Abstract
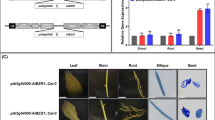
Improving oil yield of Indian mustard (Brassica juncea) is exigent as it is a major oilseed crop of Indian subcontinent, which has severe shortage of vegetable oil production in the world. Some of the regulators of seed development have been shown to improve oil yield in Arabidopsis. Arabidopsis KLUH (AtKLUH), a maternal regulator of seed size, has been shown to control seed oil content. In this study, we identified three homologs of AtKLUH in B. juncea, BjKLUH1, BjKLUH2-1 and BjKLUH2-2. We observed that BjKLUH1 differentially expresses in developing seeds in B. juncea accessions with varying seed size and oil content. Further, analyses for seed oil content in B. juncea transgenics carrying AtKLUH demonstrated an increase in seed oil up to 8.3% compared to wild-type plants. The results of this study suggest that KLUH may have a role in seed development and is a good candidate for engineering seed oil accumulation in B. juncea.
Similar content being viewed by others
Abbreviations
- ARF2:
-
auxin response factor2
- bp:
-
base pairs
- CDS:
-
coding sequence
- DAP:
-
days after pollination
- FA:
-
fatty acid
- GAPDH:
-
glyceraldehyde-3-phosphate dehydrogenase
- nptII:
-
neomycin phosphotransferase II
- ORF:
-
open reading frame
- PCR:
-
polymerase chain reaction
- qRT-PCR:
-
quantitative reverse-transcription PCR
- QTL:
-
quantitative trait locus
- TAG:
-
triacylglycerol
References
Adamski N.M., Anastasiou E., Eriksson S., O’Neill C.M. & Lenhard M. 2009. Local maternal control of seed size by KLUH/CYP78A5-dependent growth signaling. Proc. Natl. Acad. Sci. USA 106: 20115–20120.
Altschul S.F., Gish W., Miller W., Myers E.W. & Lipman D.J. 1990. Basic local alignment search tool. J. Mol. Biol. 215: 403–410.
An D. & Suh M.C. 2015. Overexpression of Arabidopsis WRI1 enhanced seed mass and storage oil content in Camelina sativa. Plant Biotechnol. Rep. 9: 137–148.
Anastasiou E., Kenz S., Gerstung M., MacLean D., Timmer J., Fleck C. & Lenhard M. 2007. Control of plant organ size by KLUH/CYP78A5-dependent intercellular signaling. Dev. Cell 13: 843–856.
Anisimova M. & Gascuel O. 2006. Approximate likelihood ratio test for branches: a fast accurate and powerful alternative. Syst. Biol. 55: 539–552.
Bak S., Beisson F., Bishop G., Hamberger B., Höfer R., Paquette S. & Werck-Reichhart D. 2011. Cytochromes P450. Arabidopsis Book 9: e0144.
Baud S., Boutin J.P., Miquel M., Lepiniec L. & Rochat C. 2002. An integrated overview of seed development in Arabidopsis thaliana ecotype WS. Plant Physiol. Biochem. 40: 151–160.
Bishnoi K.C. & Singh K. 1979. Effect of sowing dates varieties and nitrogen levels on yield and yield attributes of raya. Indian J. Agron. 24: 123–129.
Castresana J. 2000. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 17: 540–552.
Cernac A., Andre C., Hoffmann-Benning S. & Benning C. 2006. WRI1 is required for seed germination and seedling establishment. Plant Physiol. 141: 745–757.
Cernac A. & Benning C. 2004. WRINKLED1 encodes an AP2/EREB domain protein involved in the control of storage compound biosynthesis in Arabidopsis. Plant J. 40: 575–585.
Chung J., Babka H.L., Graef G.L., Staswick P.E., Lee D.J. & Cregan P.B. 2003. The seed protein oil and yield QTL on soybean linkage group I. Crop Sci. 43: 1053–1067.
Dereeper A., Guignon V., Blanc G., Audic S., Buffet S., Chevenet F., Dufayard J.F., Guindon S., Lefort V., Lescot M., Claverie J.M. & Gascuel O. 2008. Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 36: W465–W469.
Diepenbrock W. 2000. Yield analysis of winter oilseed rape (Brassica napus L.): a review. Field Crops Res. 67: 35–49.
Edgar R.C. 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32: 1792–1797.
Fatihi A., Zbierzak A.M. & Dormann P. 2013. Alterations in seed development gene expression affect size and oil content of Arabidopsis seeds. Plant Physiol. 163: 973–985.
