available at jpn.ca/170141-a1). We assessed the severity of depressive symptoms via clinician report on the Montgomery–Åsberg Depression Rating Scale,44 which correlates highly with participant report. Inter-item reliability of all symptom assessments was high at each time point (α = 0.69–0.96).
Interpersonal behaviour
We assessed participant interpersonal behaviour as communal (i.e., agreeable and quarrelsome) and agentic (i.e., dominant and submissive) behaviour from the interpersonal circumplex model.31 Participants indicated their behaviour at each event by endorsing behaviours representing those dimensions on the Social Behaviour Inventory (SBI).45 Items were summed and ipsatized at each event to centre reports within the event and individual.45 The SBI has demonstrated moderate to high inter-item reliability, strong convergent validity, high test–retest reliability39 and suitability for use in clinical samples.3 We derived intraindividual variability for each behaviour or dispersion in type of interpersonal behaviour reported (i.e., spin) from each participant’s event-level reports.46
Affect
Participants recorded event-level positive and negative affect on 9 items balanced for affective arousal.47 Items were averaged at each event to compute event-level positive and negative affect. Inter-item reliability across the 5 waves of data collection was high for negative (α = 0.89–0.92) and positive affect (α = 0.95–0.97).
Perception of others’ behaviour
At each event, participants reported their perception of the behaviour of the person or people they interacted with48 by placing a mark on a 9 × 9 grid derived from the 2 dimensions of the interpersonal circumplex model (i.e., communal and agentic behaviour).31 Inter-rater reliability, test–retest reliability and convergent validity have been demonstrated previously.48
Design
This study followed an open-label, single-arm, nonrandomized, uncontrolled design. We assessed symptom severity, affect, interpersonal behaviour and perception of others’ behaviour using ECR in a measurement burst design. Over the 5-month study, participants received paroxetine controlled-release (Paxil CR) titrated from 12.5 mg/d to 25 mg/d to minimize potential side effects. Participants met with the prescribing psychiatrist at 4-week intervals (i.e., at 4, 8, 12 and 16 weeks from the initiation of treatment) to evaluate adverse effects and complete symptom assessments. Participants completed 1 week of the ECR procedure after each clinical meeting; by the end of the study, participants had completed 35 days of the ECR procedure over 5 periods of 1 week.
This report describes all measures and conditions. As described previously, data were excluded if missing; participants were excluded based on inclusion and exclusion criteria. Financial constraints dictated sample-size determination.
Procedure
Following telephone screens, participants attended an information session. During this baseline session, participants provided informed consent and participated in an in-person assessment conducted by study psychiatrists to establish psychiatric diagnosis and suitability for paroxetine treatment. Subsequently, participants began a baseline 1-week period of the ECR procedure (see below). For 2 participants drawn from a previously collected sample,3 the final week of a previous 20-day ECR procedure provided the baseline.
Event-contingent recording
During each ECR period, participants were asked to complete a form after each substantial interaction, defined as an interaction lasting longer than 5 minutes.39 Participants used the forms to report on their affect, their interpersonal behaviour and the behaviour of the person with whom they interacted. To reduce participant burden, a subset of behaviour items were included on each form. To reduce the potential for participants to develop a response set over events, SBI items were rotated over days. There were 4 forms, each with 3 items for each behaviour dimension.
Ethics
This study was approved by the McGill University Research Ethics Board. All procedures contributing to this work complied with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Statistical analysis
We fit a mixed-effects (i.e., multilevel) growth curve model to each measure of syndrome severity to evaluate change in the severity of anxiety and depressive symptoms over the 5 months of the study.
We fit location–scale models to model change in the mean (i.e., location) and variance (i.e., scale) of interpersonal behaviour, affect and perception of others’ behaviour over time, while adjusting for correlation between the mean and variance.49 We used a 3-level mixed-effects location–scale model, adapted from Hedeker and Mermelstein,50 to account for nesting of events within measurement wave (i.e., month), which were nested within person (see Appendix 1). Variance was log-transformed. Tables report original estimates; the results section provides estimates exponentiated to compute a variance ratio (VR) representing the average proportion of change in variance each month. We assessed change in spin, an aggregate index of intraindividual variability in interpersonal behaviour,46 using a mixed-effects growth curve model. We then added monthly anxiety symptoms as main effects for both the mean and variance. Because of multicollinearity, we ran each model separately with each index of anxiety symptomatology. Anxiety assessments were centred within-person. 50 We entered the person-level and centred scores as predictors, thereby disaggregating within- and between-person associations.
