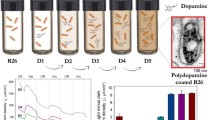
Abstract
Photovoltaic organic semiconductors are emerging in many therapeutic applications, including drug delivery and optical control of cell function. However, for their safe use the possible concomitant elicitation of undesired responses in target cells need to be carefully evaluated. Here we describe molecular responses activated by semiconducting polymer nanoparticles based on poly(3-hexyl)thiophene (P3HT) in the model Hydra vulgaris, previously shown to respond to P3HT-NP photostimulation and showed a decrease in the total antioxidant capacity and an increase in the DNA and protein oxidation levels, paving the way to a novel use of photovoltaic devices to control intracellular redox equilibrium.




Similar content being viewed by others
References
N.A. Repina, A. Rosenbloom, A. Mukherjee, D.V. Schaffer, and R.S. Kane: At light speed: advances in optogenetic systems for regulating cell signaling and behavior. Annu. Rev. Chem. Biomol. Eng. 8, 13 (2017).
T. Kushibiki, S. Okawa, T. Hirasawa, and M. Ishihara: Optogenetics: novel tools for controlling mammalian cell functions with light. Int. J. Photoenergy 2014, 10 (2014).
B. Tandon, A. Magaz, R. Balint, J.J. Blaker, and S.H. Cartmell: Electroactive biomaterials: vehicles for controlled delivery of therapeutic agents for drug delivery and tissue regeneration. Adv. Drug Deliv. Rev. (in press). doi: 10.1016/j.addr.2017.12.012.
M.R. Antognazza, N. Martino, D. Ghezzi, P. Feyen, E. Colombo, D. Endeman, F. Benfenati, and G. Lanzani: Shedding light on living cells. Adv. Mater. 27, 7662 (2015).
M. Zangoli, F. Di Maria, E. Zucchetti, C. Bossio, M.R. Antognazza, G. Lanzani, R. Mazzaro, F. Corticelli, M. Baroncini, and G. Barbarella: Engineering thiophene-based nanoparticles to induce phototransduction in live cells under illumination. Nanoscale 9, 9202 (2017).
L. Lu, T. Zheng, Q. Wu, A.M. Schneider, D. Zhao, and L. Yu: Recent advances in bulk heterojunction polymer solar cells. Chem. Rev. 115, 12666 (2015).
D. Ghezzi, M.R. Antognazza, M. Dal Maschio, E. Lanzarini, F. Benfenati, and G. Lanzani: A hybrid bioorganic interface for neuronal photoactivation. Nat. Commun. 2, 166 (2011).
V. Benfenati, N. Martino, M.R. Antognazza, A. Pistone, S. Toffanin, S. Ferroni, G. Lanzani, and M. Muccini: Photostimulation of whole-cell conductance in primary rat neocortical astrocytes mediated by organic semiconducting thin films. Adv. Healthcare Mater. 3, 392 (2014).
N. Martino, P. Feyen, M. Porro, C. Bossio, E. Zucchetti, D. Ghezzi, F. Benfenati, G. Lanzani, and M.R. Antognazza: Photothermal cellular stimulation in functional bio-polymer interfaces. Sci. Rep. 5, 8911 (2015).
P. Feyen, E. Colombo, D. Endeman, M. Nova, L. Laudato, N. Martino, M.R. Antognazza, G. Lanzani, F. Benfenati, and D. Ghezzi: Light-evoked hyperpolarization and silencing of neurons by conjugated polymers. Sci. Rep. 6, 22718 (2016).
F. Lodola, N. Martino, G. Tullii, G. Lanzani, and M.R. Antognazza: Conjugated polymers mediate effective activation of the mammalian ion channel transient receptor potential vanilloid 1. Sci. Rep. 7, 8477 (2017).