Garcia D., Fitz Gerald J.N. & Berger F. 2005. Maternal control of integument cell elongation and zygotic control of endosperm growth are coordinated to determine seed size in Arabidopsis. Plant Cell 17: 52–60.
Guindon S., Dufayard J.F., Lefort V., Anisimova M., Hordijk W. & Gascuel O. 2010. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst. Biol. 59: 307–321.
Jako C., Kumar A., Wei Y., Zou J., Barton D.L., Giblin E.M. & Taylor D.C. 2001. Seed-specific over-expression of an Arabidopsis cDNA encoding a diacylglycerol acyltransferase enhances seed oil content and seed weight. Plant Physiol. 126: 861–874.
Jofuku K.D., Omidyar P.K., Gee Z. & Okamuro J.K. 2005. Control of seed mass and seed yield by the floral homeotic gene APETALA2. Proc. Natl. Acad. Sci. USA. 102: 3117–3122.
Jönsson R. 1978. Breeding for improved oil and meal quality in rape (Brassica napus L.) and turnip rape (Brassica campestris L.) Hereditas 87: 205–218.
Katepa-Mupondwa F., Raney J.P. & Rakow G. 2005. Recurrent selection for increased protein content in yellow mustard (Sinapis alba L.). Plant Breed. 124: 382–387.
Lewers K.S., StMartin S.K., Hedges B.R. & Palmer R.G. 1998. Testcross evaluation of soybean germplasm. Crop Sci. 38: 1143–1149.
Ma M., Zhao H., Li Z., Hu S., Song W. & Liu X. 2015. TaCYP78A5 regulates seed size in wheat (Triticum aestivum). J. Exp. Bot. 67: 1397–1410.
Martínez-Andújar C., Martin R.C. & Nonogaki H. 2012. Seed traits and genes important for translational biology: highlights from recent discoveries. Plant Cell Physiol. 53: 5–15.
Murray H.G. & Thompson W.F. 1980. Rapid isolation of high molecular weight DNA. Nucleic Acids Res. 8: 4321–4325.
Nagasawa N., Hibara K., Heppard E.P., Vander Velden K.A., Luck S., Beatty M., Nagato Y. & Sakai H. 2013. GIANT EMBRYO encodes CYP78A13 required for proper size balance between embryo and endosperm in rice. Plant J. 75: 592–605.
Okushima Y., Mitina I., Quach H.L. & Theologis A. 2005. AUXIN RESPONSE FACTOR2 (ARF2): a pleiotropic developmental regulator. Plant J. 43: 29–46.
Olsson G. 1960. Some relations between number of seeds per pod seed size and oil content and the effects of selection for these characters in Brassica and Sinapis. Hereditas 46: 29–70.
Qian W., Sass O., Meng J., Li M., Frauen M. & Jung C. 2007. Het-erotic patterns in rapeseed (B. napus L.): I. Crosses between spring and Chinese semi-winter lines. Theor. Appl. Genet. 115: 27–34.
Radchuk V. & Borisjuk L. 2014. Physical metabolic and developmental functions of the seed coat. Front. Plant Sci. 5: 510.
Ramanathan T. 2004. Applied genetics of oilseed crops. Daya Publishing House, ISBN: 978-8170353201, 448 pp.
Ramchiary N., Padmaja K.L., Sharma S., Gupta V., Sodhi Y.S., Mukhopadhyay A., Arumugam N., Pental D. & Pradhan A.K. 2007. Mapping of yield influencing QTL in Bjuncea: implications for breeding of major oilseed crop of dry land areas. Theor. Appl. Genet. 115: 807–817.
Roesler K., Shen B., Bermudez E., Li C., Hunt J., Damude H.G., Ripp K.G., Everard J.D., Booth J.R., Castaneda L., Feng L. & Meyer K. 2016. An improved variant of soybean type 1 diacylglycerol acyltransferase increases the oil content and decreases the soluble carbohydrate content of soybeans. Plant Physiol. 171: 878–893.
Saran G. & De R. 1979. Influence of seeding dates varieties and rates and methods of nitrogen application on the seed yield and quality of rapeseed grown on rainfed land. Indian J. Agricult. Sci. 49: 197–201.
Savadi S., Lambani N., Kashyap P.L. & Bisht D.S. 2016a. Genetic engineering approaches to enhance oil content in oilseed crops. Plant Growth Regul. doi:10.1007/s10725-016-0236-1.
