Summary
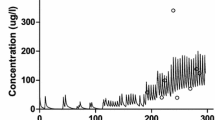
The plasma concentrations and renal clearance values of morphine, morphine-3-glucuronide (M3G) and morphine-6-glucuronide (M6G) were determined in 11 adult cancer patients maintained on a long term oral morphine dosage (10 to 100mg every 4h). Concentrations in plasma and urine were determined by a specific high performance liquid chromatography assay. In this group of patients, whose creatinine clearance values ranged from 52 to 180 ml/min (3.12 to 10.8 L/h), average steady-state plasma concentrations of morphine, M3G and M6G were related (p < 0.01) to the morphine dose per kilogram of bodyweight. The mean total urinary recovery as morphine, M3G and M6G was 74.6 ± 26.5% of the dose. Renal clearance values for M3G and M6G were closely related (r2 = 0.80; p < 0.0005). It was not possible to detect a relationship between the renal clearance of morphine, M3G and M6G, and that of creatinine. The renal tubular handling of all 3 compounds showed wide interindividual variation, and there was evidence of either net renal tubular secretion or reabsorption. There was no apparent relationship between plasma morphine and M6G concentrations and pain relief.
Similar content being viewed by others
References
Abbott FV, Palmour EM. Morphine-6-glucuronide: analgesic effects and receptor binding profile in rats. Life Sciences 43: 1685–1695, 1988
Bodd E, Jacobsen D, Lund E, Ripel Å, Mørland J, et al. Morphinc-6-glucuronide might mediate the prolonged opioid effect of morphine in acute renal failure. Human and Experimental Toxicology 9: 317–321, 1990
Chen ZR, Irvine RJ, Somogyi AA, Bochner F. Mu receptor binding of some commonly used opioids and their metabolites. Life Sciences 48: 2165–2171, 1991
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 16: 31–41, 1976
Glare PA, Walsh TD. Clinical pharmacokinetics of morphine. Therapeutic Drug Monitoring 13: 1–23, 1991
Hasselström J, Berg U, Löfgren A, Säwe J. Long lasting respiratory depression induced by morphine-6-glucuronide. British Journal of Clinical Pharmacology 27: 515–518, 1989
Inturrisi CE. Effects of other drugs and pathologic states on opioid disposition and response. Advances in Pain Research and Therapy 14: 171–180, 1990
Jensen MP, Karoly P, Braver S. The measurement of clinical pain intensity: a comparison of six methods. Pain 27: 117–126, 1986
May DG, Fujimoto JM, Inturrisi CE. The tubular transport and metabolism of morphine-N-methyl-C14 by the chicken kidney. Journal of Pharmacology and Experimental Therapeutics 157: 626–635, 1967
McQuay HJ, Carroll D, Faura CC, Gavaghan DJ, Hand CW, et al. Oral morphine in cancer pain: influences on morphine and metabolite concentration. Clinical Pharmacology and Therapeutics 48: 236–244, 1990
Milne RW, Nation RL, Reynolds GD, Somogyi AA, van Crugten JT. High-performance liquid Chromatographic determination of morphine and its 3- and 6-glucuronide metabolites: improvements to the method and application to stability studies. Journal of Chromatography 565: 457–464, 1991
Milne RW, Nation RL, Somogyi AA, Bochner F, Griggs W. The influence of renal function on the renal clearance of morphine and its glucuronide metabolites in intensive-care patients. British Journal of Clinical Pharmacology 34: 53–59, 1992
Muirhead M, Bochner F, Somogyi A. Pharmacokinetic drug interactions between triamterene and ranitidine in humans: alterations in renal and hepatic clearances and gastrointestinal absorption. Journal of Pharmacology and Experimental Therapeutics 244: 734–739, 1988
Neumann PB, Henriksén H, Grosman N, Christensen CB. Plasma morphine concentrations during chronic oral administration in patients with cancer pain. Pain 13: 247–252, 1982
Olsen GD. Morphine binding to human plasma proteins. Clinical Pharmacology and Therapeutics 17: 31–35, 1975