D. Ghezzi, M.R. Antognazza, R. Maccarone, S. Bellani, E. Lanzarini, N. Martino, M. Mete, G. Pertile, S. Bisti, G. Lanzani, and F. Benfenati: A polymer optoelectronic interface restores light sensitivity in blind rat retinas. Nat. Photonics 7, 400 (2013).
J.F. Maya-Vetencourt, D. Ghezzi, M.R. Antognazza, E. Colombo, M. Mete, P. Feyen, A. Desii, A. Buschiazzo, M. Di Paolo, S. Di Marco, F. Ticconi, L. Emionite, D. Shmal, C. Marini, I. Donelli, G. Freddi, R. Maccarone, S. Bisti, G. Sambuceti, G. Pertile, G. Lanzani, and F. Benfenati: A fully organic retinal prosthesis restores vision in a rat model of degenerative blindness. Nat. Mater. 16, 681 (2017).
A. Ambrosone, M. Roopin, B. Pelaz, A.M. Abdelmonem, L.M. Ackermann, L. Mattera, M. Allocca, A. Tino, M. Klapper, W.J. Parak, O. Levy, and C. Tortiglione: Dissecting common and divergent molecular pathways elicited by CdSe/ZnS quantum dots in freshwater and marine sentinel invertebrates. Nanotoxicology 11, 289 (2017).
M. Moros, A. Ambrosone, G. Stepien, F. Fabozzi, V. Marchesano, A. Castaldi, A. Tino, J.M. de la Fuente, and C. Tortiglione: Deciphering intracellular events triggered by mild magnetic hyperthermia in vitro and in vivo. Nanomedicine 10, 2167 (2015).
Tortiglione: An ancient model organism to test in vivo novel functional nanocrystals in Biomedical Engineering: from theory to application edited by R. Fazel-Rezai (InTech—Open Access Publisher 2011), pp. 225.
C. Tortiglione, M.R. Antognazza, A. Tino, C. Bossio, V. Marchesano, A. Bauduin, M. Zangoli, S.V. Morata, and G. Lanzani: Semiconducting polymers are light nanotransducers in eyeless animals. Sci. Adv. 3, e1601699 (2017).
B. Wang, H. Yuan, C. Zhu, Q. Yang, F. Lv, L. Liu, and S. Wang: Polymer-drug conjugates for intracellar molecule-targeted photoinduced inactivation of protein and growth inhibition of cancer cells. Sci. Rep. 2, 766 (2012).
R.M. Giron, J. Marco-Martinez, S. Bellani, A. Insuasty, H.C. Rojas, G. Tullii, M.R. Antognazza, S. Filippone, and N. Martin: Synthesis of modified fullerenes for oxygen reduction reactions. J. Mater. Chem. A 4, 14284 (2016).
G. Tullii, A. Desii, C. Bossio, S. Bellani, M. Colombo, N. Martino, M.R. Antognazza, and G. Lanzani: Bimodal functioning of a mesoporous, light sensitive polymer/electrolyte interface. Org. Electron. 46, 88 (2017).
I. Dalle-Donne, D. Giustarini, R. Colombo, R. Rossi, and A. Milzani: Protein carbonylation in human diseases. Trends Mol. Med. 9, 169 (2003).
K. Kannan and S.K. Jain: Oxidative stress and apoptosis. Pathophysiology 7, 153 (2000).
J. Pecher and S. Mecking: Nanoparticles of conjugated polymers. Chem. Rev. 110, 6260 (2010).
P.K. Smith, R.I. Krohn, G.T. Hermanson, A.K. Mallia, F.H. Gartner, M.D. Provenzano, E.K. Fujimoto, N.M. Goeke, B.J. Olson, and D.C. Klenk: Measurement of protein using bicinchoninic acid. Anal Biochem. 150, 76 (1985).
E. Zucchetti, M. Zangoli, I. Bargigia, C. Bossio, F. Di Maria, G. Barbarella, C. D’Andrea, G. Lanzani, and M.R. Antognazza: Poly(3-hexylthiophene) nanoparticles for biophotonics: study of the mutual interaction with living cells. J. Mater. Chem. B 5, 565 (2017).