We conducted all analyses using SAS software version 9.451 and maximum likelihood estimation with the NLMIXED procedure. We imported estimated fixed effects into R version 3.2.352 and plotted them using the ggplot2 package.53
Results
Participants
Of the 39 participants who began paroxetine administration, 19 (48.7%) were female. Participant ages ranged from 20 to 60 years (mean ± standard deviation 33.92 ± 11.49).
Psychiatric comorbidity was low but included concurrent dysthymic disorder (n = 1), recurrent major depressive disorder (n = 1), panic disorder (n = 1), alcohol abuse (n = 1) and substance abuse (n= 1). At baseline, all participants scored above a recommended clinical cutoff on the LSAS.54 Mean baseline anxiety severity (SPS and SIAS) was high relative to extant research on SAD.55 Baseline assessment of depressive symptoms (Montgomery–Åsberg Depression Rating Scale) indicated mild depressive symptoms (score > 10)56 in 10 participants (27%).
The sample included 21 participants (53.8%) whose first language was English. Twelve participants (30.8%) reported employment at least 30 hours per week, 24 (61.5%) were employed less than 30 hours per week and 3 (7.7%) did not provide employment information. Sixteen participants (41.0%) had a bachelor’s degree, 15 (38.5%) had some college education, 5 (12.9%) had high school education or less and 3 (7.7%) had a postgraduate degree. The distribution of educational makeup was similar to that of Montréal, Que., Canada, where the study was conducted.57
Change in symptoms
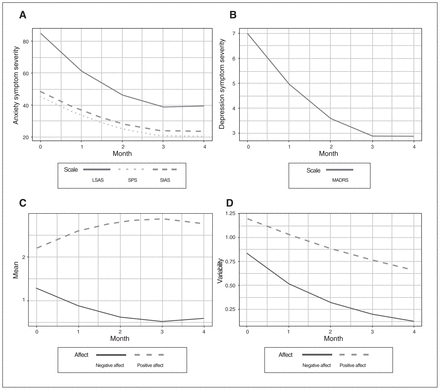
As expected, following initiation of paroxetine treatment, clinician-rated (i.e., LSAS) and participant-rated (i.e., SPS and SIAS) anxiety (Fig. 1A) and depressive symptoms (Fig. 1B) decreased. There was a monotonic downward pattern with deceleration over time (Table 1 and Appendix 1, Table S1, for random effects).
Changes in symptom severity (A, B) and event-level affect mean and variability (C, D). LSAS = Liebowitz Social Anxiety Scale; MADRS = Montgomery–Åsberg Depression Rating Scale; SIAS = Social Interaction and Anxiety Scale; SPS = Social Phobia Scale.
Change in anxiety and depression symptom severity over treatment
Change in affect
As hypothesized, over the course of treatment, participants reported increased positive and decreased negative affect (Table 2, Fig. 1C; see Appendix 1, Table S2, for random effects). Similar to the pattern of change in symptom severity, improvements in mean affect (e.g., decreased negative affect) decelerated over time. As hypothesized, intraindividual variability (i.e., variance, scale) in affect decreased over time for both positive and negative affect by 14% (VR = 0.86) and 37% (VR = 0.63) per month, respectively (Fig. 1D).
Change in affect, perception and interpersonal behaviour over treatment, mean (location) and variance (scale)
Change in perception of others’ interpersonal behaviour
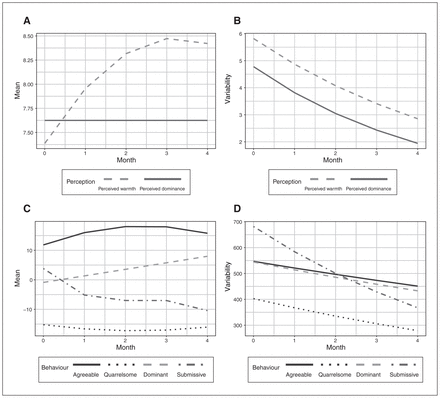
As hypothesized, participants’ tendency to perceive others as behaving agreeably increased. There was a decelerating rate of change over time (Table 2, Fig. 2A; see Appendix 1, Table S2, for random effects). Participants’ tendency to perceive others as behaving dominantly did not change over time. As hypothesized, intraindividual variability (i.e., variance, scale) in participants’ perceptions of warmth decreased over time by 16% (VR = 0.84) per month (Fig. 2B). Intraindividual variability in the perception of others’ dominant behaviour decreased over time by 21% (VR = 0.79) per month.