Savadi S., Naresh V., Kumar V. & Bhat S.R. 2016b. Seed-specific overexpression of Arabidopsis DGAT1 in Indian mustard Brassica juncea increases seed oil content and seed weight. Botany 94: 177–184.
Savadi S., Naresh V., Kumar V., Dargan S., Gupta N.C., Chamola R. & Bhat S.R. 2015. Effect of overexpression of Arabidopsis thaliana SHB1 and KLUH genes on seed weight and yield contributing traits in Indian mustard (Brassica juncea L Cz-ern). Indian J. Genet. Plant Breed. 75: 349–356.
Shen B., Sinkevicius K.W., Selinger D.A. & Tarczynski M.C. 2006. The homeobox gene GLABRA2 affects seed oil content in Arabidopsis. Plant Mol. Biol. 60: 377–387.
Shi L., Katavic V., Yu Y., Kunst L. & Haughn G. 2012. Arabidop-sis glabra2 mutant seeds deficient in mucilage biosynthesis produces more oil. Plant J. 69: 37–46.
Sun R., Ye R., Gao L., Zhang L., Wang R., Mao T., Zheng Y., Li D. & Lin Y. 2017. Ectopic expression of CoWRI1 from coconut (Cocos nucifera L.) endosperm changes the seeds oil content in transgenic Arabidopsis and rice (Oryza sativa L.). Front. Plant Sci. 8: 63.
Taylor D.C., Katavic V., Zou J., MacKenzie S.L., Keller W.A., An J. & Ge Y. 2002. Field testing of transgenic rapeseed cv. Hero transformed with a yeast sn-2 acyltransferase results in increased oil content erucic acid content and seed yield. Mol. Breed. 8: 317–322.
Tian Y., Zhang M., Hu X., Wang L., Dai J., Xu Y. & Chen F. 2016. Overexpression of CYP78A98 a cytochrome P450 gene from Jatropha curcas L increases seed size of transgenic tobacco. Electronic J. Biotechnol. 19: 15–22.
Vigeolas H., Huhn D. & Geigenberger P. 2011. Nonsymbiotic hemoglobin-2 leads to an elevated energy state and to a combined increase in polyunsaturated fatty acids and total oil content when overexpressed in developing seeds of transgenic Arabidopsis plants. Plant Physiol. 155: 1435–1444.
Vigeolas H., Möhlmann T., Martini N., Neuhaus H.E. & Geigenberger P. 2004. Embryo-specific reduction of ADP-Glc py-rophosphorylase leads to an inhibition of starch synthesis and a delay in oil accumulation in developing seeds of oilseed rape. Plant Physiol. 136: 2676–2686.
Wang H.Z., Lui G.H., Wang X.F., Liu J., Yang Q. & Hua W. 2009. Heterosis and breeding of high oil content in rapeseed (Brassica napus L.). 16th Australian Research Assembly on Brassicas Australian Oilseeds Federation, Ballarat, Victoria.
Wilcox J.R. 1998. Increasing seed protein in soybean with eight cycles of recurrent selection. Crop Sci. 38: 1536–1540.
Yadava D.K. Vasudev S., Singh N., Mohapatra T. & Prabhu K.V. 2012. Breeding major oil crops: present status and future research needs, pp. 17–51. In: Gupta S. (ed.) Technological Innovations in Major World Oil Crops, Vol. 1. Springer, New York.
Zhao B., Dai A., Wei H., Yang S., Wang B., Jiang N. & Feng X. 2016. Arabidopsis KLU homologue GmCYP78A72 regulates seed size in soybean. Plant Mol. Biol. 90: 33–47.
Zhou Y., Zhang X., Kang X., Zhao X., Zhang X. & Ni M. 2009. SHORT HYPOCOTYL UNDER BLUE1 associates with MINISEED3 and HAIKU2 promoters in vivo to regulate Arabidopsis seed development. Plant Cell 21: 106–117.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
11756_2017_72091023_MOESM1_ESM.pdf
The online version of this article (DOI: https://doi.org/10.1515/biolog-2017-0123) contains supplementary material, which is available to authorized users.
Rights and permissions
About this article
Cite this article
Savadi, S., Bisht, D.S. & Bhat, S.R. Isolation and expression analyses of KLUH gene in developing seeds and enhanced seed oil in KLUH overexpressing Brassica juncea transgenics. Biologia 72, 1023–1030 (2017). https://doi.org/10.1515/biolog-2017-0123
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2017-0123