Olsen GD, Bennett WM, Porter GA. Morphine and phenytoin binding to plasma proteins in renal and hepatic failure. Clinical Pharmacology and Therapeutics 17, 677–684, 1975
Osborne RJ, Joel SP, Slevin ML. Morphine intoxication in renal failure: the role of morphine-6-glucuronide. British Medical Journal 292: 1548–1549, 1986
Osborne RJ, Joel SP, Trew D, Slevin ML. The analgesic activity of morphine-6-glucuronide. Lancet 1: 828, 1988
Osborne RJ, Joel SP, Trew D, Slevin ML. Morphine and metabolite behaviour after different routes of administration: demonstration of the importance of the active metabolite morphine-6-glucuronide. Clinical Pharmacology and Therapeutics 47: 12–19, 1990
Osborne R, Thompson P, Joel S, Trew D, Patel N, et al. The analgesic activity of morphine-6-glucuronide. British Journal of Clinical Pharmacology 34: 130–138, 1992
Pasternak GW, Bodnar RJ, Clark JA, Inturrisi CE. Morphine-6-glucuronide, a potent mu agonist. Life Sciences 41: 2845–2849, 1987
Paul D, Standifer KM, Inturrisi CE, Pasternak GW. Pharmacological characterization of morphine-6β-glucuronide, a very potent morphine metabolite. Journal of Pharmacology and Experimental Therapeutics 251: 477–483, 1989
Peterson GM. Randall CTC, Paterson J. Plasma levels of morphine and morphine glucuronides in the treatment of cancer pain: relationship to renal function and route of administration. European Journal of Clinical Pharmacology 38: 121–124, 1990
Poulain P, Hoskin PJ, Hanks GW. A-Omar O, Walker VA, et al. Relative bioavailability of controlled release morphine tablets (MST Continus) in cancer patients. British Journal of Anaesthesia 61: 569–574, 1988
Säwe J. Morphine and its 3- and 6-glucuronides in plasma and urine during chronic oral administration in cancer patients. Advances in Pain Research and Therapy 8: 45–55, 1986
Säwe J, Kager L, Svensson Eng J-O, Rane A. Oral morphine in cancer patients: in vivo kinetics and in vitro hepatic glucuronidation. British Journal of Clinical Pharmacology 19: 495–501, 1985
Säwe J, Svensson JO, Rane A. Morphine metabolism in cancer patients on increasing oral doses — no evidence for autoinduction or dose-dependence. British Journal of Clinical Pharmacology 16: 85–93, 1983
Schali C, Roch-Ramel F. Transport and metabolism of [3H]morphine in isolated, nonperfused proximal tubular segments of the rabbit kidney. Journal of Pharmacology and Experimental Therapeutics 223: 811–815, 1982
Shimomura K, Kamata O, Ueki S, Ida S, Yoshimura H, et al. Analgesic effect of morphine glucuronides. Tohoku Journal of Experimental Medicine 105: 45–52, 1971
Smith MT, Watt JA, Cramond T. Morphine-3-glucuronide — a potent antagonist of morphine analgesia. Life Sciences 47: 579–585, 1990
Takeda F. WHO cancer pain relief programme. In Bond et al. (Eds) Proceedings of the VIth World Congress on Pain, pp. 467–474, Elsevier, Amsterdam, 1991
van Crugten JT, Sallustio BC, Nation RL, Somogyi AA. Renal tubular transport of morphine, morphine-6-glucuronide and morphine-3-glucuronide in the isolated perfused rat kidney. Drug Metabolism and Disposition 19: 1087–1092, 1991a
van Crugten JT, Somogyi AA, Nation RL. Kinetic-dynamic relationships of morphine (M) and morphine-6-glucuronide (M6G) in the rat. Clinical and Experimental Pharmacology and Physiology (Suppl. 18): 63, 1991b
Watt JA, Cramond T, Smith MT. Morphine-6-glucuronide: analgesic effects antagonized by morphine-3-glucuronide. Clinical and Experimental Pharmacology and Physiology (Suppl. 17): 83, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Somogyi, A.A., Nation, R.L., Olweny, C. et al. Plasma Concentrations and Renal Clearance of Morphine, Morphine-3-Glucuronide and Morphine-6-Glucuronide in Cancer Patients Receiving Morphine. Clin. Pharmacokinet. 24, 413–420 (1993). https://doi.org/10.2165/00003088-199324050-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199324050-00005