Changes in mean and variability of participants’ perception of others’ behaviour (A, B) and participants’ own interpersonal behaviour (C, D).
Change in participants’ interpersonal behaviour
As hypothesized, over time participants reported increased communal behaviour; agreeable behaviour increased while quarrelsome behaviour decreased (Table 2, Fig. 2C; see Appendix 1, Table S3, for random effects). In addition, agentic behaviour increased as reflected in increased dominant and decreased submissive behaviour. Improvements in agreeable, quarrelsome and submissive behaviour decelerated over time, though dominant behaviour increased at a steady rate. As hypothesized, variability in participant reports of interpersonal behaviour across interactions decreased for agreeable, quarrelsome, dominant and submissive behaviour by 5% (VR = 0.95), 9% (VR = 0.91), 6% (VR = 0.94) and 14% (VR = 0.86) per month, respectively (Fig. 2D). Spin, which reflects dispersion in interpersonal behaviour,46 also decreased in a linear trajectory over time (B = −0.10, p < 0.001, 95% confidence interval −0.15 to −0.06), even after adjusting for concurrent mean communal and agentic behaviour.
Change in the mean-squared successive difference
Additional mixed-effects growth curve models were fit to estimate change in intraindividual variability in affect, perception of others’ interpersonal behaviour and participants’ interpersonal behaviour when computed as the mean-squared successive difference. Results were consistent with the location–scale approach for positive and negative affect, perception of others’ behaviour as dominant and warm, and participants’ agreeable and submissive behaviour.
Discussion
During 4-month treatment with paroxetine, people with SAD reported improved mood (i.e., increased positive and decreased negative affect), greater affiliative/communal behaviour (i.e., increased agreeable and decreased quarrelsome behaviour) and increased agentic behaviour (i.e., increased dominant and reduced submissive behaviour). Participants perceived others’ behaviour as increasingly warm. They also demonstrated decreased intraindividual variability in affect, interpersonal behaviour and perception of others’ behaviour. Anxiety and depressive symptomatology decreased as measured by both participant and clinician report.
Because it lacked a placebo control, the present study provides only preliminary evidence that serotonin modulation influences affect, interpersonal behaviour and interpersonal perception. However, results are consistent with previous findings. For example, evidence of decreased quarrelsome behaviour during paroxetine administration extends evidence among nonclinical community participants16,25 that administration of tryptophan, a serotonin precursor,12 decreased quarrelsome behaviour in daily life compared with placebo. Similarly, evidence of increased agreeable behaviour is consistent with previous research in nonhuman primates17 and healthy human participants during a laboratory cooperation task13–15,18 and naturalistic assessment.15,16 The present findings further implicate serotonin in the modulation of agentic behaviour. This is consistent with increased dominant16,25 and reduced submissive behaviour13,15 following tryptophan and SSRI/SNRI administration to healthy human participants, respectively.
Beyond mean behavioural patterns, intensive repeated-measures methodologies such as ECR permit assessment of intraindividual variability in participant-reported affect, behaviour and perceptions over the range of situations experienced in daily life.46 Elevated variability, which has been associated with general anxiety34–36,58 and SAD,59 may be associated with interpersonal difficulties.37,38 Prior research demonstrated reduced intraindividual variability in interpersonal behaviour following tryptophan administration compared with placebo among healthy participants with elevated irritability.20 The present study further suggests reduced intraindividual variability in affect, behaviour and perception during paroxetine administration.