A. Ambrosone, L. Mattera, V. Marchesano, A. Quarta, A.S. Susha, A. Tino, A.L. Rogach, and C. Tortiglione: Mechanisms underlying toxicity induced by CdTe quantum dots determined in an invertebrate model organism. Biomaterials 33, 1991 (2012).
A.A.V. Marchesano, J. Bartelmess, F. Strisciante, A. Tino, L. Echegoyen, C. Tortiglione, and S. Giordani: Impact of carbon nano-onions on hydra vulgaris as a Model organism for nanoecotoxicology. Nanomaterials 5, 1331 (2015).
A. Ambrosone and C. Tortiglione: Methodological approaches for nanotoxicology using cnidarian models. Toxicol. Mech. Methods 23, 207 (2013).
E. Mosconi, P. Salvatori, M.I. Saba, A. Mattoni, S. Bellani, F. Bruni, B. Santiago Gonzalez, M.R. Antognazza, S. Brovelli, G. Lanzani, H. Li, J.-L. Brédas, and F. De Angelis: Surface polarization drives photoinduced charge separation at the P3HT/water interface. ACS Energy Lett. 1, 454 (2016).
A. Lewinska, M. Wnuk, E. Slota, and G. Bartosz: Total anti-oxidant capacity of cell culture media. Clin. Exp. Pharmacol. Physiol. 34, 781 (2007).
I.S. Young: Measurement of total antioxidant capacity. J. Clin. Pathol. 54, 339 (2001).
A. Valavanidis, T. Vlachogianni, and C. Fiotakis: 8-hydroxy-2′-deoxyguanosine (8-OHdG): a critical biomarker of oxidative stress and carcinogenesis. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 27, 120 (2009).
V. Rajendran, M. Lehnig, and C.M. Niemeyer: Photocatalytic activity of colloidal CdS nanoparticles with different capping ligands. J. Mater. Chem. 19, 6348 (2009).
N. Waiskopf, Y. Ben-Shahar, M. Galchenko, I. Carmel, G. Moshitzky, H. Soreq, and U. Banin: Photocatalytic reactive oxygen species formation by semiconductor-metal hybrid nanoparticles. Toward light-induced modulation of biological processes. Nano Lett. 16, 4266 (2016).
J. Ge, M. Lan, B. Zhou, W. Liu, L. Guo, H. Wang, Q. Jia, G. Niu, X. Huang, H. Zhou, X. Meng, P. Wang, C.S. Lee, W. Zhang, and X. Han: A graphene quantum dot photodynamic therapy agent with high singlet oxygen generation. Nat. Commun. 5, 4596 (2014).
B. Poljsak, D. Suput, and I. Milisav: Achieving the Balance between ROS and antioxidants: when to use the synthetic antioxidants. Oxid. Med. Cell. Longev. 2013, 956792 (2013).
M. Schieber and N.S. Chandel: ROS function in redox signaling and oxidative stress. Curr. Biol. 24, R453 (2014).
Acknowledgment
Technical assistance of Giuseppe Marino and Ilaria Panico (ISASI-CNR, Pozzuoli) for animal culturing is gratefully acknowledged. We also thank for technical assistance in animal irradiation using white light LED source Dr. Massimo Rippa (ISASI-CNR, Pozzuoli). MM acknowledges the European Union’s Horizon 2020 research and innovation programme (Marie Skiodowska-Curie grant agreement No. 660228).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Supplementary material
Supplementary material
The supplementary material for this article can be found at {rs|https://doi.org/10.1557/mrc.2018.104|url|}.
Rights and permissions
About this article
Cite this article
Moros, M., Lewinska, A., Onorato, G. et al. Light-triggered modulation of cell antioxidant defense by polymer semiconducting nanoparticles in a model organism. MRS Communications 8, 918–925 (2018). https://doi.org/10.1557/mrc.2018.104
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/mrc.2018.104