The changes reported during paroxetine treatment in the present study parallel affective, behavioural and cognitive deficits characteristics of SAD. Reduced submissive behaviour2,3 and increased agreeable behaviour4 may decrease interpersonal stress in close relationships.7 Increases in the perception of warmth in others’ behaviour are consistent with construing social situations as less threatening,8 and expecting less negative evaluation from others could contribute specifically to reduced anxiety and more generally to reduced negative affect.5
Change in interpersonal behaviour and perceptions of others’ behaviour is consistent with 2 recent proposals for behavioural and cognitive mechanisms of serotonergic pharmacotherapy. Increased communal and agentic behaviour is consistent with the Social Interaction Model, which suggests that symptom improvement may result from improved interpersonal behaviour and, subsequently, reduced negative interpersonal consequences.24 Evidence of increased perception of warmth in others’ behaviour, along with improved affect, supports the Cognitive Neuropsychological Model, in which serotonergic medication impacts on mood and symptomatology by modifying cognitions, such as the tendency to perceive others as acting in a quarrelsome manner.27,28
Previous support for both theories has been limited to research in healthy community participants or laboratory-based research. While the present findings do not support one model more strongly than the other, they provide the first evidence to demonstrate changes in naturally occurring patterns of affect, interpersonal behaviour and perception during SSRI treatment of individuals diagnosed with SAD. Future research is needed to rule out potential concomitants, such as the influence of reflecting on one’s interactions during ECR data collection. Nonetheless, the present study provides an important initial step to demonstrate consistent changes in mean affect, interpersonal behaviour and perception of others’ behaviour, as well as reduction in intraindividual variability.
Limitations
Several limitations warrant consideration and further research. The present study provides a preliminary investigation of change in affect, interpersonal behaviour and interpersonal perception without a comparison placebo condition. While the present study suggests changes during standard pharmacological treatment of individuals with SAD, future research is needed to rule out alternative explanations, such as the possibility that change is due to reflection on one’s interactions. However, among community adults, previous research has demonstrated moderate to high stability over consecutive weeks of the ECR measures used in the present study, which suggests that the procedure may not substantially influence participants’ reported affect and behaviour.39,46
During paroxetine treatment, participants reported that others behaved more agreeably. This finding may indicate changes in participants’ perception of others’ behaviour or it may signify actual changes in others’ behaviour. Additionally, previous work has implicated affect and others’ behaviour in modulating participants’ behaviour.58,60 Future use of the ECR procedure and related EMA methodology is needed to examine the temporal patterning of changes in affect, interpersonal behaviour and perception of others’ behaviour.
The SBI and Interpersonal Grid assess the range of interpersonal behaviours manifested in typical interpersonal interactions among community participants.45,48 While these measures inform research with clinical samples, they may not include clinically relevant behaviours. For example, evidence implicating serotonergic dysregulation in aggression suggests future research on changes in aggression beyond quarrelsome behaviour.
Conclusion
In the present study, individuals with SAD demonstrated increased communal (more agreeable, less quarrelsome) and increased agentic (more dominant, less submissive) behaviours, improved affect and greater perception of warmth in others’ behaviour during SSRI pharmacotherapy for SAD. Moreover, participants demonstrated decreased intraindividual variability across all 8 measures of behaviour, affect and perception, which suggests reduced instability.
Although further research is needed, the present findings provide preliminary evidence of reductions in characteristic affective, behavioural and perceptual patterns among individuals with SAD during pharmacotherapy with paroxetine. Findings also encourage the use of repeated assessment of patients’ experiences in daily life. This methodological approach enhanced generalizability through naturalistic assessment across a range of interpersonal encounters. High temporal density recording permitted not only the assessment of mean levels, but also intraindividual variability in patient characteristics, a novel approach to the representation of patients’ features. As piloted in the present study, the assessment of symptoms and clinical outcomes in real time may inform the development of personalized treatments and provide novel indices of treatment response, such as reduced affective, behavioural or cognitive variability.
Acknowledgments
The authors thank Carolina Pansera, Melissa Levesque and David Paul for assistance in data collection, Helen Wang for assistance with computational resources and Daniel S. Pine for feedback on an earlier manuscript draft.
Footnotes
Funding: This research was funded by a grant from GlaxoSmithKline to D.S. Moskowitz and from fellowship support to L. Rappaport from the National Institute of Mental Health (T32MH020030).
Competing interests: The authors do not have any financial interests that might influence this research. Some data from the present study were previously published in the doctoral dissertation by L. Rappaport; the data were analyzed differently to address different scientific aims. P. Bleau reports a grant from GlaxoSmithKline and other funding from the National Institute of Mental Health during the conduct of the study.
Contributors: J. Russell, P. Bleau and D. Moskowitz designed the study. J. Russell, G. Pinard, P. Bleau and D. Moskowitz acquired the data, which L. Rappaport, D. Hedeker, P. Bleau and D. Moskowitz analyzed. L. Rappaport and D. Moskowitz wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received July 24, 2017.
- Revision received October 31, 2017.
- Accepted January 18, 2018.